Revolutionizing Benzophenone Production: Continuous Flow Technology for Commercial Scale-Up
Revolutionizing Benzophenone Production: Continuous Flow Technology for Commercial Scale-Up
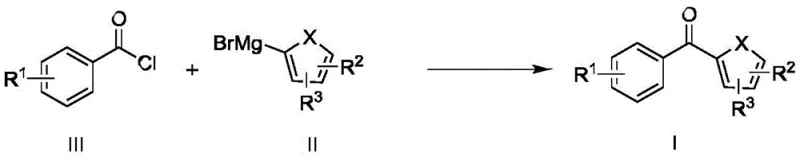
The landscape of fine chemical manufacturing is undergoing a paradigm shift, driven by the urgent need for safer, more efficient, and environmentally sustainable processes. A groundbreaking patent, CN108409516B, filed in June 2020, introduces a novel method for synthesizing benzophenone derivatives utilizing a continuous flow microreactor system. This technology represents a significant leap forward for the industry, addressing long-standing challenges associated with the traditional batch synthesis of these critical intermediates. By leveraging the superior mass and heat transfer characteristics of microreactors, this method enables the rapid reaction between aryl Grignard reagents and acyl chlorides at ambient temperatures. For global procurement leaders and R&D directors, this innovation signals a new era of reliability in the supply chain for pharmaceutical intermediates, offering a pathway to drastically reduce operational risks while enhancing overall process efficiency and product purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of benzophenone and its derivatives has relied heavily on methods such as the Friedel-Crafts acylation or traditional batch Grignard reactions. The Friedel-Crafts route, while established, is plagued by severe environmental drawbacks, including the generation of substantial amounts of hazardous waste due to the stoichiometric use of Lewis acids like aluminum trichloride and ferric trichloride. These processes create significant disposal burdens and purification challenges. Alternatively, the batch synthesis using aryl Grignard reagents and acyl chlorides presents acute safety hazards. These reactions are intensely exothermic, requiring rigorous temperature control often necessitating cryogenic conditions around -20°C to prevent thermal runaway and the formation of unwanted tertiary alcohol by-products. Furthermore, batch processes suffer from the notorious "amplification effect," where reaction performance degrades significantly upon scaling up from the laboratory to the plant due to inefficient mixing and heat dissipation, resulting in inconsistent yields typically hovering between a mere 10% and 20%.
The Novel Approach
In stark contrast, the continuous flow microreactor technology described in the patent offers a transformative solution that effectively neutralizes these historical pain points. By confining the reaction within micro-channels, the system achieves near-instantaneous mixing and exceptional heat exchange, allowing the highly exothermic Grignard addition to proceed safely at room temperature (25°C). This elimination of cryogenic requirements not only enhances operational safety but also leads to substantial energy savings. The process demonstrates remarkable robustness, overcoming the "amplification effect" entirely; parameters optimized in the lab translate directly to commercial production without loss of efficiency. Moreover, the integration of a solvent recovery system allows for the recycling of 2-methyltetrahydrofuran, aligning the process with modern green chemistry principles and significantly reducing the environmental footprint compared to traditional solvent-intensive batch operations.
Mechanistic Insights into Microreactor-Enhanced Grignard Acylation
The core of this technological advancement lies in the precise control over the reaction kinetics and thermodynamics afforded by the microreactor environment. In a conventional batch reactor, the addition of a Grignard reagent to an acyl chloride creates localized hot spots where the concentration of the nucleophile is high, promoting over-addition to form tertiary alcohols rather than the desired ketone. The microreactor architecture mitigates this by ensuring that the reagents are mixed at the molecular level almost instantaneously upon entering the mixing module. This homogeneous distribution prevents local excesses of the Grignard reagent, thereby kinetically favoring the formation of the ketone intermediate and suppressing further nucleophilic attack. The high surface-to-volume ratio of the microchannels facilitates the rapid removal of the heat of reaction, maintaining a uniform temperature profile throughout the reaction zone. This thermal stability is crucial for preserving the integrity of sensitive functional groups often present in complex pharmaceutical intermediates.
Furthermore, the impurity profile of the final product is significantly improved due to the reduced residence time distribution inherent in plug-flow systems. Unlike batch reactors where some molecules may remain in the reaction zone longer than others, leading to decomposition or side reactions, the continuous flow system ensures that every molecule experiences the exact same reaction history. This uniformity is critical for achieving the high purity specifications required by regulatory bodies for API intermediates. The ability to operate at ambient temperature also reduces the thermal stress on the reagents, minimizing degradation pathways that are accelerated at elevated temperatures or prolonged exposure to harsh conditions. Consequently, the downstream purification burden is lightened, as the crude product contains fewer by-products, streamlining the overall manufacturing workflow and improving the final isolated yield.
How to Synthesize Benzophenone Derivatives Efficiently
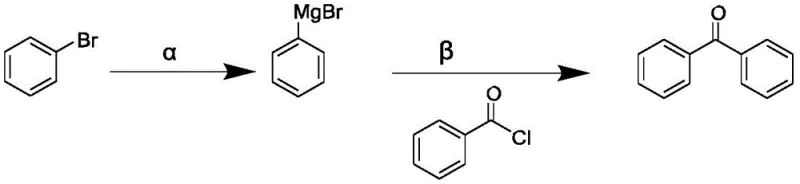
The implementation of this continuous flow methodology requires a systematic approach to reagent preparation and flow parameter optimization. The process begins with the in-situ or separate preparation of the aryl Grignard reagent, typically generated by reacting an aryl bromide with magnesium turnings in 2-methyltetrahydrofuran under an inert nitrogen atmosphere. Once the Grignard solution is prepared, it is pumped alongside a solution of the corresponding acyl chloride into the microreactor system. The precision of the metering pumps is vital here, as maintaining the correct stoichiometric ratio and flow rate ensures optimal conversion. The reaction mixture then traverses the residence time unit, where the transformation occurs under controlled thermal conditions before being quenched and processed.
- Preparation of Aryl Grignard Reagent: React aryl bromide with magnesium turnings in 2-methyltetrahydrofuran at 50°C under nitrogen atmosphere.
- Continuous Flow Reaction: Pump the Grignard solution and acyl chloride solution separately into a mixing module at 25°C with a residence time of 60 minutes.
- Workup and Purification: Quench the effluent with hydrochloric acid, separate phases, wash with brine, and recover the solvent via distillation to isolate the crude product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this continuous flow technology translates into tangible strategic advantages that extend far beyond simple chemical yield improvements. The shift from batch to continuous processing fundamentally alters the cost structure and risk profile of manufacturing benzophenone derivatives. By eliminating the need for expensive cryogenic cooling infrastructure and reducing the reliance on hazardous Lewis acid catalysts, the operational expenditure (OPEX) is significantly lowered. The ability to recycle the reaction solvent further contributes to cost reduction in pharmaceutical intermediate manufacturing, as solvent purchase and waste disposal costs are among the largest variable expenses in fine chemical production. Additionally, the enhanced safety profile reduces insurance premiums and minimizes the risk of production stoppages due to safety incidents, ensuring a more stable and predictable supply for downstream customers.
- Cost Reduction in Manufacturing: The transition to room temperature operation eliminates the massive energy consumption associated with maintaining -20°C conditions in large batch reactors. Furthermore, the high atom economy and reduced formation of by-products mean that raw materials are utilized more efficiently, lowering the cost per kilogram of the final product. The recycling of 2-methyltetrahydrofuran creates a closed-loop system that drastically cuts down on solvent procurement costs, providing a competitive pricing advantage in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Traditional batch processes are prone to variability and batch-to-batch inconsistencies, which can lead to supply disruptions if a batch fails quality control. The continuous nature of this microreactor process ensures a steady, uninterrupted output of high-purity material. The "no amplification effect" characteristic means that scaling up production to meet surging demand does not require extensive re-optimization or risk performance drops. This reliability makes the supplier a more dependable partner for long-term contracts, securing the supply chain against volatility and ensuring just-in-time delivery capabilities for critical drug substances.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental protection intensify globally, the green credentials of this process become a major asset. The reduction in hazardous waste generation and the use of a recyclable, greener solvent position the manufacturing process favorably against strict environmental regulations. The modular nature of microreactor systems allows for flexible capacity expansion; increasing production volume can be achieved by running the system for longer periods or numbering up reactor units, rather than building massive new infrastructure. This scalability ensures that the supply can grow in tandem with market demand while maintaining full compliance with environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the continuous flow synthesis of benzophenone derivatives. These insights are derived directly from the patented technology and practical implementation data, providing clarity on how this method compares to legacy processes. Understanding these details is essential for technical teams evaluating the feasibility of integrating this technology into their existing supply chains or R&D pipelines.
Q: How does the microreactor method improve safety compared to traditional batch Grignard reactions?
A: Traditional batch Grignard reactions are highly exothermic and require cryogenic conditions (-20°C) to prevent runaway reactions. The microreactor method utilizes superior heat transfer capabilities to maintain the reaction safely at room temperature (25°C), eliminating the need for energy-intensive cooling and significantly reducing thermal risk.
Q: What is the yield improvement achieved by this continuous flow process?
A: While traditional batch methods for synthesizing benzophenone derivatives via Grignard reagents often suffer from low yields around 10-20% due to side reactions and poor mixing, this continuous flow technology consistently achieves yields exceeding 60%, with some optimized conditions reaching up to 85%.
Q: Is the solvent used in this process environmentally sustainable?
A: Yes, the process utilizes 2-methyltetrahydrofuran (2-MeTHF), which is a greener alternative to traditional ethers. Furthermore, the continuous flow setup allows for the efficient recovery and recycling of the solvent through an integrated distillation module, minimizing waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzophenone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing technologies like continuous flow microreactors is key to remaining competitive in the global fine chemicals market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN108409516B are fully realized in practical, large-scale operations. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our capability to handle complex chemistries, such as the moisture-sensitive Grignard reactions described herein, underscores our position as a leader in the synthesis of high-value pharmaceutical intermediates.
We invite potential partners to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your project requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to innovation and quality can drive value for your organization and secure your supply of critical benzophenone derivatives.
