Advanced Synthesis of Chiral Isoquinoline Compounds for High-Potency Cardiovascular Therapeutics
Advanced Synthesis of Chiral Isoquinoline Compounds for High-Potency Cardiovascular Therapeutics
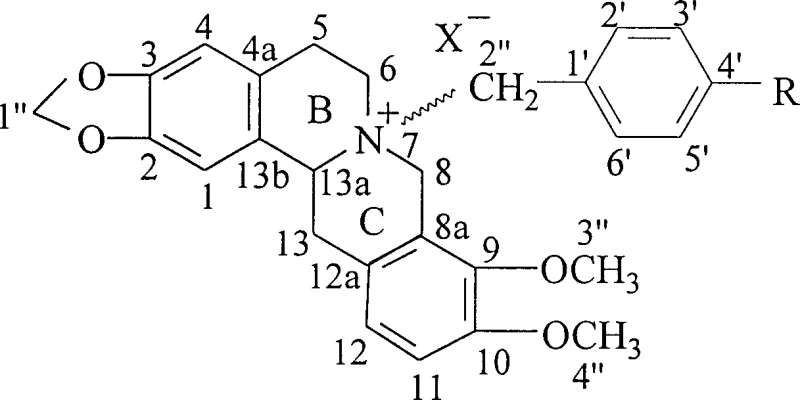
The pharmaceutical landscape for cardiovascular therapeutics is undergoing a significant paradigm shift towards enantiopure intermediates, driven by the need for higher efficacy and reduced off-target toxicity. Patent CN1156474C introduces a groundbreaking methodology for synthesizing N-substituted chiral tetrahydroberberine quaternary ammonium compounds, which serve as critical precursors for next-generation anti-arrhythmic and anti-ischemic agents. Unlike traditional racemic mixtures that often suffer from poor pharmacokinetic profiles, these novel chiral isoquinoline derivatives exhibit optimized activity against L-type calcium channels and alpha-adrenoceptors. The core innovation lies in the precise stereochemical control achieved through di-p-toluoyl tartrate (DPT) resolution, yielding distinct isomers such as (+)-13aR and (-)-13aS tetrahydroberberine. These intermediates are subsequently quaternized to produce a library of compounds (1-12) with tailored substituents, addressing the historical limitations of solubility and bioavailability associated with protoberberine alkaloids. As a reliable pharmaceutical intermediates supplier, understanding the structural nuances of these molecules is essential for developing safer cardiac medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of protoberberine-based cardiovascular drugs, such as the racemic CPU-86017, has been hindered by significant pharmacological drawbacks that limit their clinical utility. The primary issue stems from the presence of multiple chiral centers within the isoquinoline scaffold, where the racemic mixture contains isomers with divergent biological activities, leading to unpredictable therapeutic outcomes. Conventional synthesis routes often fail to distinguish between these stereoisomers, resulting in a final product where the less active or even toxic enantiomer competes with the therapeutic one for receptor binding sites. Furthermore, the parent berberine structure possesses a rigid planar conformation due to the methylenedioxy bridge, which contributes to poor aqueous solubility and low oral bioavailability, necessitating intravenous administration that carries higher risks of acute toxicity. The non-selective blocking of various ion channels by racemic mixtures can cause severe side effects, including drastic changes in blood pressure and heart rate, making tolerance management difficult in emergency care settings. Additionally, the lack of specific substituent optimization in older methods means that the drugs often exhibit strong alpha-blocking effects that are not clinically desirable for all patient populations, thereby restricting their therapeutic window.
The Novel Approach
The methodology outlined in patent CN1156474C represents a sophisticated evolution in synthetic strategy, transitioning from crude racemic production to precise enantiomeric isolation and functionalization. By employing chiral resolving agents like (-)-DPT or (+)-DPT, the process effectively splits the racemic tetrahydroberberine into pure (+)-13aR and (-)-13aS enantiomers, allowing for the independent evaluation and utilization of each isomer's unique pharmacological profile. This resolution step is followed by a targeted N-quaternization reaction with specific benzyl halides, such as p-chlorobenzyl chloride or p-nitrobenzyl bromide, which introduces steric bulk and electronic modifications that significantly enhance water solubility. The resulting quaternary ammonium salts demonstrate improved membrane permeability and receptor selectivity, specifically optimizing the blockade of calcium and potassium channels while minimizing unwanted alpha-adrenergic interference. This approach not only mitigates the toxicity issues associated with intravenous administration but also expands the clinical adaptability of the compounds to treat conditions like pulmonary hypertension and myocardial infarction more effectively. The ability to systematically vary the R and X groups on the N-substituent allows medicinal chemists to fine-tune the lipophilicity and metabolic stability of the final API, ensuring cost reduction in API manufacturing through higher success rates in preclinical trials.
Mechanistic Insights into Chiral Resolution and Quaternization
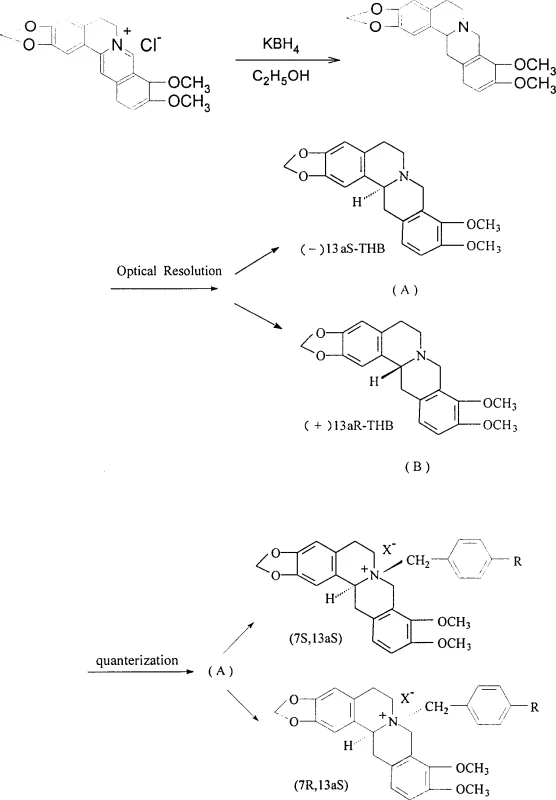
The stereochemical integrity of the final cardiovascular intermediate is established through a classic yet highly effective diastereomeric salt formation mechanism during the resolution phase. When racemic tetrahydroberberine interacts with optically pure di-p-toluoyl tartrate (DPT) in a methanol solvent system, the two enantiomers form diastereomeric salts with distinct physical properties, particularly solubility and crystallization kinetics. The (+)-13aR enantiomer preferentially complexes with the (-)-DPT resolving agent to form a less soluble crystalline salt that precipitates out of the solution, while the (-)-13aS enantiomer remains largely in the mother liquor, allowing for physical separation via filtration. This thermodynamic control is critical because the chiral center at C-13a influences the spatial orientation of the nitrogen lone pair, which subsequently dictates the stereochemical outcome of the quaternization step. Following isolation and alkalization to recover the free base, the enantiopure tetrahydroberberine undergoes nucleophilic substitution at the nitrogen atom. The mechanism involves the attack of the tertiary amine nitrogen on the benzylic carbon of the alkylating agent, proceeding through an SN2-like transition state that inverts the configuration at the nitrogen if it were chiral, but primarily establishes the quaternary ammonium center. The choice of solvent, typically anhydrous acetonitrile or acetone, is crucial to prevent hydrolysis of the alkylating agent and to maintain the solubility of the intermediate species throughout the reflux period.
Impurity control in this synthesis is managed through a combination of selective crystallization and gradient elution chromatography, ensuring that the final product meets stringent purity specifications required for pharmaceutical applications. During the quaternization reaction, potential side products include over-alkylated species or hydrolysis byproducts if moisture is present, which are effectively removed by silica gel column chromatography using a dichloromethane and ethanol gradient system. The separation efficiency relies on the polarity differences between the quaternary ammonium salts and any unreacted tertiary amine precursors, with the more polar quaternary salts eluting later in the gradient. Analytical monitoring via thin-layer chromatography (TLC) allows for the precise identification of fractions containing the desired isomers, such as Compound 1 (7R, 13aR) versus Compound 2 (7S, 13aR), based on their distinct Rf values. This rigorous purification protocol ensures that the impurity profile is minimized, which is vital for reducing the risk of immunogenic reactions or unexpected metabolic interactions in patients. Furthermore, the stability of the methylenedioxy bridge and the dimethoxy groups on the aromatic rings is preserved under the mild reflux conditions employed, preventing degradation of the core isoquinoline scaffold.
How to Synthesize Chiral Tetrahydroberberine Efficiently
The synthesis of these high-value cardiovascular intermediates requires a disciplined approach to reaction conditions and purification to ensure consistent optical purity and yield. The process begins with the reduction of commercially available berberine using potassium borohydride, followed by the critical optical resolution step that defines the stereochemistry of the final product. Operators must maintain strict temperature control during the crystallization of the DPT salts to maximize the enantiomeric excess of the isolated tetrahydroberberine. Subsequent quaternization requires anhydrous conditions to prevent the decomposition of reactive benzyl halides, and the reaction progress should be monitored closely to avoid over-reaction. For detailed operational parameters, safety guidelines, and specific stoichiometric ratios, please refer to the standardized synthesis protocol below.
- Reduce Berberine to Tetrahydroberberine (THB) using potassium borohydride in ethanol, followed by recrystallization to obtain the racemic precursor.
- Perform optical resolution of racemic THB using chiral resolving agents like (-)-DPT or (+)-DPT in methanol to isolate (+)-13aR and (-)-13aS enantiomers.
- React the resolved enantiomers with substituted benzyl halides (e.g., p-chlorobenzyl chloride) in anhydrous acetonitrile under reflux to form the final quaternary ammonium salts.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this chiral synthesis route offers substantial benefits for procurement managers and supply chain directors looking to optimize their API supply chains. The reliance on classical resolution techniques rather than expensive asymmetric catalysis significantly lowers the barrier to entry for production, as it utilizes commodity chemicals like tartaric acid derivatives and common organic solvents that are readily available in the global market. This reduces dependency on specialized catalyst suppliers and mitigates the risk of supply disruptions caused by the scarcity of precious metals or complex ligands. Moreover, the scalability of the process is enhanced by the use of standard unit operations such as reflux, filtration, and column chromatography, which can be easily translated from laboratory scale to multi-ton commercial production without requiring exotic reactor configurations. The improved solubility of the final quaternary ammonium salts also simplifies downstream formulation processes, potentially reducing the need for complex solubilizing excipients and lowering the overall cost of goods sold for the finished drug product. By securing a reliable pharmaceutical intermediates supplier who masters this specific chiral technology, companies can ensure a steady flow of high-quality materials that meet regulatory standards for chiral drugs.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the need for costly and technically challenging heavy metal removal steps, which are often required to meet strict ICH guidelines for residual metals in pharmaceuticals. This simplification of the purification train leads to significant savings in both material costs and processing time, as fewer unit operations are needed to achieve the required purity levels. Additionally, the high selectivity of the resolution process minimizes the formation of hard-to-separate diastereomeric impurities, thereby increasing the overall yield of the desired active isomer and reducing waste disposal costs associated with failed batches. The use of recyclable solvents like acetonitrile and ethanol further contributes to a leaner manufacturing budget, aligning with green chemistry principles that are increasingly valued by stakeholders.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including berberine and substituted benzyl halides, are produced by a robust global chemical industry, ensuring that raw material availability is not a bottleneck for production schedules. Unlike proprietary catalysts that may be sourced from a single vendor, the reagents used in this process are commoditized, allowing procurement teams to negotiate better pricing and secure multiple supply sources to mitigate geopolitical or logistical risks. The robustness of the reaction conditions, which tolerate minor variations in temperature and mixing without compromising product quality, adds another layer of reliability to the manufacturing process, reducing the likelihood of batch failures that could disrupt supply continuity. This stability is crucial for maintaining just-in-time inventory levels and meeting the demanding delivery timelines of downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The process design inherently supports commercial scale-up of complex chiral molecules, as the exothermic nature of the reduction and quaternization steps can be safely managed in large-scale reactors with standard cooling systems. The absence of hazardous reagents like strong Lewis acids or pyrophoric organometallics simplifies the environmental health and safety (EHS) profile of the facility, reducing the regulatory burden and insurance costs associated with handling dangerous chemicals. Waste streams generated during the chromatography and crystallization steps are primarily organic solvents that can be recovered and reused through distillation, minimizing the environmental footprint of the manufacturing site. This alignment with sustainable manufacturing practices not only fulfills corporate social responsibility goals but also future-proofs the supply chain against tightening environmental regulations in key markets like Europe and North America.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these chiral isoquinoline intermediates. Understanding the specific advantages of this patented route helps stakeholders make informed decisions about integrating these materials into their development pipelines. The answers provided are derived directly from the technical specifications and experimental data contained within the patent documentation.
Q: Why is chiral resolution critical for CPU-86017 derivatives?
A: Chiral resolution separates enantiomers that exhibit distinct pharmacological profiles; specific isomers demonstrate superior selectivity for calcium channels and reduced toxicity compared to the racemic mixture.
Q: How does the N-substitution improve drug properties?
A: N-substitution with groups like p-chlorobenzyl enhances water solubility and bioavailability while optimizing the steric configuration for better binding to cardiovascular receptors.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process utilizes standard reflux conditions and common solvents like methanol and acetonitrile, avoiding expensive transition metal catalysts, which facilitates easy commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydroberberine Supplier
As the demand for enantiopure cardiovascular therapeutics continues to rise, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM provides a strategic advantage in bringing these complex molecules to market. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the preclinical phase or preparing for full-scale launch. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee the optical purity and chemical identity of every batch we deliver. Our team of process chemists is adept at optimizing the resolution and quaternization steps described in patent CN1156474C to maximize yield and minimize cost, providing you with a competitive edge in the marketplace.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. By collaborating with us, you gain access to specific COA data and route feasibility assessments that will accelerate your development timeline and reduce technical risk. Let us help you navigate the complexities of chiral synthesis and secure a reliable supply of high-quality intermediates for your next-generation cardiovascular drugs.
