Advanced Synthetic Route for 5,6-Dimethyl-2-(4-fluorophenylamino)-4-(1-methyl-1,2,3,4-tetrahydroisoquinolin-2-yl)pyrimidine Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways for potent therapeutic agents, particularly those targeting gastrointestinal disorders. Patent CN1217722A introduces a groundbreaking methodology for the preparation of 5,6-dimethyl-2-(4-fluorophenylamino)-4-(1-methyl-1,2,3,4-tetrahydroisoquinolin-2-yl)pyrimidine and its acid addition salts, compounds known for their reversible proton-pump inhibitory effects useful as anti-ulcer agents. Unlike prior art methods which suffered from significant byproduct formation due to non-selective reactivity, this invention utilizes a specifically designed mono-halogenated pyrimidine intermediate that reacts cleanly with 1-methyl-1,2,3,4-tetrahydroisoquinoline. This strategic modification not only streamlines the purification process but also drastically enhances the overall yield and purity profile of the final active pharmaceutical ingredient (API) precursor. For a reliable pharmaceutical intermediate supplier, mastering such selective coupling reactions is critical to meeting the stringent quality standards demanded by global regulatory bodies.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex pyrimidine derivatives often relied on starting materials possessing multiple reactive functional groups, which introduced significant challenges in process control and product isolation. Specifically, earlier protocols described in related patent applications utilized dichloro-substituted pyrimidines that contained two chemically equivalent or similar reaction sites. When these substrates were subjected to nucleophilic attack by amines, the reaction lacked the necessary regioselectivity, inevitably producing a mixture of the desired mono-substituted product and unwanted bis-substituted byproducts. This lack of selectivity necessitated extensive and costly downstream purification steps, such as repeated recrystallization or chromatography, to remove impurities that possessed similar physical properties to the target molecule. Furthermore, the formation of these byproducts inherently capped the maximum theoretical yield, making the process economically inefficient for large-scale commercial production where material throughput is paramount.
The Novel Approach
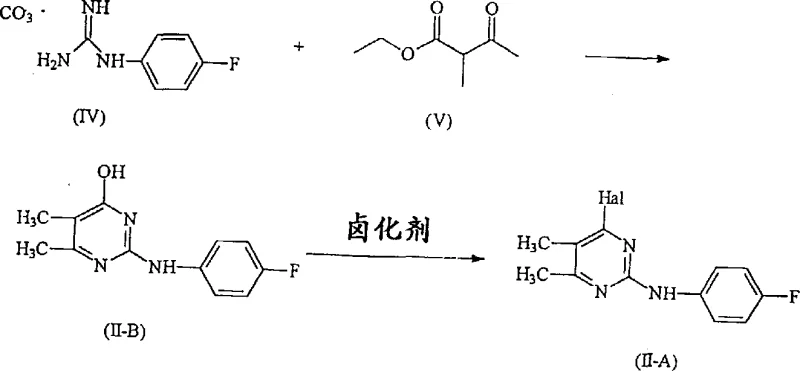
The inventive process detailed in CN1217722A overcomes these historical bottlenecks by employing a pyrimidine derivative represented by Formula II-A, wherein only a single halogen atom serves as the leaving group for the nucleophilic substitution. By restricting the reactivity to a single site on the heterocyclic ring, the reaction with 1-methyl-1,2,3,4-tetrahydroisoquinoline proceeds with exceptional specificity, effectively eliminating the generation of bis-substituted impurities. This high degree of chemical selectivity translates directly into a simplified work-up procedure, as the crude reaction mixture contains predominantly the desired product along with easily removable inorganic salts and excess reagents. Additionally, the process allows for the use of excess tetrahydroisoquinoline, which is a liquid under reaction conditions and can be readily separated post-reaction, further driving the equilibrium towards completion without compromising purity. This approach represents a significant leap forward in cost reduction in API manufacturing by minimizing waste and maximizing resource utilization.
Mechanistic Insights into Nucleophilic Aromatic Substitution and Lewis Acid Cyclization
The core of this synthetic strategy relies on a nucleophilic aromatic substitution mechanism where the secondary amine nitrogen of the tetrahydroisoquinoline moiety attacks the electron-deficient carbon at the 4-position of the pyrimidine ring. The presence of electron-withdrawing nitrogen atoms within the pyrimidine core activates the ring towards nucleophilic attack, while the specific positioning of the halogen leaving group ensures that substitution occurs exclusively at the intended site. The reaction is typically conducted in high-boiling polar solvents such as ethylene glycol or propylene glycol at temperatures ranging from 110°C to 160°C, which provides the necessary thermal energy to overcome the activation barrier for the displacement of the halide. The use of organic bases like triethylamine or inorganic bases like potassium acetate facilitates the deprotonation of the amine nucleophile, enhancing its reactivity and ensuring smooth conversion to the final coupled product without degrading the sensitive fluorophenyl amino group.
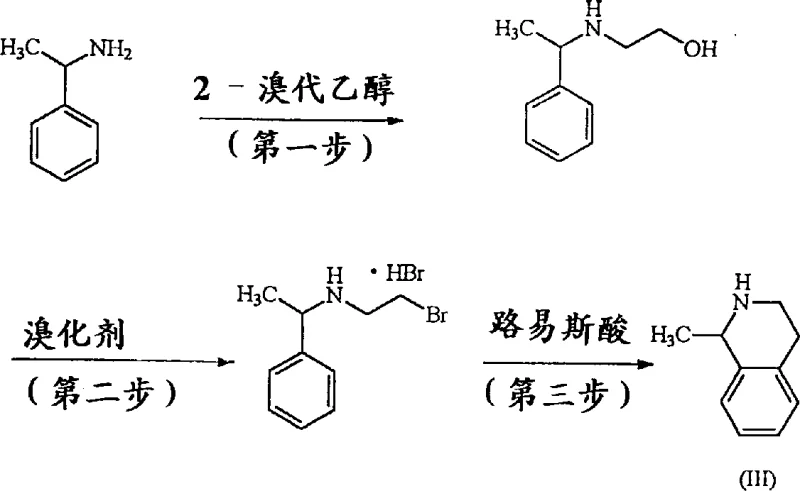
Beyond the final coupling step, the patent elucidates a novel and safer pathway for constructing the 1-methyl-1,2,3,4-tetrahydroisoquinoline scaffold itself, which is crucial for controlling the stereochemical outcome of the final drug substance. Traditional methods for synthesizing this chiral building block often involved hazardous reagents such as alpha-chloro-alpha-(methylthio)-acetyl chloride, which is unstable and poses explosion risks during handling and storage. In contrast, the disclosed method utilizes a sequential reaction of alpha-methylbenzylamine with 2-bromoethanol followed by bromination and a Lewis acid-catalyzed intramolecular Friedel-Crafts cyclization. This cyclization step, mediated by catalysts like aluminum chloride or zinc chloride, efficiently closes the ring to form the tetrahydroisoquinoline structure with high fidelity. By avoiding unstable intermediates, this route significantly improves the safety profile of the manufacturing process, making it more suitable for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 5,6-Dimethyl-2-(4-fluorophenylamino)-4-(1-methyl-1,2,3,4-tetrahydroisoquinolin-2-yl)pyrimidine Efficiently
The efficient production of this high-value pyrimidine derivative requires precise control over reaction parameters and a deep understanding of the interplay between the two key fragments. The process begins with the condensation of 4-fluorophenyl guanidine carbonate with ethyl 2-methylacetoacetate to form a hydroxyl-pyrimidine intermediate, which is subsequently halogenated using agents like phosphoryl chloride or phosphorus tribromide to yield the reactive Formula II-A species. Parallel to this, the chiral tetrahydroisoquinoline component is prepared via the aforementioned Lewis acid cyclization route, allowing for the introduction of specific (R) or (S) stereochemistry depending on the starting alpha-methylbenzylamine enantiomer. The final convergence of these two streams involves heating the components in a glycol-based solvent system, where the kinetics favor the formation of the C-N bond while maintaining the integrity of the chiral center. Detailed standardized synthesis steps see the guide below.
- Prepare the key intermediate 4-halo-2-(4-fluorophenylamino)-5,6-dimethylpyrimidine by reacting 4-fluorophenyl guanidine carbonate with ethyl 2-methylacetoacetate followed by halogenation.
- Synthesize 1-methyl-1,2,3,4-tetrahydroisoquinoline by reacting alpha-methylbenzylamine with 2-bromoethanol, followed by bromination and Lewis acid-catalyzed cyclization.
- Perform the final coupling reaction between the halogenated pyrimidine intermediate and the tetrahydroisoquinoline derivative in a high-boiling solvent like ethylene glycol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthetic route offers substantial strategic benefits that extend beyond simple chemical yield improvements. The elimination of byproduct-forming side reactions means that raw material consumption is optimized, as fewer starting materials are wasted on generating impurities that must ultimately be discarded. This efficiency gain directly contributes to substantial cost savings in the overall production budget, allowing for more competitive pricing structures in the global marketplace. Furthermore, the avoidance of explosive and unstable reagents in the tetrahydroisoquinoline synthesis reduces the need for specialized containment infrastructure and lowers insurance and safety compliance costs associated with hazardous chemical handling. These factors combined create a more resilient and economically viable supply chain for this critical pharmaceutical intermediate.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the dramatic simplification of the purification workflow. Because the reaction between the mono-halogenated pyrimidine and the tetrahydroisoquinoline is highly selective, the resulting crude product requires minimal purification, often needing only a simple filtration or recrystallization rather than expensive chromatographic separation. This reduction in downstream processing time and solvent usage significantly lowers the operational expenditure per kilogram of produced material. Additionally, the ability to use excess liquid amine reagent which is easily recovered and recycled further enhances the economic efficiency of the process, ensuring that the cost of goods sold remains low even at varying scales of production.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of robust, commercially available starting materials such as 4-fluoroaniline and alpha-methylbenzylamine, which are produced by multiple vendors globally, mitigating the risk of single-source dependency. The synthetic route does not rely on exotic catalysts or reagents with long lead times, ensuring that production schedules can be maintained without interruption due to material shortages. Moreover, the improved safety profile of the tetrahydroisoquinoline synthesis removes potential bottlenecks caused by strict regulatory controls on explosive precursors, allowing for smoother logistics and warehousing operations. This reliability is essential for reducing lead time for high-purity pharmaceutical intermediates and ensuring consistent delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing solvents like ethylene glycol and propylene glycol which have high boiling points and allow reactions to proceed at atmospheric pressure without the need for complex high-pressure reactor vessels. This simplifies the engineering requirements for scaling up from pilot plant to multi-ton commercial production, reducing capital expenditure on equipment. From an environmental perspective, the high atom economy and reduced generation of hazardous waste align with modern green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations. The ability to operate safely and cleanly at large volumes makes this technology an attractive option for sustainable chemical manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology, derived directly from the specific advantages and mechanisms detailed in the patent documentation. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios. The answers provided reflect the empirical data and logical deductions found within the intellectual property, offering clarity on yield expectations, safety protocols, and stereochemical control capabilities.
Q: How does this process improve upon previous methods for synthesizing this pyrimidine derivative?
A: Previous methods utilized dichloro-pyrimidine starting materials which possessed two reactive sites, inevitably leading to bis-substituted byproducts and reduced yields. This patented process employs a mono-halogenated intermediate (Formula II-A) with only one reactive site, ensuring high regioselectivity and eliminating the formation of difficult-to-remove impurities.
Q: What are the safety advantages of the new tetrahydroisoquinoline synthesis route?
A: Conventional routes relied on unstable and potentially explosive reagents like alpha-chloro-alpha-(methylthio)-acetyl chloride. The novel method described in the patent utilizes stable, commercially available starting materials such as alpha-methylbenzylamine and 2-bromoethanol, significantly reducing operational hazards and facilitating safer commercial scale-up.
Q: Can this process produce specific optical isomers of the final compound?
A: Yes, the process is highly versatile regarding stereochemistry. By using optically pure (R)- or (S)-alpha-methylbenzylamine as the starting material for the tetrahydroisoquinoline synthesis, the corresponding (R)-(+)- or (S)-(-)-isomers of the final pyrimidine derivative can be obtained effectively without the need for complex resolution steps post-synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5,6-Dimethyl-2-(4-fluorophenylamino)-4-(1-methyl-1,2,3,4-tetrahydroisoquinolin-2-yl)pyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced synthetic methodologies to deliver high-quality pharmaceutical intermediates that meet the rigorous demands of the global healthcare sector. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch of 5,6-dimethyl-2-(4-fluorophenylamino)-4-(1-methyl-1,2,3,4-tetrahydroisoquinolin-2-yl)pyrimidine conforms to the highest standards of identity and potency required for subsequent drug substance synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this innovative process can be tailored to your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this superior synthetic route for your anti-ulcer agent programs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will enhance the competitiveness and reliability of your pharmaceutical product portfolio.
