Advanced Visible Light-Promoted Oxidative Ring-Opening of Morpholine Derivatives for Pharmaceutical Intermediates
Advanced Visible Light-Promoted Oxidative Ring-Opening of Morpholine Derivatives for Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient methods for constructing complex molecular architectures, particularly when dealing with the selective activation of inert bonds. Patent CN113072460B introduces a groundbreaking methodology for the oxidative ring-opening of morpholine derivatives, leveraging visible light photocatalysis to achieve C(sp3)-C(sp3) bond cleavage under exceptionally mild conditions. This innovation addresses the long-standing challenge of activating non-strained carbon-carbon bonds without relying on harsh reagents or expensive transition metals. By utilizing molecular oxygen as the terminal oxidant and organic photocatalysts, this technology offers a greener alternative that aligns perfectly with modern green chemistry principles, providing a robust pathway for generating valuable open-chain intermediates from cyclic amines.
For research and development teams focused on drug discovery, the ability to selectively functionalize morpholine rings is of paramount importance, as these structures are ubiquitous in bioactive molecules. The method described in CN113072460B not only expands the synthetic toolbox available to medicinal chemists but also ensures high purity profiles by avoiding metal contamination. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such advanced synthetic routes allows us to better serve clients who demand high-quality building blocks for their API synthesis. The transition from traditional thermal or metal-catalyzed processes to photochemical methods represents a significant paradigm shift, enabling access to chemical spaces that were previously difficult or economically unviable to explore.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the oxidative cleavage of C(sp3)-C(sp3) bonds in morpholine derivatives has been fraught with significant challenges that limit its practical application in large-scale manufacturing. Early attempts, such as those by Albini's group, relied on high-energy UV irradiation (310 nm) in aqueous conditions, which often resulted in negligible yields and poor selectivity, making the process unsuitable for complex molecule synthesis. Furthermore, other conventional approaches utilized hazardous oxidants like ozone or stoichiometric amounts of toxic heavy metal reagents such as lead tetraacetate, posing severe safety risks and environmental burdens. These traditional methods frequently required extreme reaction conditions, including high temperatures and pressures, which can lead to the decomposition of sensitive functional groups commonly found in pharmaceutical intermediates.
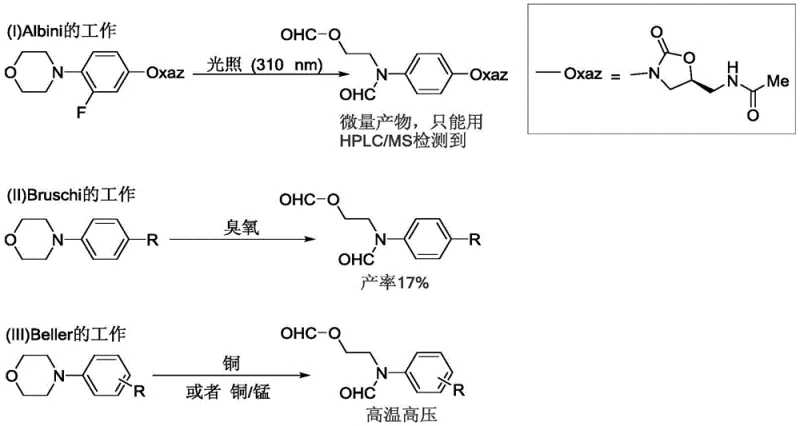
The reliance on transition metal catalysts, as seen in Beller's work involving copper or cobalt-manganese systems, introduces another layer of complexity regarding downstream processing. The removal of trace metal residues to meet stringent regulatory standards for pharmaceutical ingredients adds substantial cost and time to the production cycle. Additionally, the use of strong chemical oxidants often lacks the chemoselectivity required for late-stage functionalization, leading to over-oxidation or unwanted side reactions. These limitations collectively hinder the efficiency of supply chains and increase the overall cost of goods, creating a pressing need for a more sustainable, metal-free, and mild alternative that can be easily scaled without compromising safety or product quality.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN113072460B utilizes visible light photocatalysis to drive the oxidative ring-opening reaction at room temperature, eliminating the need for thermal energy input. By employing organic photocatalysts like 4CzIPN, the process avoids the introduction of transition metals entirely, thereby simplifying the purification workflow and ensuring a cleaner final product profile. The use of molecular oxygen from the air as the sole oxidant not only reduces reagent costs drastically but also generates water as the primary byproduct, significantly minimizing waste generation. This approach demonstrates exceptional functional group tolerance, successfully accommodating substrates with electron-withdrawing and electron-donating groups, halogens, and even sensitive heterocycles without degradation.
The operational simplicity of this novel method cannot be overstated, as it proceeds under ambient pressure using standard blue LED light sources, making it highly amenable to scale-up in flow chemistry reactors or batch processes. For procurement managers, this translates to a reduction in the dependency on specialized high-pressure equipment and expensive metal catalysts, leading to substantial cost reduction in pharmaceutical intermediate manufacturing. The ability to achieve yields up to 83% across a diverse range of thirty different substrates underscores the robustness and versatility of this protocol. By shifting to this visible light-promoted strategy, manufacturers can achieve cost reduction in fine chemical intermediates production while adhering to increasingly strict environmental regulations regarding heavy metal discharge and hazardous waste disposal.
Mechanistic Insights into Visible Light-Promoted C(sp3)-C(sp3) Bond Cleavage
The mechanistic pathway elucidated in the patent provides a fascinating glimpse into the power of single-electron transfer (SET) processes in organic synthesis. Upon irradiation with visible light, the organic photocatalyst 4CzIPN is excited to its singlet or triplet state, becoming a potent oxidant capable of accepting an electron from the morpholine substrate. This initial single-electron transfer generates a highly reactive radical cation intermediate and the reduced form of the photocatalyst. The radical cation subsequently undergoes deprotonation at the alpha-position relative to the nitrogen atom, facilitated by the base additive, to form a neutral alpha-amino radical. This step is critical as it sets the stage for the interaction with molecular oxygen, which acts as the radical trap to form a peroxyl radical species.
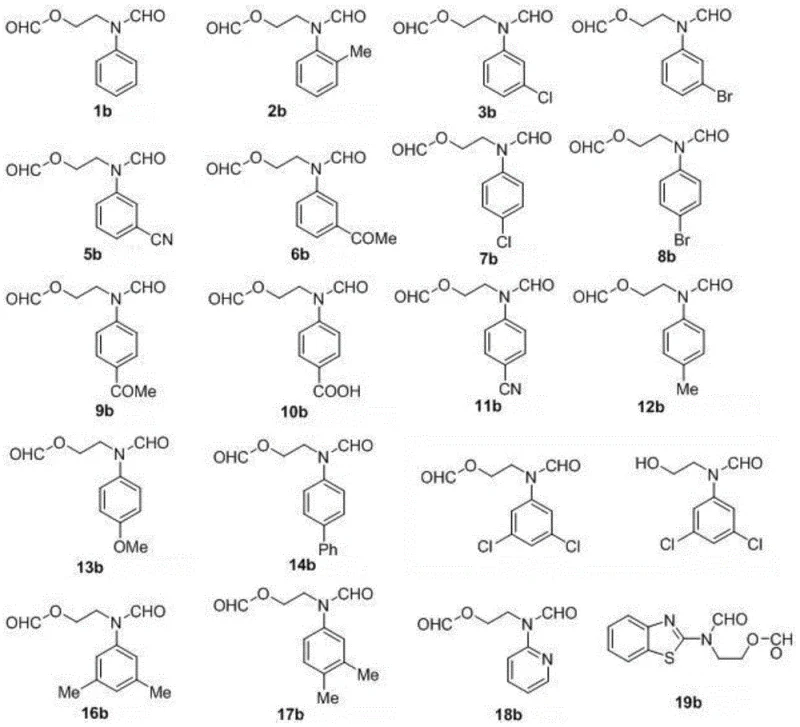
Further mechanistic studies involving radical trapping experiments with TEMPO and BHT confirmed the involvement of radical intermediates, as the reaction was completely inhibited in their presence. Isotopic labeling experiments using 18O2 unequivocally demonstrated that the oxygen atoms incorporated into the carbonyl groups of the final product originate from molecular oxygen, validating the oxidative nature of the transformation. The peroxyl radical eventually cyclizes to form a transient dioxetane intermediate, which is thermally unstable and spontaneously decomposes to yield the final ring-opened formamide ester product. This intricate cascade of events occurs under mild conditions, preventing the formation of complex impurity profiles often associated with high-thermal stress reactions. Understanding this mechanism allows R&D directors to predict substrate compatibility and optimize reaction parameters for new analogues, ensuring high-purity pharmaceutical intermediates are consistently produced.
The control over impurity formation is a direct consequence of the mild reaction conditions and the specificity of the photocatalytic cycle. Unlike thermal oxidation which can lead to non-selective burning of the organic framework, the photo-induced electron transfer is highly selective for the amine moiety. This selectivity ensures that other sensitive functional groups on the aromatic ring, such as nitriles, esters, or halides, remain intact throughout the transformation. For quality control teams, this means a simpler impurity profile that is easier to characterize and control, reducing the risk of batch failures during commercial production. The ability to tune the reaction by adjusting the photocatalyst loading or light intensity provides an additional layer of process control, enabling the fine-tuning of reaction kinetics to maximize yield and minimize byproduct formation.
How to Synthesize N-Phenylmorpholine Derivatives Efficiently
The synthesis of these valuable open-chain intermediates follows a straightforward protocol that can be implemented in standard laboratory or pilot plant settings with minimal equipment modifications. The process begins by dissolving the morpholine derivative substrate in a suitable organic solvent such as acetonitrile, followed by the addition of the organic photocatalyst and a mild base additive. The reaction vessel is then purged with oxygen and sealed, typically with an oxygen balloon, to maintain a constant supply of the oxidant throughout the reaction duration. Irradiation with a blue LED light source at room temperature drives the reaction to completion, which can be monitored conveniently using thin-layer chromatography.
- Dissolve the morpholine derivative substrate, photocatalyst 4CzIPN, and additive (such as 2,6-lutidine) in an organic solvent like acetonitrile.
- Maintain an oxygen atmosphere (using an oxygen balloon) and irradiate the reaction mixture with a 9W blue light lamp at room temperature.
- Monitor the reaction progress via TLC, then concentrate the mixture under reduced pressure and purify the crude product using silica gel column chromatography.
Following the reaction, the workup procedure is remarkably simple, involving the removal of the solvent under reduced pressure to afford the crude product. Purification is typically achieved via standard silica gel column chromatography using common eluent systems like petroleum ether and ethyl acetate. This ease of isolation is a significant advantage over metal-catalyzed methods that often require complex extraction procedures to remove metal salts. The scalability of this method is supported by the use of inexpensive reagents and ambient conditions, making it an attractive option for the commercial scale-up of complex pharmaceutical intermediates. By adhering to these optimized conditions, manufacturers can reliably produce high-quality intermediates with consistent batch-to-batch reproducibility.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this visible light-mediated technology offers profound benefits that extend beyond mere chemical efficiency. The elimination of transition metal catalysts removes a major bottleneck in the supply chain associated with the sourcing of precious metals like palladium, iridium, or rhodium, which are subject to volatile market prices and geopolitical supply risks. Furthermore, the absence of heavy metals simplifies the regulatory compliance landscape, as there is no need for extensive and costly metal scavenging steps or rigorous testing for residual metal limits in the final active pharmaceutical ingredient. This streamlining of the production process directly contributes to cost reduction in pharmaceutical intermediate manufacturing by lowering both material and operational expenses.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with inexpensive organic photocatalysts like 4CzIPN significantly lowers the raw material costs associated with the synthesis. Additionally, the use of molecular oxygen from the air as the oxidant eliminates the need for purchasing hazardous and costly stoichiometric oxidizing agents. The mild reaction conditions also reduce energy consumption by removing the requirement for high-temperature heating or high-pressure reactors, leading to substantial cost savings in utility bills. These factors combined result in a more economically viable process that enhances the overall profit margin for the final product.
- Enhanced Supply Chain Reliability: By relying on readily available organic reagents and common light sources, the supply chain becomes more resilient to disruptions caused by the scarcity of specialized catalytic metals. The robustness of the reaction across a wide range of substrates ensures that production schedules are less likely to be impacted by substrate-specific failures or optimization delays. This reliability is crucial for maintaining continuous supply to downstream customers, reducing lead time for high-purity pharmaceutical intermediates and ensuring that project timelines are met without unexpected delays caused by reagent shortages or complex purification bottlenecks.
- Scalability and Environmental Compliance: The metal-free nature of this process aligns perfectly with green chemistry initiatives, reducing the environmental footprint of chemical manufacturing by minimizing hazardous waste generation. The simplified workup and purification procedures facilitate easier scale-up from gram to kilogram and ton scales without the need for specialized waste treatment facilities for heavy metal disposal. This environmental compliance not only mitigates regulatory risks but also enhances the corporate sustainability profile, making the supply chain more attractive to eco-conscious partners and stakeholders who prioritize responsible sourcing and manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light oxidative ring-opening technology. These insights are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing clarity on the practical aspects of adopting this method. Understanding these details helps stakeholders make informed decisions about integrating this technology into their existing production workflows.
Q: What are the advantages of this visible light method over traditional transition metal catalysis?
A: This method eliminates the need for expensive and toxic transition metal catalysts like copper or cobalt, thereby reducing heavy metal residue concerns in pharmaceutical intermediates and simplifying the purification process.
Q: Does this oxidative ring-opening method tolerate diverse functional groups?
A: Yes, the protocol demonstrates excellent functional group tolerance, successfully processing substrates with halogens, nitriles, ketones, and carboxylic acids without significant degradation or side reactions.
Q: What is the primary oxidant used in this green chemistry approach?
A: Molecular oxygen (O2) serves as the terminal oxidant, which is environmentally benign, cost-effective, and avoids the use of hazardous stoichiometric oxidants like ozone or lead tetraacetate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Morpholine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible light photocatalysis in modernizing the synthesis of complex pharmaceutical intermediates. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN113072460B can be seamlessly translated into robust industrial processes. We are committed to delivering high-purity morpholine derivatives and their open-chain counterparts with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to guarantee product quality and consistency.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us to request specific COA data for our available inventory or to discuss route feasibility assessments for custom synthesis projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that is not only reliable and compliant but also at the forefront of sustainable chemical innovation, ensuring your competitive edge in the global marketplace.
