Optimizing Pyrimidine Derivative Production: A Strategic Guide for Pharmaceutical Sourcing and Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways for active pharmaceutical ingredients (APIs) and their precursors, particularly for gastro-intestinal therapeutics. Patent CN1097591C introduces a significant advancement in the preparation of 5,6-dimethyl-2-(4-fluorophenylamino)-4-(1-methyl-1,2,3,4-tetrahydroisoquinolin-2-yl)pyrimidine and its acid addition salts. This compound functions as a reversible proton-pump inhibitor, making it a critical candidate for anti-ulcer medication. The disclosed technology addresses long-standing challenges in heterocyclic chemistry by optimizing reaction selectivity and yield. For R&D directors and procurement specialists, understanding this patented methodology is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials. The innovation lies not just in the final molecule, but in the strategic design of the intermediate compounds that streamline the entire manufacturing workflow.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of similar pyrimidine derivatives relied on starting materials with multiple reactive sites, which introduced significant inefficiencies into the production line. As illustrated in the prior art reaction schemes, conventional methods often utilized dichloro-substituted pyrimidines where both chlorine atoms were susceptible to nucleophilic attack. This lack of regioselectivity meant that the first reaction step inevitably produced unwanted bis-substituted by-products alongside the desired mono-substituted compound. These impurities not only lowered the overall yield of the target API intermediate but also necessitated complex and costly purification processes to meet stringent pharmaceutical standards. The presence of these side reactions created a bottleneck in commercial scale-up of complex pharmaceutical intermediates, driving up costs and extending lead times for high-purity batches.
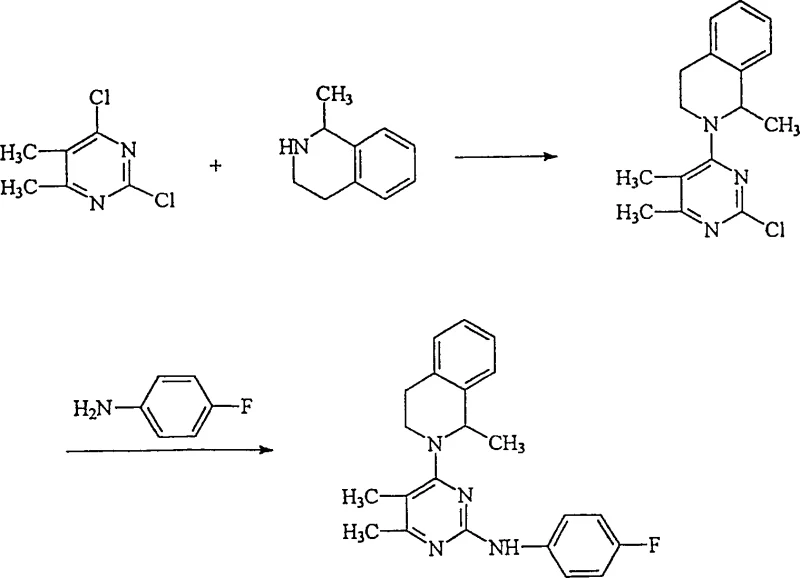
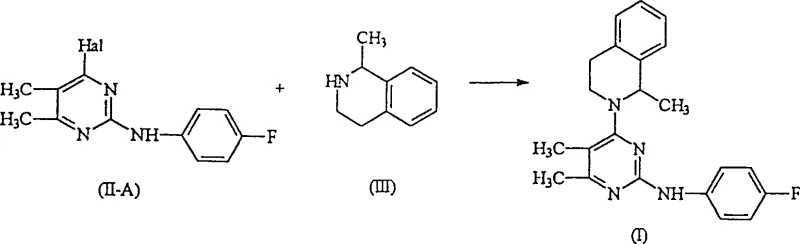
The Novel Approach
The breakthrough detailed in CN1097591C circumvents these issues by employing a specifically designed intermediate, 4-halo-2-(4-fluorophenylamino)-5,6-dimethyl-pyrimidine, represented by formula (II-A). Unlike the dichloro predecessors, this intermediate possesses a single reactive halogen site, ensuring that the subsequent nucleophilic substitution with 1-methyl-1,2,3,4-tetrahydroisoquinoline occurs exclusively at the intended position. This structural modification effectively eliminates the formation of bis-substituted by-products, thereby optimizing the yield of the desired formula (I) compound. The reaction flow demonstrates a clean transformation where the amine nitrogen of the tetrahydroisoquinoline ring attacks the pyrimidine core without competing side reactions. This level of control is paramount for cost reduction in API manufacturing, as it simplifies downstream processing and maximizes material utilization.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core of this synthetic strategy relies on a nucleophilic aromatic substitution mechanism where the secondary amine of the 1-methyl-1,2,3,4-tetrahydroisoquinoline acts as the nucleophile. The electron-deficient nature of the pyrimidine ring, enhanced by the electronegative halogen atom at the 4-position, facilitates this attack. The reaction is typically conducted in high-boiling polar solvents such as ethylene glycol or 1,2-propanediol, which stabilize the transition state and allow the reaction to proceed efficiently at temperatures ranging from 110°C to 160°C. The use of a base, such as triethylamine or potassium acetate, further promotes the reaction by scavenging the hydrogen halide by-product, driving the equilibrium towards the formation of the final pyrimidine derivative. This mechanistic precision ensures that the process is not only chemically sound but also reproducible on a large scale, a key requirement for any reliable agrochemical intermediate supplier or pharma partner.
Furthermore, the patent addresses the stereochemical complexity of the target molecule. The final compound contains an asymmetric carbon atom, marked by an asterisk in the structural formulas, which gives rise to (R)-(+)- and (S)-(-)-isomers. The synthetic route is versatile enough to accommodate chiral starting materials; by using optically pure (R)- or (S)-1-methyl-1,2,3,4-tetrahydroisoquinoline, manufacturers can produce the corresponding enantiomerically enriched final products. This capability is crucial for modern drug development, where specific isomers often exhibit superior pharmacological activity and safety profiles. The ability to control chirality without resorting to difficult resolution steps post-synthesis represents a significant technical advantage, ensuring that the impurity profile remains within tight specifications required by global regulatory bodies.
How to Synthesize 5,6-dimethyl-2-(4-fluorophenylamino)-4-(1-methyl-1,2,3,4-tetrahydroisoquinolin-2-yl)pyrimidine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing this valuable intermediate with high efficiency. The process begins with the preparation of the key halo-pyrimidine intermediate, followed by its coupling with the tetrahydroisoquinoline derivative under controlled thermal conditions. The detailed experimental examples demonstrate that yields can reach upwards of 90% under optimized conditions, validating the robustness of the method. For technical teams looking to implement this route, the following guide summarizes the critical operational parameters derived from the patent data. Detailed standardized synthesis steps are provided in the section below to ensure reproducibility and safety during scale-up operations.
- Prepare the key intermediate 4-halo-2-(4-fluorophenylamino)-5,6-dimethyl-pyrimidine by reacting 4-fluorophenylguanidine carbonate with ethyl 2-methylacetoacetate followed by halogenation.
- React the halo-pyrimidine intermediate with 1-methyl-1,2,3,4-tetrahydroisoquinoline in a high-boiling solvent like ethylene glycol or 1,2-propanediol at 110-160°C.
- Isolate the final product by converting it into an acid addition salt, such as the hydrochloride, through treatment with hydrochloric acid in ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible strategic benefits beyond mere chemical elegance. The elimination of by-products fundamentally alters the economics of production by reducing the burden on purification infrastructure. Traditional methods requiring extensive chromatography or recrystallization to remove bis-substituted impurities are resource-intensive; by contrast, this cleaner reaction profile allows for simpler work-up procedures, such as direct salt formation and filtration. This streamlining translates directly into substantial cost savings and a more predictable production schedule, mitigating the risks associated with batch failures or off-spec material. Additionally, the use of commercially available and relatively inexpensive solvents like glycols enhances the economic viability of the process.
- Cost Reduction in Manufacturing: The primary driver for cost efficiency in this process is the drastic simplification of the purification workflow. Because the reaction does not generate significant amounts of structural by-products, the need for expensive separation technologies is minimized. The high conversion rates observed in the patent examples mean that raw material consumption is optimized, reducing the cost per kilogram of the final active ingredient. Furthermore, the ability to perform the reaction in a single vessel without isolating unstable intermediates reduces labor hours and equipment usage, contributing to a leaner manufacturing operation that is highly attractive for long-term supply contracts.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on exotic or unstable reagents. This process utilizes stable, commodity-grade starting materials such as 4-fluoroaniline and ethyl 2-methylacetoacetate, which are readily available from multiple global sources. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent mixtures, ensures that production is less susceptible to minor fluctuations in process parameters. This resilience is critical for maintaining consistent inventory levels and meeting the rigorous delivery timelines demanded by downstream pharmaceutical manufacturers, thereby strengthening the overall reliability of the supply chain.
- Scalability and Environmental Compliance: The process is inherently scalable, as evidenced by the patent examples which describe reactions performed on multi-hundred-gram to kilogram scales without loss of efficiency. The preference for glycol-based solvents over more volatile or toxic organic solvents aligns with modern green chemistry principles, simplifying waste management and environmental compliance. Reduced solvent toxicity and lower energy requirements for solvent recovery contribute to a smaller environmental footprint. This alignment with sustainability goals is increasingly important for multinational corporations seeking to partner with suppliers who prioritize eco-friendly manufacturing practices and regulatory adherence.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and claims within CN1097591C, providing clarity for stakeholders evaluating this synthesis route. Understanding these nuances is vital for making informed decisions about sourcing and process development. The answers reflect the specific advantages of the mono-halo intermediate strategy over legacy methods.
Q: Why is the mono-halo intermediate superior to the dichloro starting material?
A: The conventional dichloro starting material possesses two reactive sites, inevitably leading to bis-substituted by-products that reduce yield and complicate purification. The novel mono-halo intermediate ensures regioselectivity, eliminating these side reactions.
Q: Can this process produce specific chiral isomers?
A: Yes, the process supports stereoselective synthesis. By utilizing optically pure (R)- or (S)-1-methyl-1,2,3,4-tetrahydroisoquinoline as the starting amine, the corresponding (R)-(+)- or (S)-(-)-isomers of the final pyrimidine derivative can be obtained.
Q: What solvents are recommended for industrial scale-up?
A: The patent highlights ethylene glycol and 1,2-propanediol as preferred solvents. These high-boiling polyols facilitate the reaction at 110-160°C, minimize reaction time, and significantly reduce the generation of by-products compared to other solvent systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5,6-dimethyl-2-(4-fluorophenylamino)-4-(1-methyl-1,2,3,4-tetrahydroisoquinolin-2-yl)pyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. Our team of expert chemists has extensively analyzed the methodology described in CN1097591C and is fully equipped to translate this laboratory-scale success into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are supported by rigorous QC labs and stringent purity specifications, guaranteeing that every batch of pyrimidine derivative meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this advanced technology for your product pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you secure not just a supplier, but a strategic ally committed to optimizing your manufacturing costs and ensuring uninterrupted supply continuity for your vital anti-ulcer therapeutics.
