Advanced Synthesis of 3-Hydroxypicolinic Acid Derivatives for Commercial Agrochemical Production
The agricultural chemical industry continuously demands novel scaffolds to combat evolving fungal resistance, and patent CN1411445A presents a significant breakthrough in the preparation of 3-hydroxypicolinic acid derivatives. This intellectual property outlines a robust synthetic methodology that enables the precise construction of highly substituted pyridine cores, which are critical for next-generation fungicidal activity. Unlike previous methods restricted to natural extraction, this approach allows for extensive structural diversification at the 4-position, opening new avenues for structure-activity relationship studies. For R&D directors seeking high-purity agrochemical intermediates, this technology offers a reliable pathway to access chemical space previously difficult to explore. The general structure defined in this patent serves as a versatile platform for developing potent crop protection agents. 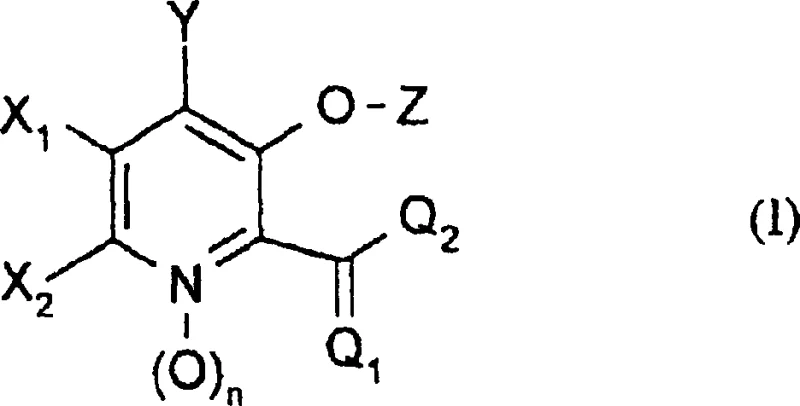
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the availability of 3-hydroxypicolinic acid derivatives was severely constrained by reliance on natural sources and fermentation processes. Prior art, such as the publications by Kuzo Shibata, indicated that obtaining compounds substituted at the 4-position was virtually impossible using traditional biological methods. These conventional routes often suffered from low yields, inconsistent batch-to-batch quality, and an inability to introduce specific functional groups required for optimized biological performance. Furthermore, the purification of natural extracts often involved complex chromatography steps that were not economically viable for large-scale commercial production. This lack of synthetic flexibility hindered the rapid development of new fungicides needed to address resistant pathogen strains in modern agriculture. Consequently, supply chains were vulnerable to biological variability and seasonal fluctuations inherent in fermentation-based sourcing.
The Novel Approach
The methodology disclosed in CN1411445A revolutionizes this landscape by introducing a fully synthetic route that bypasses the limitations of natural fermentation. By utilizing pyridine N-oxide precursors, the process enables the strategic introduction of substituents at the 4-position through nucleophilic aromatic substitution mechanisms. This chemical strategy provides unparalleled control over the molecular architecture, allowing chemists to tailor the electronic and steric properties of the final active ingredient. The ability to systematically vary groups such as halogens, alkoxy, and amino functionalities ensures that procurement managers can source materials with consistent specifications. This shift from biological extraction to chemical synthesis represents a fundamental improvement in cost reduction in agrochemical manufacturing, as it eliminates the unpredictability associated with biological systems. The result is a more stable and predictable supply of critical intermediates for the global crop protection market.
Mechanistic Insights into N-Oxide Activated Cyclization
The core innovation of this patent lies in the activation of the pyridine ring via N-oxidation, which significantly enhances the electrophilicity of the carbon atoms adjacent to the nitrogen. 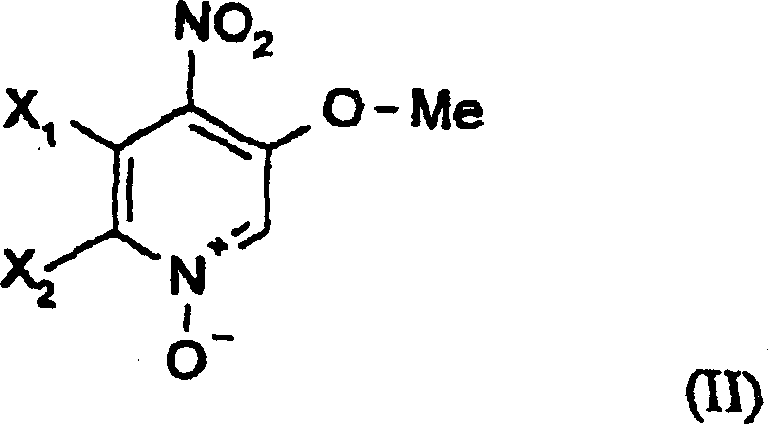 This electronic activation facilitates the subsequent cyanation step, where a cyano group is introduced to form the 2-cyano-3-methoxy-4-nitropyridine intermediate. The mechanism proceeds through a nucleophilic attack on the activated ring system, followed by elimination of the leaving group, driven by the strong electron-withdrawing nature of the N-oxide moiety. For technical teams, understanding this mechanistic nuance is crucial for optimizing reaction parameters such as temperature and solvent polarity to maximize conversion rates. The careful control of these conditions ensures that the desired regioisomer is formed exclusively, minimizing the generation of difficult-to-remove side products. This level of mechanistic precision is essential for maintaining the high purity standards required by regulatory bodies for agrochemical registration.
This electronic activation facilitates the subsequent cyanation step, where a cyano group is introduced to form the 2-cyano-3-methoxy-4-nitropyridine intermediate. The mechanism proceeds through a nucleophilic attack on the activated ring system, followed by elimination of the leaving group, driven by the strong electron-withdrawing nature of the N-oxide moiety. For technical teams, understanding this mechanistic nuance is crucial for optimizing reaction parameters such as temperature and solvent polarity to maximize conversion rates. The careful control of these conditions ensures that the desired regioisomer is formed exclusively, minimizing the generation of difficult-to-remove side products. This level of mechanistic precision is essential for maintaining the high purity standards required by regulatory bodies for agrochemical registration.
Following the initial functionalization, the process involves a critical halogenation step that further prepares the molecule for downstream derivatization. 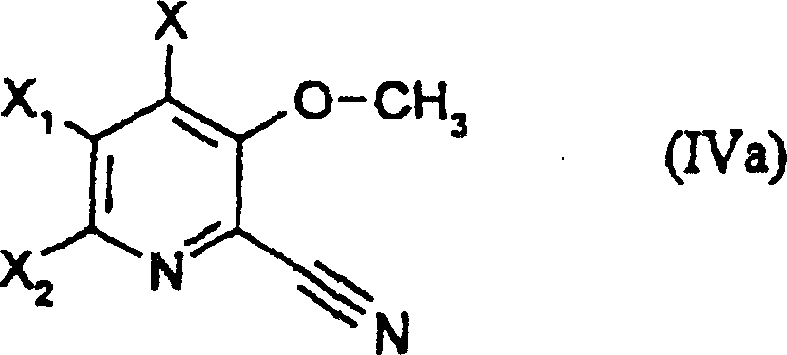 The conversion of the nitro group to a halogen atom, typically bromine or chlorine, creates a versatile handle for subsequent cross-coupling or nucleophilic displacement reactions. This transformation is achieved under controlled thermal conditions using acyl halides, which act as both reagents and dehydrating agents to drive the reaction to completion. The resulting halogenated intermediate is remarkably stable yet reactive enough to undergo hydrolysis to the final acid without compromising the integrity of the sensitive 3-hydroxy group. This sequence demonstrates a sophisticated understanding of protecting group chemistry and orthogonal reactivity, ensuring that the final 3-hydroxypicolinic acid scaffold is obtained with minimal impurity burden. Such robust chemistry is vital for scaling up production while maintaining stringent quality control metrics.
The conversion of the nitro group to a halogen atom, typically bromine or chlorine, creates a versatile handle for subsequent cross-coupling or nucleophilic displacement reactions. This transformation is achieved under controlled thermal conditions using acyl halides, which act as both reagents and dehydrating agents to drive the reaction to completion. The resulting halogenated intermediate is remarkably stable yet reactive enough to undergo hydrolysis to the final acid without compromising the integrity of the sensitive 3-hydroxy group. This sequence demonstrates a sophisticated understanding of protecting group chemistry and orthogonal reactivity, ensuring that the final 3-hydroxypicolinic acid scaffold is obtained with minimal impurity burden. Such robust chemistry is vital for scaling up production while maintaining stringent quality control metrics.
How to Synthesize 3-Hydroxypicolinic Acid Derivatives Efficiently
The synthesis of these valuable intermediates follows a logical progression of activation, functionalization, and hydrolysis steps designed for operational simplicity and high yield. The detailed standardized synthesis steps see the guide below, which outlines the specific reagents, stoichiometry, and workup procedures required to replicate the patent examples successfully. Implementing this route requires careful attention to the handling of cyanide sources and the management of exothermic events during the N-oxide formation. However, the use of common organic solvents such as dichloroethane and dimethylformamide ensures that the process can be integrated into existing manufacturing infrastructure with minimal capital expenditure. By adhering to the specified reaction times and temperatures, production teams can achieve consistent results that meet the rigorous demands of the agrochemical supply chain.
- Activation of pyridine N-oxide precursors followed by cyanation to establish the core nitrile functionality.
- Halogenation of the nitro-cyano intermediate using acyl halides to introduce reactive handles at the 4-position.
- Hydrolysis of the nitrile group under acidic or basic conditions to yield the final 3-hydroxypicolinic acid scaffold.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this synthetic methodology offers substantial strategic benefits beyond mere technical feasibility. The elimination of fermentation-dependent steps removes a significant bottleneck in the production timeline, leading to drastically simplified logistics and reduced lead time for high-purity agrochemical intermediates. Since the process relies on commodity chemicals and standard unit operations, it mitigates the risk of supply disruptions caused by biological contamination or raw material scarcity. This reliability is paramount for maintaining continuous manufacturing schedules for finished fungicide formulations. Furthermore, the synthetic route allows for the optimization of atom economy, which translates into significant cost savings in waste treatment and raw material consumption. These factors collectively enhance the overall resilience of the supply chain against market volatility.
- Cost Reduction in Manufacturing: The synthetic pathway described eliminates the need for expensive enzymatic catalysts or complex extraction protocols associated with natural product isolation. By utilizing readily available chemical reagents and avoiding the high overhead of sterile fermentation facilities, the overall cost of goods sold is significantly lowered. The process efficiency is further enhanced by the high yields reported in the patent examples, which minimize the loss of valuable starting materials. Additionally, the ability to recycle solvents and recover byproducts contributes to a more sustainable and economically viable production model. These cumulative efficiencies allow for competitive pricing structures without compromising on the quality of the final intermediate.
- Enhanced Supply Chain Reliability: Chemical synthesis offers a level of predictability that biological processes cannot match, ensuring that delivery schedules are met with greater consistency. The independence from agricultural cycles or microbial culture health means that production can be ramped up or down based on market demand without long lag times. This flexibility is crucial for responding to sudden outbreaks of crop diseases that require immediate deployment of fungicidal solutions. Moreover, the stability of the intermediates allows for safer storage and transportation, reducing the risk of degradation during transit. This reliability strengthens the partnership between suppliers and formulators, fostering long-term strategic alliances.
- Scalability and Environmental Compliance: The reaction conditions employed are amenable to scale-up from kilogram to multi-ton quantities using standard stainless steel reactors. The process avoids the use of heavy metal catalysts that often require costly removal steps and generate hazardous waste streams. Instead, the byproducts are primarily inorganic salts that can be treated using conventional wastewater management systems. This alignment with green chemistry principles simplifies regulatory compliance and reduces the environmental footprint of the manufacturing site. As global regulations on agrochemical production become stricter, adopting such environmentally benign processes provides a distinct competitive advantage in the marketplace.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology in industrial settings. These answers are derived directly from the experimental data and claims presented in the original intellectual property documentation. Understanding these nuances helps stakeholders make informed decisions about integrating these intermediates into their development pipelines. The clarity provided here aims to bridge the gap between laboratory research and commercial application, ensuring smooth technology transfer.
Q: What are the primary advantages of this synthetic route over fermentation?
A: This synthetic method overcomes the limitations of natural fermentation by allowing precise substitution at the 4-position of the pyridine ring, which is not achievable through traditional biological extraction methods.
Q: How does the N-oxide chemistry improve impurity control?
A: The use of pyridine N-oxides activates the ring for nucleophilic substitution, enabling milder reaction conditions that reduce the formation of thermal degradation byproducts and simplify downstream purification.
Q: Is this process scalable for industrial fungicide production?
A: Yes, the methodology utilizes standard chemical reagents and solvent systems compatible with large-scale reactor setups, ensuring consistent quality and supply continuity for commercial agrochemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxypicolinic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to bring this advanced synthetic route to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-hydroxypicolinic acid derivatives meets the highest industry standards. Our commitment to quality assurance means that you can rely on us as a consistent source of high-performance agrochemical intermediates for your formulation development. We understand the critical nature of supply continuity in the crop protection sector and are dedicated to being a partner you can trust.
We invite you to contact our technical procurement team to discuss how we can support your specific requirements with a Customized Cost-Saving Analysis. By collaborating with us, you gain access to specific COA data and route feasibility assessments tailored to your project timeline. Let us help you optimize your supply chain and accelerate the development of your next-generation fungicide products. Reach out today to explore the possibilities of this innovative chemistry and secure a competitive edge in the global agrochemical market.
