Advanced Chiral Resolution and Quaternization for High-Purity Cardiovascular Intermediates
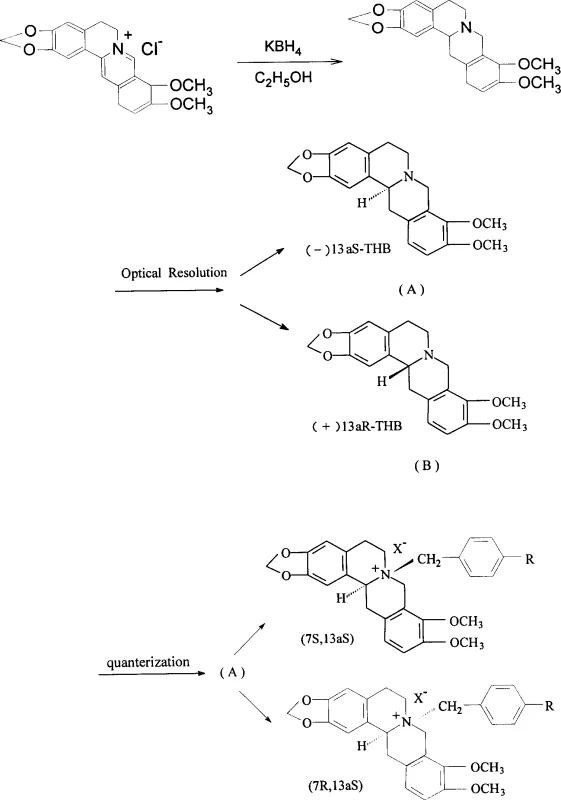
The pharmaceutical industry is constantly seeking methods to enhance the therapeutic index of cardiovascular agents, and patent CN1403456A presents a significant advancement in this domain by detailing the synthesis of N-substituted chiral tetrahydroberberine quaternary ammonium compounds. This technology addresses critical limitations associated with earlier racemic mixtures, specifically the compound known as CPU-86017, by introducing a robust optical resolution strategy followed by precise N-quaternization. The core innovation lies in the ability to isolate specific enantiomers, namely (+)-13aR and (-)-13aS tetrahydroberberine, which demonstrate superior pharmacological profiles including optimized calcium channel blocking activity and reduced intravenous toxicity. For R&D directors and procurement specialists, this patent represents a viable pathway to producing high-value intermediates that offer better bioavailability and safety margins, ultimately facilitating the development of next-generation treatments for conditions such as pulmonary hypertension and heart failure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for protoberberine derivatives often resulted in racemic mixtures that exhibited non-selective biological activity and significant safety concerns, particularly regarding intravenous administration tolerance. The parent compound CPU-86017, while effective in blocking ion channels, suffered from low solubility and high toxicity due to the presence of both optical isomers which interacted differently with biological macromolecules. In many cases, one enantiomer contributed to the desired therapeutic effect while the other exacerbated side effects or contributed to toxicity without adding efficacy, leading to a narrow therapeutic window. Furthermore, conventional methods lacked the specificity to control the stereochemistry at the C13a and N7 centers, resulting in batch-to-batch variability in pharmacological performance. This lack of stereochemical control posed a significant risk for clinical translation, as regulatory bodies increasingly demand single-enantiomer drugs to ensure predictable pharmacokinetics and minimized adverse events in patients suffering from acute arrhythmias.
The Novel Approach
The novel approach outlined in the patent utilizes a chemical resolution technique employing di-p-toluoyl tartaric acid (DPT) to effectively separate the racemic tetrahydroberberine into its constituent enantiomers before the quaternization step. By resolving the precursor first, the synthesis ensures that each subsequent N-substitution reaction yields diastereomers that are easier to separate and characterize, thereby enhancing the overall purity of the final active pharmaceutical ingredient. This method allows for the systematic evaluation of all four possible stereoisomers, identifying specific configurations like the (7S, 13aR) form that possess superior calcium channel blocking activity with reduced hemodynamic side effects. The process is designed to be reproducible and scalable, utilizing common solvents like methanol and acetonitrile, which simplifies the transition from laboratory discovery to commercial manufacturing. This strategic shift from racemic synthesis to chiral resolution fundamentally improves the quality profile of the intermediate, making it a more attractive candidate for downstream drug development.
Mechanistic Insights into Chiral Resolution and Quaternization
The mechanistic foundation of this synthesis relies on the formation of diastereomeric salts between the basic tetrahydroberberine and the chiral resolving agent DPT, which exhibit different solubilities in methanol allowing for fractional crystallization. Once the pure (+)-13aR or (-)-13aS tetrahydroberberine is obtained, it undergoes a nucleophilic substitution reaction where the tertiary amine nitrogen attacks the benzylic carbon of halides such as p-chlorobenzyl chloride. This quaternization step creates a permanent positive charge on the nitrogen atom, which is crucial for the compound's interaction with negatively charged regions of ion channels and receptors. The stereochemistry at the C13a position influences the three-dimensional orientation of the molecule, dictating how well it fits into the binding pockets of L-type calcium channels and alpha-adrenergic receptors. Understanding this structure-activity relationship is vital for R&D teams, as it explains why specific isomers show markedly higher pIC50 values in vasoconstriction assays compared to the racemic mixture.
Impurity control is inherently managed through the resolution step, as the crystallization of the diastereomeric salt selectively precipitates the desired enantiomer while leaving the unwanted isomer in the mother liquor. Following quaternization, the resulting mixture of diastereomers is separated using silica gel column chromatography with a dichloromethane and ethanol gradient, which effectively resolves compounds based on their polarity differences arising from their distinct spatial arrangements. This dual-stage purification strategy ensures that the final product meets stringent purity specifications required for cardiovascular drugs, minimizing the risk of off-target effects caused by trace impurities. The robustness of this chromatographic separation is a key factor in the commercial viability of the process, as it provides a reliable method to consistently produce high-purity material regardless of minor fluctuations in reaction conditions. Consequently, the mechanistic design prioritizes both stereochemical integrity and chemical purity, addressing the two most critical quality attributes for this class of therapeutic intermediates.
How to Synthesize Chiral Tetrahydroberberine Efficiently
The synthesis protocol described in the patent offers a clear and actionable roadmap for producing these high-value chiral intermediates, starting from readily available berberine which is reduced to tetrahydroberberine using potassium borohydride. The detailed procedure involves dissolving the racemic tetrahydroberberine in methanol and adding the chiral resolving agent DPT, followed by a standing period at room temperature to allow for the crystallization of the diastereomeric salt. After isolation and basification to recover the free base, the pure enantiomer is reacted with the appropriate benzyl halide in anhydrous acetonitrile under reflux conditions to drive the quaternization to completion. For a comprehensive understanding of the specific reaction times, temperatures, and workup procedures, please refer to the standardized synthesis steps provided in the guide below which encapsulates the critical operational parameters for successful replication.
- Resolve racemic tetrahydroberberine using di-p-toluoyl tartaric acid (DPT) in methanol to isolate (+)-13aR and (-)-13aS enantiomers.
- React the isolated chiral tetrahydroberberine enantiomers with benzyl halides such as p-chlorobenzyl chloride or p-nitrobenzyl bromide in anhydrous acetonitrile or acetone.
- Purify the resulting quaternary ammonium compounds using silica gel column chromatography with a dichloromethane and ethanol gradient elution system.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented process offers substantial advantages by utilizing widely available starting materials and avoiding the need for expensive transition metal catalysts often required in asymmetric synthesis. The reliance on chemical resolution rather than enzymatic or catalytic asymmetric methods reduces the complexity of the supply chain, as chiral acids like DPT are commodity chemicals with stable pricing and consistent availability globally. This simplification translates directly into cost reduction in cardiovascular drug manufacturing, as it eliminates the need for specialized catalyst recovery systems and reduces the risk of heavy metal contamination which requires additional purification steps. Furthermore, the use of standard solvents such as methanol, acetonitrile, and dichloromethane ensures that the process can be implemented in existing multipurpose chemical plants without requiring significant capital investment in new infrastructure or equipment. These factors collectively enhance the economic feasibility of producing these chiral intermediates at a commercial scale.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the use of straightforward crystallization techniques significantly lower the raw material and processing costs associated with producing chiral intermediates. By avoiding complex asymmetric catalysis, the process reduces the burden on waste treatment systems and minimizes the loss of valuable materials during catalyst removal, leading to a more efficient overall mass balance. The ability to recycle solvents and the high yield of the resolution step further contribute to a leaner manufacturing cost structure, making the final API more competitive in the global market. Additionally, the robustness of the chromatographic purification reduces the likelihood of batch failures, ensuring that production resources are utilized effectively without the need for costly reprocessing.
- Enhanced Supply Chain Reliability: Sourcing strategies are simplified because the key reagents, including berberine and benzyl halides, are produced by multiple suppliers worldwide, mitigating the risk of single-source bottlenecks. The process does not rely on biologically derived enzymes which can suffer from stability issues or supply constraints, thereby ensuring a continuous and predictable flow of materials for production scheduling. This reliability is crucial for maintaining uninterrupted supply to downstream pharmaceutical partners who depend on consistent quality and timely delivery for their own clinical and commercial timelines. The established nature of the chemical transformations involved means that technology transfer to contract manufacturing organizations is straightforward, further diversifying the potential supply base and strengthening supply chain resilience.
- Scalability and Environmental Compliance: The synthesis route is inherently scalable, as the unit operations involved such as crystallization, filtration, and column chromatography are well-understood and easily adapted from kilogram to ton-scale production. The process generates waste streams that are manageable within standard environmental compliance frameworks, avoiding the generation of hazardous heavy metal waste that complicates disposal and increases regulatory overhead. The use of common organic solvents allows for efficient recovery and reuse systems, aligning with green chemistry principles and reducing the overall environmental footprint of the manufacturing operation. This scalability ensures that the supply can grow in tandem with clinical demand, supporting the commercialization of new cardiovascular therapies without facing production capacity constraints.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these chiral tetrahydroberberine derivatives, based on the data provided in the patent documentation. These answers are designed to clarify the specific benefits of the chiral approach over traditional racemic synthesis and to provide insight into the practical aspects of the manufacturing process. Understanding these details is essential for stakeholders evaluating the feasibility of incorporating these intermediates into their drug development pipelines. The information reflects the specific experimental findings and structural data reported in the patent to ensure accuracy and relevance.
Q: What is the primary advantage of using chiral tetrahydroberberine over the racemic mixture?
A: The chiral forms exhibit significantly improved pharmacological selectivity, reduced toxicity upon intravenous administration, and enhanced solubility compared to the racemic CPU-86017 mixture.
Q: Which resolving agent is utilized in this patented synthesis process?
A: The process employs di-p-toluoyl tartaric acid (DPT), specifically using the (-) or (+) enantiomers to effectively separate the tetrahydroberberine racemate into pure optical isomers.
Q: How are the final quaternary ammonium compounds purified?
A: The final products are purified via silica gel column chromatography using a gradient elution of dichloromethane and ethanol, ensuring high purity suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Tetrahydroberberine Supplier
NINGBO INNO PHARMCHEM stands ready to support your development needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply requirements are met with precision and consistency. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of chiral tetrahydroberberine intermediate meets the highest standards required for cardiovascular drug applications. We understand the critical nature of stereochemical purity in this therapeutic area and have optimized our processes to deliver the specific enantiomers needed to maximize therapeutic efficacy and minimize toxicity. Our team of experts is dedicated to providing seamless technology transfer and process optimization services to help you bring your innovative cardiovascular therapies to market faster.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and project timelines. By engaging with us early in your development cycle, you can leverage our expertise to optimize your supply chain and secure specific COA data and route feasibility assessments that will de-risk your project. Let us partner with you to transform this promising patented technology into a commercial reality, ensuring a reliable supply of high-quality intermediates for your next-generation cardiovascular medications.
