Advanced Synthetic Routes for High-Purity Piperazine Intermediates in Antihistamine Production
Advanced Synthetic Routes for High-Purity Piperazine Intermediates in Antihistamine Production
The pharmaceutical industry continuously seeks robust and scalable synthetic pathways for high-volume antihistamine active pharmaceutical ingredients (APIs), particularly second-generation compounds like cetirizine and efletirizine. Patent CN1072661C introduces a significant technological advancement by disclosing novel substituted [2-(1-piperazinyl)ethoxy]methyl compounds of Formula I, which serve as versatile and high-purity precursors for these critical therapeutic agents. These intermediates address longstanding challenges in impurity control and process efficiency, offering a streamlined alternative to legacy synthetic methods that often suffer from poor selectivity. By utilizing specific protecting group strategies on the piperazine ring, this innovation enables the production of complex ether-linked piperazines with superior chemical integrity. 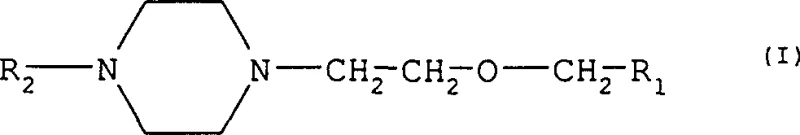 For procurement and supply chain leaders, understanding the structural flexibility of these intermediates is crucial, as the variable R1 and R2 groups allow for the tailored synthesis of various salt forms and derivatives required for different global pharmacopeial standards. This patent represents a pivotal shift towards more predictable and economically viable manufacturing processes for non-sedating antihistamines.
For procurement and supply chain leaders, understanding the structural flexibility of these intermediates is crucial, as the variable R1 and R2 groups allow for the tailored synthesis of various salt forms and derivatives required for different global pharmacopeial standards. This patent represents a pivotal shift towards more predictable and economically viable manufacturing processes for non-sedating antihistamines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for producing cetirizine and related piperazine-based antihistamines have historically relied on the direct alkylation of piperazine derivatives with halo-ether precursors. While conceptually straightforward, these conventional methods frequently encounter significant hurdles regarding regioselectivity and the formation of difficult-to-remove bis-alkylated byproducts. The lack of effective protection on the secondary amine of the piperazine ring often leads to competitive reactions, resulting in complex crude mixtures that require extensive and costly purification steps such as repeated recrystallization or preparative chromatography. Furthermore, older processes described in prior art, such as those found in Canadian Patent 1,199,918, often utilize starting materials that are either expensive or prone to degradation under the harsh reaction conditions necessary to drive the alkylation to completion. These inefficiencies not only inflate the cost of goods sold (COGS) but also introduce variability in the supply chain, posing risks for manufacturers aiming for consistent commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
The methodology outlined in Patent CN1072661C overcomes these deficiencies by introducing a modular synthesis strategy centered on the novel Formula I intermediates. Instead of direct, uncontrolled alkylation, this approach employs protected piperazine derivatives (Formula V) that react selectively with substituted [2-haloethoxy]methyl compounds (Formula VI). This strategic use of protecting groups, such as carbamates or amides on the piperazine nitrogen, effectively masks the reactive site until the desired ether linkage is formed, thereby drastically reducing the formation of unwanted di-substituted impurities. The resulting intermediates are stable, easily isolable solids or oils that can be purified to high standards before undergoing the final coupling step with benzhydryl halides. This decoupling of the ether formation from the final C-N bond formation allows for rigorous quality control at the intermediate stage, ensuring that only high-purity materials enter the final API synthesis, which is a critical factor for reliable pharmaceutical intermediate supplier operations.
Mechanistic Insights into Nucleophilic Substitution and Deprotection
The core chemical transformation in this novel pathway involves a nucleophilic substitution reaction where the nitrogen atom of the protected piperazine acts as the nucleophile attacking the electrophilic carbon of the halo-ether side chain. The reaction is typically conducted in polar aprotic or moderately polar solvents such as methylethylketone (MEK), toluene, or acetonitrile, often in the presence of an acid acceptor like sodium carbonate or triethylamine to neutralize the hydrogen halide byproduct. The choice of solvent and temperature is critical; for instance, when using iodine-containing precursors, lower temperatures below 40°C are preferred to prevent decomposition, whereas chloro-precursors may require reflux conditions to achieve acceptable reaction rates. 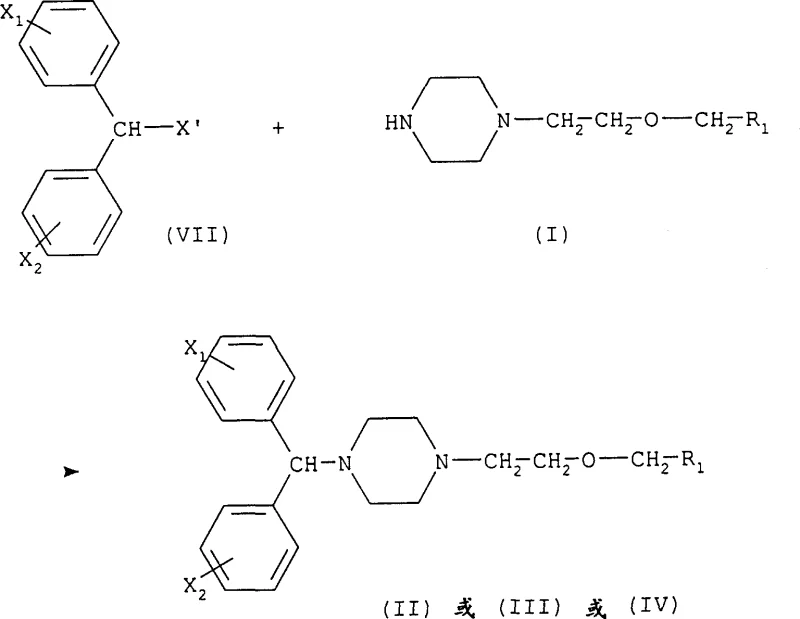 Following the formation of the protected intermediate, the process involves a carefully controlled deprotection step. Depending on the nature of the protecting group (R2), this can be achieved through catalytic hydrogenation, acidic hydrolysis, or thermal treatment. For example, benzyl carbamate groups can be cleaved via hydrogenolysis using palladium on carbon, while tert-butyl esters are removed under acidic conditions. This mechanistic flexibility allows manufacturers to select the most cost-effective and operationally simple deprotection method compatible with their existing infrastructure, thereby facilitating cost reduction in pharmaceutical intermediates manufacturing without compromising on the stringent purity specifications required for human consumption.
Following the formation of the protected intermediate, the process involves a carefully controlled deprotection step. Depending on the nature of the protecting group (R2), this can be achieved through catalytic hydrogenation, acidic hydrolysis, or thermal treatment. For example, benzyl carbamate groups can be cleaved via hydrogenolysis using palladium on carbon, while tert-butyl esters are removed under acidic conditions. This mechanistic flexibility allows manufacturers to select the most cost-effective and operationally simple deprotection method compatible with their existing infrastructure, thereby facilitating cost reduction in pharmaceutical intermediates manufacturing without compromising on the stringent purity specifications required for human consumption.
Furthermore, the subsequent conversion of these intermediates into the final antihistamine precursors (Formula II, III, or IV) proceeds via another nucleophilic substitution, where the free piperazine nitrogen attacks a diphenylmethyl halide (Formula VII). This step benefits immensely from the high purity of the incoming Formula I intermediate, as the absence of competing nucleophiles ensures a clean reaction profile. The ability to isolate the Formula I intermediate also provides a strategic buffer in the supply chain; manufacturers can produce and stockpile this stable intermediate in large batches, decoupling the production timeline from the final API demand fluctuations. This level of process control is essential for maintaining supply continuity in the highly regulated pharmaceutical sector, where any deviation in impurity profiles can lead to batch rejection and significant financial losses.
How to Synthesize Substituted Piperazine Intermediates Efficiently
The synthesis of these high-value intermediates follows a logical progression designed to maximize yield and minimize waste generation. The process begins with the preparation of the protected piperazine, followed by the etherification reaction to install the functionalized side chain. Detailed operational parameters, including specific molar ratios, solvent volumes, and workup procedures, are critical for reproducing the high yields reported in the patent examples. For R&D teams looking to implement this technology, it is essential to note that the reaction conditions are relatively mild, avoiding the need for cryogenic temperatures or high-pressure equipment, which simplifies the engineering requirements for commercial reactors. The standardized synthesis steps见下方的指南 ensure that the process can be transferred smoothly from the laboratory bench to pilot and production scales.
- React a protected piperazine derivative (Formula V) with a substituted [2-haloethoxy]methyl compound (Formula VI) in a solvent like methylethylketone or toluene with an acid acceptor.
- Isolate the novel intermediate (Formula I) through filtration, solvent removal, and purification via silica gel chromatography or crystallization.
- Couple the purified intermediate with a diphenylmethyl halide (Formula VII) under reflux conditions to generate the final antihistamine precursor (Formula II).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits for procurement managers and supply chain directors focused on optimizing the total cost of ownership for antihistamine APIs. The primary advantage lies in the significant simplification of the purification workflow. By preventing the formation of complex byproduct mixtures through the use of protected intermediates, the need for resource-intensive purification techniques is drastically reduced. This translates directly into lower solvent consumption, reduced waste disposal costs, and shorter cycle times, all of which contribute to a more lean and efficient manufacturing operation. Additionally, the starting materials required for this process, such as piperazine derivatives and simple halo-ethers, are commodity chemicals that are readily available from multiple global sources, mitigating the risk of raw material shortages that can plague more specialized synthetic routes.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in solvent usage for purification steps lead to a structurally lower cost base for production. By avoiding the generation of difficult-to-separate impurities, the overall yield of the process is improved, meaning less raw material is required to produce the same amount of final API. This efficiency gain is compounded by the ability to recycle solvents like toluene and methylethylketone, which are used in large volumes during the reaction and workup phases. Consequently, manufacturers can achieve substantial cost savings without sacrificing the quality or safety profile of the final product, making this route highly attractive for generic drug production where margin pressure is intense.
- Enhanced Supply Chain Reliability: The modular nature of this synthesis enhances supply chain resilience by allowing for the stocking of stable intermediates. Unlike unstable reactive species that must be used immediately, the Formula I compounds described in the patent are robust and can be stored for extended periods, providing a buffer against upstream supply disruptions. Furthermore, the reliance on widely available commodity reagents reduces dependency on single-source suppliers for exotic starting materials. This diversification of the supply base ensures that production schedules can be maintained even in the face of geopolitical or logistical challenges, securing the continuous flow of critical medications to the market.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations such as filtration, distillation, and crystallization that are standard in modern chemical plants. The reaction conditions do not require extreme pressures or temperatures, reducing the energy footprint of the manufacturing process. Moreover, the reduced generation of hazardous waste aligns with increasingly stringent environmental regulations, minimizing the regulatory burden on the manufacturing site. This environmental compatibility not only lowers compliance costs but also enhances the corporate sustainability profile of the manufacturer, which is becoming an increasingly important factor in supplier selection criteria for major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthetic technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this route for their specific production needs. The clarity provided here aims to bridge the gap between theoretical patent claims and practical industrial application.
Q: What are the primary advantages of using Formula I intermediates over direct alkylation methods?
A: The use of Formula I intermediates allows for better control over regioselectivity and prevents over-alkylation of the piperazine ring, leading to higher purity profiles and simplified downstream purification compared to traditional direct alkylation routes.
Q: Can this synthetic route be scaled for commercial API manufacturing?
A: Yes, the process utilizes common industrial solvents such as toluene and methylethylketone and operates at moderate temperatures, making it highly amenable to scale-up from kilogram to multi-ton production scales without requiring exotic catalysts.
Q: How does the protecting group strategy impact the overall yield and cost?
A: The strategic use of removable protecting groups on the piperazine nitrogen minimizes side reactions and byproduct formation, which significantly reduces waste generation and improves the effective yield of the valuable final API precursor.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Piperazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic pathways in the competitive landscape of antihistamine production. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative routes described in Patent CN1072661C can be seamlessly integrated into your supply chain. We are committed to delivering high-purity substituted piperazine intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to manage complex deprotection and coupling chemistries allows us to offer a reliable source of these key building blocks, reducing the technical risk associated with in-house development.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this novel intermediate. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your volume needs. Let us collaborate to optimize your antihistamine supply chain with proven, scalable, and cost-effective chemical solutions.
