Scalable Production of 3,5-Dichloro-2-Pentanone via Solvent-Free Chlorination for Global Markets
Introduction to Advanced Intermediate Synthesis
The chemical industry is constantly evolving towards more sustainable and cost-effective manufacturing processes, particularly for high-value intermediates like 3,5-dichloro-2-pentanone. As detailed in patent CN114685253A, a groundbreaking preparation method has been developed that fundamentally shifts the paradigm from traditional sulfonyl chloride-based routes to a more efficient chlorine gas-mediated pathway. This innovation addresses critical pain points in the supply chain of prothioconazole, a vital fungicide, by streamlining the synthetic route into a cohesive one-pot operation. The significance of this technology lies not only in its chemical elegance but also in its potential to redefine cost structures for reliable agrochemical intermediate suppliers globally. By eliminating the need for expensive sulfonating agents and complex solvent systems, this method offers a robust solution for large-scale production.
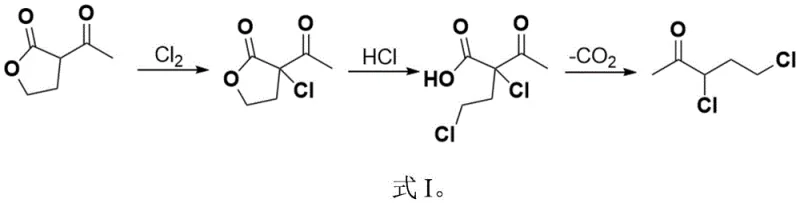
The core of this technological advancement involves the direct chlorination of alpha-acetyl-gamma-butyrolactone using elemental chlorine under strictly solvent-free conditions. This initial step is crucial as it sets the stage for a cascade of reactions including ring-opening and decarboxylation, all managed within a unified reaction vessel. The ability to perform these transformations without isolating unstable intermediates significantly reduces processing time and material loss. Furthermore, the process ingeniously utilizes the byproduct hydrogen chloride generated in the first step for the subsequent acid-catalyzed reactions, creating a closed-loop system that minimizes waste. This holistic approach ensures that the production of high-purity 3,5-dichloro-2-pentanone is not only chemically feasible but also economically superior to legacy methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,5-dichloro-2-pentanone has relied heavily on sulfonyl chloride as the chlorinating agent, a method fraught with significant operational and environmental drawbacks. Traditional protocols often require the use of glacial acetic acid or other organic solvents to facilitate the reaction, which subsequently necessitates energy-intensive distillation processes for solvent recovery and product isolation. Moreover, the use of sulfonyl chloride inevitably generates sulfur dioxide as a stoichiometric byproduct, imposing a heavy burden on wastewater treatment facilities and increasing the overall environmental footprint of the manufacturing plant. The formation of substantial amounts of tar during these reactions further complicates purification, often requiring multiple distillation steps that degrade yield and increase energy consumption. These inefficiencies create a bottleneck for commercial scale-up of complex agrochemical intermediates, driving up costs and limiting supply reliability.
The Novel Approach
In stark contrast, the novel approach outlined in the patent data leverages elemental chlorine gas to achieve chlorination under solvent-free conditions, effectively bypassing the generation of sulfur-containing waste. This method allows for the direct conversion of alpha-acetyl-gamma-butyrolactone into the chlorinated intermediate without the need for external solvents, thereby simplifying the reaction matrix and reducing the volume of waste streams. The process is designed as a telescoped sequence where the crude chlorinated product undergoes immediate ring-opening and decarboxylation upon the addition of catalytic water and hydrogen chloride. This seamless integration of reaction steps eliminates the need for intermediate isolation and purification, drastically cutting down on processing time and equipment requirements. The result is a streamlined workflow that enhances cost reduction in agrochemical intermediate manufacturing while maintaining high product integrity.
Mechanistic Insights into Chlorination and Ring-Opening Dynamics
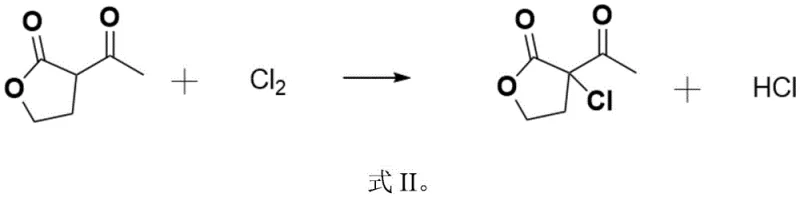
The mechanistic pathway of this synthesis is a sophisticated interplay of electrophilic substitution and acid-catalyzed hydrolysis. The process initiates with the electrophilic chlorination of the alpha-position of the acetyl group in alpha-acetyl-gamma-butyrolactone. Under the optimized conditions of 0 to 60°C, chlorine gas acts as a potent electrophile, selectively substituting the alpha-hydrogen to form alpha-chloro-alpha-acetyl-gamma-butyrolactone. This step is critical as it introduces the first chlorine atom necessary for the final structure while generating hydrogen chloride as a byproduct. The absence of solvent during this phase ensures high concentration of reactants, driving the reaction kinetics forward efficiently. The precise control of temperature and chlorine flow rate is paramount to preventing over-chlorination or degradation of the lactone ring, ensuring the formation of the desired mono-chlorinated intermediate with high selectivity.
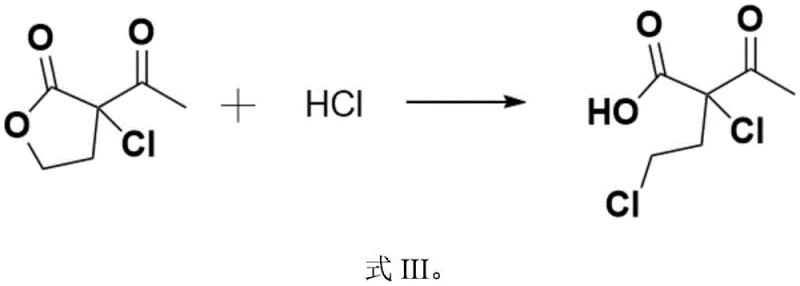
Following the initial chlorination, the reaction system transitions into the ring-opening and decarboxylation phase through the introduction of water and additional hydrogen chloride. The presence of water, even in catalytic amounts, facilitates the hydrolysis of the lactone ring, while the acidic environment provided by HCl promotes the cleavage of the ester bond. This ring-opening event exposes the carboxylic acid functionality, which is inherently unstable under the heated conditions employed in the subsequent step. As the temperature is raised to the range of 40 to 120°C, the beta-keto acid intermediate undergoes thermal decarboxylation, releasing carbon dioxide and yielding the final 3,5-dichloro-2-pentanone skeleton. The second chlorine atom is introduced during the ring-opening phase, likely via an SN2-type displacement or acid-catalyzed substitution on the hydroxyalkyl chain, completing the dichloro motif essential for the biological activity of the downstream fungicide.
How to Synthesize 3,5-Dichloro-2-Pentanone Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and purity. The protocol begins with the careful introduction of chlorine gas into the starting material at low temperatures to manage exothermicity, followed by a controlled heating ramp to facilitate the cascade of ring-opening and decarboxylation reactions. The beauty of this method lies in its simplicity; by avoiding the use of Lewis acids or phase transfer catalysts, the workup procedure is reduced to a simple phase separation and distillation. The aqueous phase separated after the reaction contains minimal organic residue and can be recycled for subsequent batches, further enhancing the sustainability of the process. For detailed operational parameters and safety guidelines regarding the handling of chlorine gas and hydrogen chloride, operators should refer to standard industrial safety protocols.
- Perform chlorination of alpha-acetyl-gamma-butyrolactone with chlorine gas under solvent-free conditions at 0-60°C to obtain alpha-chloro-alpha-acetyl-gamma-butyrolactone.
- Add catalytic water and introduce hydrogen chloride to the crude product for ring-opening and chlorination at 20-60°C.
- Heat the system to 40-120°C to induce decarboxylation, followed by phase separation and vacuum distillation to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route presents a compelling value proposition centered on cost efficiency and operational reliability. The substitution of expensive sulfonyl chloride with commodity chlorine gas results in a drastic reduction in raw material expenditure, directly impacting the bottom line of production costs. Furthermore, the elimination of organic solvents in the initial chlorination step removes the logistical and financial burden associated with solvent procurement, storage, and recovery. This simplification of the bill of materials translates into a more resilient supply chain, less susceptible to fluctuations in solvent markets. The one-pot nature of the reaction also implies a shorter cycle time per batch, allowing manufacturers to respond more agilely to market demand spikes for prothioconazole intermediates without requiring significant capital investment in new reactor capacity.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of high-cost sulfonating agents with inexpensive chlorine gas, which is widely available and cost-effective. Additionally, the avoidance of acetic acid as a solvent eliminates the energy costs associated with distilling and recovering large volumes of solvent, leading to substantial utility savings. The reduction in tar formation also means less product is lost to waste, improving the overall mass balance and effective yield of the process. These factors combine to create a significantly leaner cost structure for the production of high-purity agrochemical intermediates.
- Enhanced Supply Chain Reliability: By simplifying the synthetic route and removing dependency on specialized catalysts like Lewis acids or phase transfer agents, the supply chain becomes more robust and less prone to disruptions. The ability to recycle the aqueous phase and utilize byproduct hydrogen chloride internally reduces the need for external chemical inputs, fostering a more self-sufficient production environment. This autonomy ensures consistent production schedules and reliable delivery timelines for downstream customers who depend on a steady flow of intermediates for their own formulation processes. The scalability of the solvent-free method further guarantees that supply can be ramped up quickly to meet global agricultural demands.
- Scalability and Environmental Compliance: From an environmental compliance perspective, this method offers a distinct advantage by completely avoiding the generation of sulfur dioxide, a regulated pollutant that requires costly scrubbing systems. The reduction in wastewater volume and the absence of heavy metal catalysts simplify effluent treatment, lowering the environmental compliance costs for the manufacturing facility. The process is inherently safer and cleaner, aligning with modern green chemistry principles and making it easier to obtain necessary environmental permits for expansion. This environmental stewardship not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a sustainable partner in the global chemical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis method. Understanding these nuances is critical for R&D teams looking to adapt this technology to their existing infrastructure. The answers provided are derived directly from the experimental data and technical disclosures within the patent literature, ensuring accuracy and relevance for industrial application. These insights cover aspects ranging from reaction stoichiometry to byproduct management, offering a comprehensive overview for technical decision-makers.
Q: What are the primary advantages of using chlorine gas over sulfonyl chloride in this synthesis?
A: Using chlorine gas eliminates the generation of sulfur dioxide byproducts and significantly reduces raw material costs compared to sulfonyl chloride methods. It also simplifies the process by avoiding the need for complex waste acid treatment associated with sulfur-containing reagents.
Q: How does the solvent-free condition impact the environmental profile of the manufacturing process?
A: Operating under solvent-free conditions during the initial chlorination step removes the need for organic solvents like acetic acid, thereby reducing VOC emissions and energy consumption required for solvent recovery. This leads to a greener process with lower three-waste discharge.
Q: Can the byproduct hydrogen chloride be recycled within this production method?
A: Yes, the hydrogen chloride generated during the chlorination step can be collected via tail gas trapping and directly reintroduced into the subsequent ring-opening reaction, maximizing atom economy and reducing the need for external acid sources.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Dichloro-2-Pentanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the competitive agrochemical landscape. Our team of expert chemists has extensively evaluated the solvent-free chlorination technology described in CN114685253A and confirmed its viability for large-scale manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to pilot plant is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3,5-dichloro-2-pentanone delivered meets the highest industry standards for prothioconazole synthesis.
We invite global partners to collaborate with us to leverage this advanced manufacturing capability for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage interested parties to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can optimize the production of critical agrochemical intermediates, driving down costs while maintaining the uncompromising quality required for modern crop protection solutions.
