Advanced Synthesis of Benzophenone Derivatives for Commercial Scale-Up
Advanced Synthesis of Benzophenone Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to produce critical intermediates, particularly benzophenone derivatives which serve as the backbone for numerous anti-inflammatory and antirheumatic agents. Patent CN1181376A introduces a groundbreaking method for producing compounds of Formula (I) with exceptional purity and yield, marking a significant departure from traditional synthesis routes. This technology specifically addresses the long-standing challenges associated with the acylation of N-phenyl amides, offering a robust solution for the reliable pharmaceutical intermediate supplier market. By utilizing a catalyst system based on metal halides rather than polyphosphonic acid, the process not only enhances reaction efficiency but also drastically simplifies the post-reaction workup, making it an ideal candidate for cost reduction in fine chemical manufacturing. The strategic shift in catalytic methodology ensures that the resulting aminobenzophenone derivatives meet the stringent quality standards required for downstream drug synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of substituted benzophenones has relied heavily on the use of polyphosphoric acid (PPA) as a condensing agent, a method that presents severe operational bottlenecks for commercial scale-up of complex intermediates. The high viscosity of polyphosphoric acid creates significant mixing difficulties, leading to uneven heat distribution and potential hot spots that can degrade sensitive functional groups on the aromatic rings. Furthermore, the quenching and separation of products from the thick PPA matrix is notoriously labor-intensive, often requiring large volumes of water and organic solvents, which increases waste generation and environmental compliance costs. The difficulty in completely removing phosphorus residues from the final product can also compromise the purity profile, necessitating additional recrystallization steps that reduce overall yield. These factors collectively contribute to extended production cycles and higher operational expenditures, making traditional PPA-based routes less attractive for modern, high-volume manufacturing environments.
The Novel Approach
In contrast, the novel approach detailed in the patent data utilizes a Friedel-Crafts catalyst system comprising metal halides, specifically tin halides, in combination with phosphorus halides like phosphorus oxychloride. This liquid-phase catalytic system offers superior fluidity compared to PPA, ensuring homogeneous mixing and precise temperature control throughout the reaction vessel. The use of solvents such as methylene dichloride allows for a homogeneous reaction medium that facilitates better contact between the N-phenyl amide and the benzoic acid derivative, leading to significantly improved conversion rates. The workup procedure is markedly simplified, as the catalyst residues can be effectively removed through standard aqueous washing and extraction techniques, eliminating the need for complex neutralization of viscous acid sludges. This methodological advancement translates directly into enhanced supply chain reliability by reducing batch-to-batch variability and ensuring consistent product quality.
Mechanistic Insights into SnCl4-Catalyzed Friedel-Crafts Acylation
The core of this technological breakthrough lies in the specific activation of the acylating agent by the Lewis acid catalyst, which generates a highly reactive acylium ion capable of attacking the electron-rich aromatic ring of the N-phenyl amide. The reaction proceeds through a classic electrophilic aromatic substitution mechanism, where the tin tetrachloride coordinates with the carbonyl oxygen of the benzoic acid or benzoyl halide, increasing the electrophilicity of the carbonyl carbon. This activation is crucial for overcoming the deactivating effect of the amide group on the aniline ring, ensuring that the acylation occurs selectively at the desired position, typically ortho to the amino group. The presence of phosphorus oxychloride further enhances the reaction kinetics by acting as a dehydrating agent, driving the equilibrium towards product formation and preventing the hydrolysis of the acylating agent. Understanding this mechanistic pathway is essential for optimizing reaction conditions to maximize yield while minimizing the formation of regioisomers or poly-acylated byproducts.

The structural integrity of the starting material, specifically the N-phenyl amide represented by Formula (II), plays a pivotal role in the success of the acylation. The amide group serves as a protecting group for the amine, preventing it from reacting with the acylating agent or the catalyst, while also directing the incoming electrophile to the ortho position through resonance effects. The substituents on Ring A, such as methoxy or hydroxyl groups, further activate the ring towards electrophilic attack, allowing the reaction to proceed under milder conditions than would be required for unsubstituted anilines. The precise control over the substitution pattern on Ring A ensures that the final benzophenone derivative possesses the exact stereochemical and electronic properties required for its biological activity in the target pharmaceutical application.
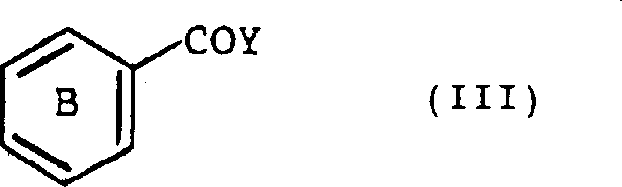
Similarly, the nature of the acylating agent, represented by Formula (III), dictates the structure of the resulting benzophenone ketone bridge. Whether using the free carboxylic acid or the corresponding acid halide, the reaction conditions are tuned to ensure complete conversion without damaging sensitive substituents on Ring B. The use of tin halides as the primary catalyst is particularly advantageous because they are less corrosive than aluminum chloride and easier to handle on a large scale. The catalyst loading is optimized to balance reaction speed with cost efficiency, typically ranging from 1 to 3 moles per mole of substrate. This careful balancing act ensures that the process remains economically viable while delivering the high-purity [high-purity pharmaceutical intermediates] demanded by regulatory bodies.
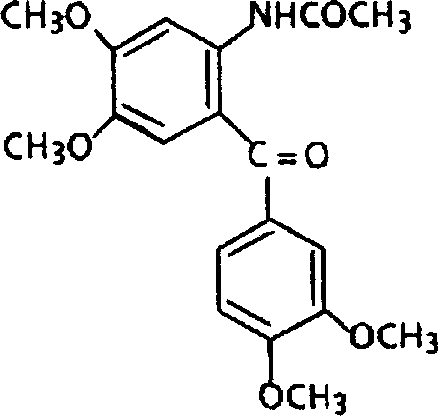
The final product, an aminobenzophenone derivative of Formula (I), is obtained with exceptional purity, often exceeding 96% without the need for extensive chromatographic purification. The impurity profile is significantly cleaner compared to PPA-based methods, primarily because the liquid catalyst system minimizes side reactions such as polymerization or over-acylation. The ability to isolate the product through simple crystallization from solvents like methanol or ethanol further enhances the economic feasibility of the process. This high level of purity is critical for downstream applications, where trace impurities can affect the efficacy and safety of the final drug product. The robust nature of this synthesis route makes it a preferred choice for the commercial scale-up of complex intermediates intended for high-value therapeutic areas.
How to Synthesize 2-Acetamido-3',4,4',5-tetramethoxybenzophenone Efficiently
The synthesis of specific high-value targets like 2-acetamido-3',4,4',5-tetramethoxybenzophenone exemplifies the practical application of this patented technology. The process begins with the suspension of N-(3,4-dimethoxyphenyl)ethanamide and 3,4-dimethoxybenzoic acid in methylene dichloride, creating a homogeneous reaction mixture. The detailed standardized synthesis steps see the guide below for precise operational parameters.
- Prepare the N-phenyl amide starting material and the benzoic acid or benzoyl halide derivative according to the specified molar ratios.
- Suspend the reactants in an inert organic solvent such as methylene dichloride and cool the mixture to below 35°C.
- Add the catalyst system, comprising tin tetrachloride and phosphorus oxychloride, and reflux the mixture for 10 to 20 hours to complete the acylation.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthesis route offers substantial cost savings and operational efficiencies that directly impact the bottom line. The elimination of polyphosphoric acid removes a major bottleneck in the production workflow, significantly reducing the time and resources required for reactor cleaning and waste disposal. This streamlining of the manufacturing process allows for faster batch turnover, enabling suppliers to respond more agilely to market demand fluctuations. Furthermore, the use of commercially available and relatively inexpensive catalysts like tin tetrachloride ensures that raw material costs remain stable and predictable. These factors combine to create a more resilient supply chain capable of delivering high-purity [pharmaceutical intermediates] with consistent reliability.
- Cost Reduction in Manufacturing: The primary driver for cost reduction lies in the simplified workup procedure, which eliminates the need for specialized equipment to handle viscous acids and reduces solvent consumption during extraction. By avoiding the extensive washing cycles required to remove phosphorus residues, manufacturers can significantly lower their utility and waste treatment costs. Additionally, the higher yield achieved with this method means that less raw material is wasted, further enhancing the overall economic efficiency of the production process. The qualitative improvement in process robustness also reduces the risk of batch failures, which can be a significant hidden cost in chemical manufacturing.
- Enhanced Supply Chain Reliability: The reliance on standard Friedel-Crafts reagents and common organic solvents ensures that the supply chain is not vulnerable to the shortages of specialized or niche chemicals. Tin halides and phosphorus oxychloride are widely produced commodities with stable global supply networks, reducing the risk of procurement delays. This availability allows for better inventory planning and reduces the need for safety stock, freeing up working capital. The consistency of the reaction also means that production schedules can be adhered to with greater confidence, reducing lead time for high-purity [pharmaceutical intermediates] and improving customer satisfaction.
- Scalability and Environmental Compliance: The liquid nature of the reaction mixture makes this process inherently easier to scale from pilot plant to full commercial production without the engineering challenges associated with mixing highly viscous media. The reduced generation of phosphorus-containing waste aligns with increasingly stringent environmental regulations, lowering the compliance burden on manufacturing facilities. Easier waste management translates to lower disposal fees and a smaller environmental footprint, which is increasingly important for corporate sustainability goals. The process is designed to be green by design, minimizing hazardous waste while maximizing atom economy.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of benzophenone derivatives using this advanced catalytic method. These answers are derived directly from the patent specifications and practical manufacturing experience to provide clarity on process capabilities.
Q: Why is polyphosphoric acid avoided in this benzophenone synthesis?
A: Polyphosphoric acid creates significant downstream processing challenges, including high viscosity and difficult product separation. The novel method uses metal halides to simplify workup and improve purity.
Q: What is the expected purity of the final benzophenone derivative?
A: The patented process consistently achieves high purity levels, often exceeding 96%, without the need for complex chromatographic purification steps.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the use of standard Friedel-Crafts catalysts and common solvents makes this route highly scalable for commercial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Acetamido-3',4,4',5-tetramethoxybenzophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of life-saving medications. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 2-acetamido-3',4,4',5-tetramethoxybenzophenone meets the highest industry standards. Our capability to implement the patented metal halide catalysis method allows us to offer a superior product profile compared to traditional suppliers.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis capabilities can support your long-term strategic goals. Let us be your partner in delivering high-quality chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
