Scalable Synthesis of Chiral Morpholine Derivatives for Pharmaceutical Applications
Scalable Synthesis of Chiral Morpholine Derivatives for Pharmaceutical Applications
The pharmaceutical industry continuously demands robust, scalable, and cost-effective methodologies for constructing complex heterocyclic scaffolds, particularly those containing chiral centers essential for biological activity. Patent CN1642928A represents a significant technological advancement in this domain, disclosing novel processes for the preparation of morpholine derivatives and their intermediates. These compounds, characterized by a substituted morpholine ring linked to various aryl or heteroaryl moieties, serve as critical building blocks in the synthesis of active pharmaceutical ingredients (APIs). The patent outlines multiple synthetic strategies, including chiral pool synthesis using enantiomerically pure epoxides and enzymatic kinetic resolution, offering manufacturers flexible options to optimize yield and purity. By leveraging these innovative routes, producers can overcome the limitations of traditional racemic synthesis followed by difficult resolution steps.
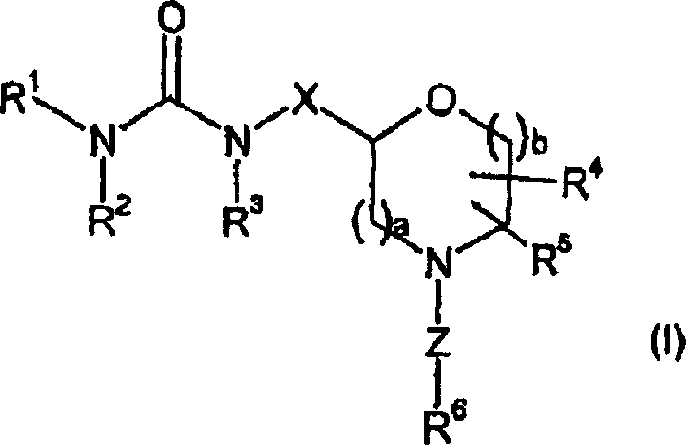
For R&D directors and process chemists, the structural versatility depicted in the general formula is paramount. The ability to vary substituents such as R1, which can range from simple alkyl groups to complex fused heterocycles, allows for the rapid generation of analog libraries during drug discovery. Furthermore, the specific definition of the linker Z and the side chain R6 provides a framework for tuning physicochemical properties like solubility and metabolic stability. This patent does not merely claim a single molecule but protects a broad genus of compounds, ensuring that any commercial entity utilizing these specific substitution patterns must navigate the intellectual property landscape carefully. Understanding the scope of these claims is essential for freedom-to-operate analyses when developing new therapeutic candidates targeting similar biological pathways.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral morpholine derivatives has relied heavily on the resolution of racemic mixtures, a process often fraught with inefficiency and high material loss. Traditional methods frequently employ preparative chiral high-performance liquid chromatography (HPLC) or fractional crystallization of diastereomeric salts. While effective on a laboratory scale, these techniques present substantial bottlenecks when transitioning to commercial manufacturing. Preparative chiral HPLC, for instance, requires expensive stationary phases and large volumes of organic solvents, leading to prohibitive operational costs and environmental burdens. Similarly, diastereomeric salt formation often necessitates multiple recrystallization cycles to achieve acceptable enantiomeric excess, resulting in overall yields that may drop below 40%. Moreover, the reliance on stoichiometric amounts of chiral resolving agents adds to the raw material costs and complicates the downstream purification process, as removing trace amounts of the resolving agent can be challenging.
The Novel Approach
In contrast, the methodology disclosed in CN1642928A introduces a paradigm shift by integrating chiral starting materials and biocatalysis directly into the synthetic route. One key embodiment involves the reaction of an amino-alcohol precursor with a chiral epoxide, such as (S)-2-(oxiran-2-ylmethyl)-1H-isoindole-1,3(2H)-dione. This approach effectively transfers the chirality from the commercially available or easily prepared epoxide to the final morpholine product, bypassing the need for post-synthesis resolution. Another groundbreaking aspect is the use of enzymatic kinetic resolution using lipases, specifically Lipase PS-C. This biocatalytic step operates under mild conditions, typically between 25-35°C, and exhibits high stereoselectivity. By combining chemical synthesis with enzymatic resolution, the process achieves high optical purity without the massive solvent consumption associated with chromatographic methods. This hybrid strategy not only improves the overall mass balance but also aligns with green chemistry principles, making it highly attractive for sustainable manufacturing.
Mechanistic Insights into Chiral Epoxide Ring-Opening and Mitsunobu Coupling
The core chemical transformation in this patent involves the construction of the morpholine ring through the nucleophilic attack of an amino-alcohol on a chiral electrophile. In the Mitsunobu variant, the reaction proceeds via the activation of the hydroxyl group on the amino-alcohol component. Typically, a mixture of the amino-alcohol and the chiral epoxide derivative is stirred in a solvent like tetrahydrofuran or toluene under an inert nitrogen atmosphere. The addition of triphenylphosphine and an azodicarboxylate, such as diisopropyl azodicarboxylate (DIAD), generates a reactive betaine intermediate. This species activates the alcohol, facilitating an intramolecular or intermolecular substitution that closes the ring or attaches the side chain with inversion of configuration. The reaction temperature is carefully controlled, often maintained below 40°C during reagent addition to prevent side reactions, and then warmed to 20-40°C to drive completion. This precise thermal management is crucial for minimizing the formation of regioisomers and ensuring the integrity of the acid-sensitive protecting groups present on the nitrogen atoms.
Alternatively, the direct ring-opening of chiral epoxides by amino-alcohols offers a more atom-economical route. In this mechanism, the nucleophilic nitrogen or oxygen of the amino-alcohol attacks the less hindered carbon of the epoxide ring. The patent highlights the use of (R)-(-)-epichlorohydrin derivatives or protected glycidyl phthalimides as key chiral synthons. The stereochemical outcome is dictated by the configuration of the epoxide and the regioselectivity of the attack. For instance, reacting 2-[(3,4-dichlorobenzyl)amino]ethanol with a chiral epoxide in 1-propanol at 15-25°C allows for the formation of the desired intermediate with high fidelity. The subsequent cyclization to form the morpholine ring can be induced by heating or by adding a base. The presence of the phthalimido group serves a dual purpose: it protects the amine functionality during the harsh reaction conditions and acts as a crystallization handle, facilitating the isolation of the intermediate as a solid rather than an oil, which is a significant advantage for purification.
Impurity control is another critical aspect addressed by the mechanistic understanding of these reactions. Common impurities include over-alkylation products, regioisomers from epoxide opening, and hydrolysis byproducts. The patent specifies rigorous work-up procedures to mitigate these risks. For example, after the Mitsunobu reaction, the mixture is concentrated and treated with alcohols like methanol or isopropanol to precipitate triphenylphosphine oxide, a major byproduct. Filtration at this stage removes the bulk of the phosphorus-containing impurities before the final crystallization. Furthermore, the use of specific solvents like methyl tert-butyl ether (MTBE) or diisopropyl ether as anti-solvents promotes the selective crystallization of the desired product while keeping soluble impurities in the mother liquor. This multi-step purification strategy ensures that the final API intermediate meets stringent purity specifications, often exceeding 98% area/area by HPLC analysis.
How to Synthesize [(2S)-4-(3,4-dichlorobenzyl)morpholin-2-yl]methanamine Efficiently
The synthesis of specific targets like [(2S)-4-(3,4-dichlorobenzyl)morpholin-2-yl]methanamine exemplifies the practical application of the patented technology. The process begins with the coupling of a benzylated amino-ethanol with a chiral epoxide synthon. Following the initial coupling, the intermediate undergoes cyclization and deprotection to reveal the free amine. To ensure high enantiomeric purity, an enzymatic resolution step is employed using Lipase PS-C in the presence of an acyl donor like ethyl octanoate. This step selectively acylates one enantiomer, allowing the desired unreacted amine to be isolated with high optical purity. The detailed standardized synthesis steps for this transformation are provided in the guide below.
- React a protected amino-alcohol with a chiral epoxide derivative under Mitsunobu conditions or via direct nucleophilic ring-opening to form the morpholine core.
- Isolate the protected intermediate, such as the phthalimido-substituted morpholine, through crystallization from alcoholic solvents.
- Perform deprotection using hydrazine or acidic hydrolysis, followed by enzymatic kinetic resolution if necessary to achieve >95% ee.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the processes described in CN1642928A offers tangible benefits in terms of cost stability and supply security. The shift away from chromatographic purification towards crystallization-based isolation significantly reduces the consumption of high-grade solvents and specialized consumables. This simplification of the downstream processing directly translates to lower manufacturing costs and a reduced environmental footprint, which is increasingly important for regulatory compliance and corporate sustainability goals. Additionally, the reliance on commercially available starting materials, such as substituted benzyl amines and epichlorohydrin derivatives, mitigates the risk of supply chain disruptions associated with custom-synthesized chiral reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent systems, further enhances the reliability of the supply chain by allowing for flexibility in manufacturing sites.
- Cost Reduction in Manufacturing: The elimination of preparative chiral HPLC and the reduction in recrystallization cycles lead to substantial cost savings. By utilizing enzymatic resolution or chiral pool synthesis, the process avoids the capital expenditure associated with chromatography columns and the operational costs of solvent recovery. The use of common industrial solvents like toluene, THF, and isopropanol ensures that raw material costs remain predictable and low. Furthermore, the high yields reported in the examples, such as the 526.9g batch in Example 4, demonstrate that the process is efficient on a larger scale, maximizing the throughput per batch and reducing the cost per kilogram of the final intermediate.
- Enhanced Supply Chain Reliability: The synthetic routes utilize readily available precursors, reducing dependency on single-source suppliers for exotic chiral catalysts. The enzymatic step uses immobilized lipases which can often be recovered and reused, adding another layer of cost efficiency and supply stability. The process is designed to be robust, with wide operating windows for temperature and reaction time, which minimizes the risk of batch failures due to minor process deviations. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of modern pharmaceutical supply chains.
- Scalability and Environmental Compliance: The patent provides clear evidence of scalability, with examples demonstrating successful execution from gram scale to multi-hundred gram scale without loss of efficiency. The work-up procedures involve standard unit operations like filtration, distillation, and liquid-liquid extraction, which are easily transferable to pilot and commercial plants. From an environmental perspective, the reduction in solvent volume and the avoidance of heavy metal catalysts or toxic reagents simplify waste treatment. The ability to isolate intermediates as solids reduces the need for energy-intensive drying processes associated with oils, contributing to a lower overall carbon footprint for the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a reliable basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this chemistry into their existing portfolios.
Q: What is the primary advantage of the enzymatic resolution method described in CN1642928A?
A: The enzymatic resolution using Lipase PS-C allows for the separation of enantiomers under mild conditions (25-35°C), avoiding the need for expensive chiral HPLC columns or complex diastereomeric salt crystallizations, thereby significantly reducing processing costs.
Q: Can this process be scaled for commercial API production?
A: Yes, the patent explicitly demonstrates scalability with examples ranging from gram scale to over 500g batches (Example 4), utilizing common industrial solvents like toluene and isopropanol, which facilitates straightforward commercial scale-up.
Q: What level of optical purity can be achieved?
A: The disclosed methods, particularly the enzymatic resolution and chiral epoxide routes, consistently achieve high enantiomeric excess, with specific examples reporting up to 96% ee and 99% area/area purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Morpholine Derivatives Supplier
The technologies outlined in CN1642928A represent a sophisticated approach to constructing high-value pharmaceutical intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate these patented methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from development to market. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical tools to verify enantiomeric excess and chemical purity at every stage of production. Our commitment to quality assurance guarantees that every batch of morpholine derivatives meets the exacting standards required for global regulatory submissions.
We invite you to collaborate with us to leverage these advanced synthetic routes for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce overall project costs. Let us be your partner in delivering high-quality chiral intermediates with speed and precision.
