Advanced Synthesis of Substituted Piperazine Intermediates for Commercial Antihistamine Production
Advanced Synthesis of Substituted Piperazine Intermediates for Commercial Antihistamine Production
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for high-volume antihistamine active pharmaceutical ingredients (APIs), particularly second-generation compounds like cetirizine and efletirizine. Patent CN1215395A introduces a significant technological advancement by disclosing novel substituted [2-(1-piperazinyl)ethoxy]methyl compounds that serve as highly versatile precursors in this domain. These intermediates are characterized by a specific structural framework where the piperazine ring is linked via an ethoxy bridge to various functional groups, offering unprecedented flexibility in downstream chemical transformations. By defining specific substituents for R1 and R2, the patent outlines a library of compounds that can be tailored for optimal reactivity and purity, addressing long-standing challenges in the manufacturing of piperazine-based therapeutics. This innovation provides a reliable pharmaceutical intermediate supplier with the necessary chemical tools to enhance production efficiency while maintaining stringent quality standards required for global regulatory compliance.
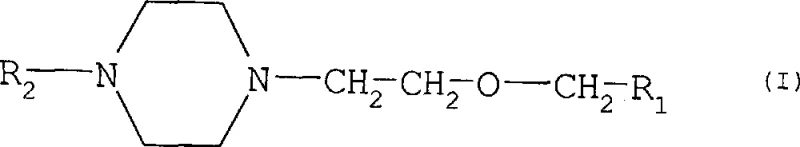
The core innovation lies in the structural versatility of Formula I, where R1 can represent diverse functionalities such as amides, nitriles, carboxylic acids, or esters, and R2 can act as a hydrogen atom or a sophisticated protecting group. This modularity allows chemists to select the optimal precursor based on the specific requirements of the final API synthesis, whether it be cetirizine or its fluorinated analog efletirizine. The ability to introduce protecting groups on the piperazine nitrogen is particularly critical for preventing unwanted side reactions, such as dialkylation, which often plague traditional synthesis routes. Consequently, this patent represents a pivotal shift towards more controlled and predictable chemical manufacturing processes, ensuring that the resulting intermediates possess the high purity profiles necessary for subsequent coupling reactions with diphenylmethyl halides.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of piperazine-based antihistamine intermediates has relied on direct alkylation strategies that frequently encounter significant hurdles regarding selectivity and yield. Traditional methods often involve reacting unsubstituted piperazine directly with halo-ether derivatives, a process that lacks precise control over the degree of substitution. This lack of control frequently results in the formation of di-alkylated byproducts, necessitating complex and costly purification steps to isolate the desired mono-alkylated intermediate. Furthermore, conventional routes described in earlier literature, such as Canadian Patent 1,199,918, often utilize specific starting materials that limit the scope of applicable derivatives, thereby restricting the ability to optimize the process for different analogs. The reliance on harsh reaction conditions or stoichiometric imbalances to drive these reactions can also lead to the degradation of sensitive functional groups, ultimately compromising the overall yield and economic viability of the manufacturing process.
The Novel Approach
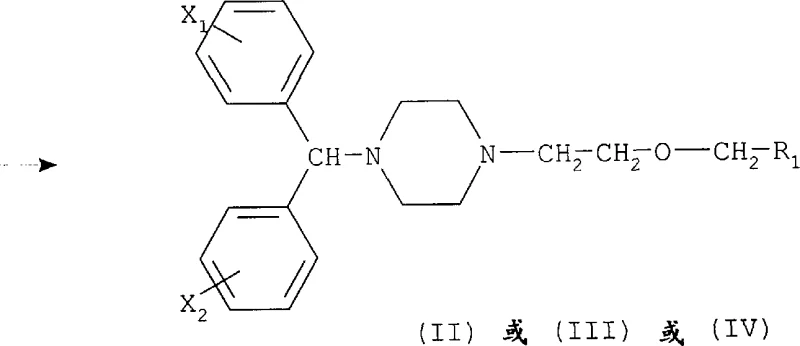
In contrast, the methodology presented in CN1215395A offers a sophisticated solution by introducing pre-functionalized intermediates that streamline the synthetic pathway. By utilizing compounds where the piperazine nitrogen is either unsubstituted or protected with specific groups like carbamates or benzyl derivatives, the new approach ensures high regioselectivity during the alkylation step. This strategic use of protecting groups effectively masks one nitrogen atom, forcing the reaction to occur exclusively at the desired site and virtually eliminating the formation of di-alkylated impurities. Additionally, the patent describes reaction conditions that are milder and more compatible with a wide range of solvents, including toluene, methylethylketone, and acetonitrile. This flexibility not only simplifies the workup procedures but also facilitates easier solvent recovery and recycling, contributing to a more sustainable and cost-effective manufacturing protocol that aligns with modern green chemistry principles.
Mechanistic Insights into Nucleophilic Substitution and Deprotection Strategies
The chemical mechanism underpinning this invention relies on a classic nucleophilic substitution reaction, specifically an SN2 type displacement, where the piperazine nitrogen acts as the nucleophile attacking the electrophilic carbon of the halo-ether moiety. The presence of an acid acceptor, such as sodium carbonate, potassium carbonate, or triethylamine, is crucial in this step to neutralize the hydrogen halide byproduct generated during the reaction, thereby driving the equilibrium towards the formation of the desired ether linkage. The patent details how varying the leaving group (chlorine, bromine, or iodine) and the solvent polarity can significantly influence the reaction kinetics and completion time. For instance, using iodo-derivatives allows the reaction to proceed at lower temperatures, typically below 40°C, which is advantageous for preserving thermally sensitive functional groups on the R1 side chain. This mechanistic understanding allows process chemists to fine-tune reaction parameters to maximize conversion rates while minimizing energy consumption.

Following the initial alkylation, the removal of the protecting group (R2) is a critical mechanistic step that restores the reactivity of the piperazine nitrogen for the final coupling with diphenylmethyl halides. The patent outlines several deprotection strategies depending on the nature of the protecting group employed; for example, carbamate groups can be cleaved via acid hydrolysis or thermal decomposition, while benzyl groups can be removed through catalytic hydrogenation. This versatility in deprotection chemistry is a major advantage, as it allows manufacturers to choose the method that best fits their existing infrastructure and safety protocols. The ability to cleanly remove the protecting group without affecting the integrity of the ether linkage or the R1 functional group ensures that the final intermediate is obtained with exceptional purity. This high level of control over the impurity profile is essential for meeting the rigorous specifications demanded by regulatory bodies for pharmaceutical raw materials.
How to Synthesize Substituted Piperazine Intermediates Efficiently
The synthesis of these valuable intermediates involves a straightforward yet highly controlled sequence of reactions that can be easily adapted for large-scale production. The process begins with the selection of the appropriate piperazine derivative and the corresponding halo-ether component, followed by heating in a suitable solvent system with an acid scavenger. Detailed operational parameters, including temperature ranges from 30°C to 180°C and reaction times spanning a few hours to several days, are provided to ensure optimal yields. The subsequent purification steps typically involve standard techniques such as filtration, solvent evaporation, and silica gel chromatography, which are well-established in industrial settings. For a comprehensive understanding of the specific experimental conditions and stoichiometric ratios required for different derivatives, please refer to the standardized synthesis guide below.
- React a protected or unsubstituted piperazine derivative with a substituted [2-haloethoxy]methyl compound in the presence of an acid acceptor like sodium carbonate or triethylamine.
- Heat the reaction mixture in a suitable solvent such as toluene, methylethylketone, or acetonitrile at temperatures ranging from 30°C to 180°C to facilitate alkylation.
- If a protecting group was utilized on the piperazine nitrogen, perform a deprotection step using acid hydrolysis, catalytic hydrogenation, or thermal cleavage to reveal the reactive amine.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented synthetic route offers substantial strategic benefits that extend beyond mere technical feasibility. The ability to produce high-purity intermediates with fewer purification steps translates directly into reduced manufacturing costs and shorter production cycles. By eliminating the need for complex separation of di-alkylated byproducts, manufacturers can achieve higher throughput and better utilization of reactor capacity. Furthermore, the use of readily available and inexpensive reagents, such as common alkali metal carbonates and standard organic solvents, mitigates the risk of supply chain disruptions associated with specialized or scarce catalysts. This robustness in raw material sourcing ensures a stable and continuous supply of critical intermediates, which is paramount for maintaining the production schedules of high-demand antihistamine medications in the global market.
- Cost Reduction in Manufacturing: The streamlined synthetic pathway significantly lowers the cost of goods sold by minimizing waste generation and reducing the consumption of expensive purification media. The elimination of transition metal catalysts, which often require costly removal steps to meet residual metal limits, further enhances the economic efficiency of the process. Additionally, the high yields reported for various derivatives mean that less raw material is required to produce the same amount of final product, leading to substantial savings in material costs. These cumulative efficiencies allow for a more competitive pricing structure without compromising on the quality or safety of the pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and standard unit operations makes this process highly resilient to external supply shocks. Unlike routes that depend on proprietary reagents or single-source suppliers, this method utilizes a broad base of commercially available starting materials that can be sourced from multiple vendors globally. This diversification of the supply base reduces the risk of bottlenecks and ensures that production can continue uninterrupted even if one supplier faces difficulties. Moreover, the scalability of the process means that production volumes can be rapidly increased to meet surges in demand, providing a reliable buffer against market volatility.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing reaction conditions and equipment that are standard in the fine chemical industry. The absence of hazardous reagents and the generation of benign byproducts simplify waste treatment and disposal, ensuring compliance with increasingly stringent environmental regulations. The ability to recycle solvents and recover reagents further reduces the environmental footprint of the manufacturing operation. This alignment with sustainability goals not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the manufacturing entity, making it a preferred partner for environmentally conscious pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these aspects is crucial for evaluating the feasibility of integrating this route into existing manufacturing workflows.
Q: What are the primary advantages of using protected piperazine intermediates in API synthesis?
A: Using protected piperazine intermediates prevents over-alkylation and ensures regioselectivity during the synthesis of complex antihistamine structures like cetirizine, leading to higher purity profiles and simplified downstream purification processes.
Q: Can this synthetic route be scaled for industrial production of antihistamine APIs?
A: Yes, the described methodology utilizes standard industrial solvents and reagents without requiring exotic catalysts, making it highly suitable for commercial scale-up from kilogram to multi-ton annual production capacities.
Q: How does this patent improve upon previous methods for cetirizine intermediate preparation?
A: This approach introduces versatile intermediates with variable R1 and R2 groups, allowing for more flexible synthetic pathways and potentially milder reaction conditions compared to older direct alkylation methods that often suffered from yield limitations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Piperazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the successful development and commercialization of life-saving medications. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to delivering substituted piperazine compounds that adhere to stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging the advanced synthetic methodologies described in patents like CN1215395A, we can offer customized solutions that optimize both cost and performance for your specific API requirements.
We invite you to collaborate with us to explore how these innovative intermediates can enhance your production efficiency and product quality. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to be your trusted partner in the complex landscape of pharmaceutical manufacturing.
