Scalable Synthesis of Chiral Triazole Alcohols via Organozinc Addition for Commercial API Production
Introduction to Advanced Triazole Intermediate Manufacturing
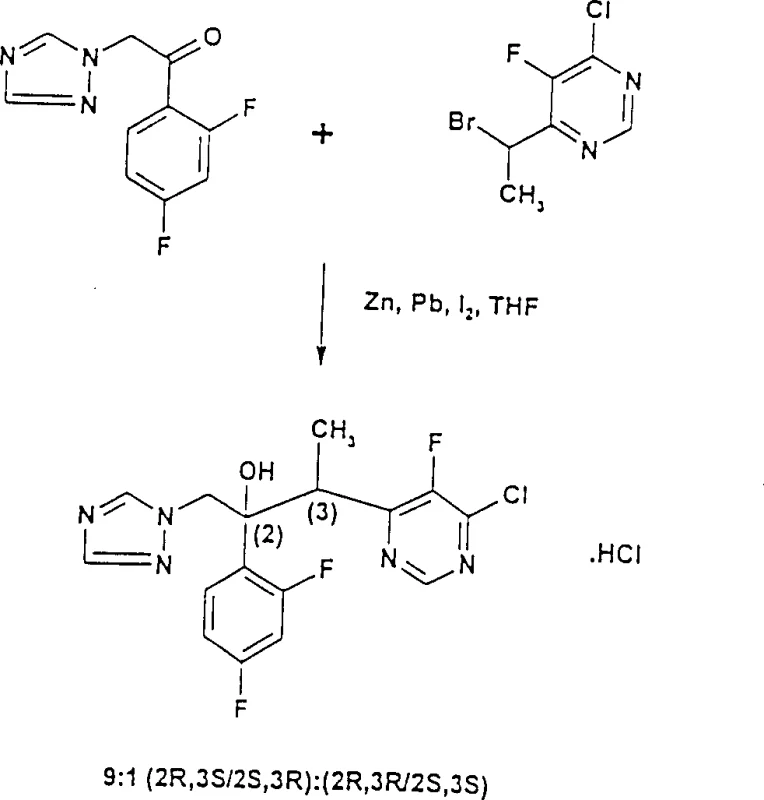
The pharmaceutical industry continuously seeks robust synthetic routes for complex chiral intermediates, particularly those serving as precursors to potent antifungal agents. Patent CN1076019C introduces a transformative methodology for the preparation of triazole compounds through the organometallic addition to ketones, specifically addressing the limitations of prior art methods. This innovation centers on the reaction of 1-phenyl-2-(1H-1,2,4-triazol-1-yl)ethanone derivatives with organometallic compounds derived from alpha-haloalkylpyrimidines. Unlike traditional approaches that rely on harsh cryogenic conditions and suffer from poor stereocontrol, this novel process utilizes a zinc-mediated system enhanced by iodine and optionally lead. The result is a highly stereoselective formation of tertiary alcohols, specifically favoring the (2R,3S/2S,3R) enantiomer pairs which are critical for biological activity. For procurement and technical teams, this represents a significant leap forward in process reliability, offering a pathway to high-purity intermediates with reduced operational complexity and improved safety profiles suitable for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key antifungal intermediates such as voriconazole precursors relied heavily on organolithium chemistry. As detailed in the background of the patent, previous methods involved the addition of organolithium derivatives to ketones at extremely low temperatures, typically ranging from -70°C to -50°C. These cryogenic conditions impose severe engineering constraints on manufacturing facilities, requiring specialized equipment and substantial energy consumption for cooling. Furthermore, the organolithium approach is plagued by competitive enolization side reactions, which drastically reduce the overall yield of the desired product. The stereoselectivity achieved was often suboptimal, resulting in approximately equimolar amounts of undesired enantiomers that required difficult and costly chromatographic separation. Consequently, the total isolated yield of the four stereoisomers was often limited to around 50%, rendering the process economically unviable for large-scale production and creating significant bottlenecks in the supply chain for high-demand antifungal medications.
The Novel Approach
In stark contrast, the process disclosed in CN1076019C utilizes an organozinc reagent generated in situ from alpha-haloalkylpyrimidines and activated zinc dust. This method operates under remarkably mild conditions, with reaction temperatures preferably maintained between -10°C and +15°C, eliminating the need for extreme cryogenics. The inclusion of iodine and optional lead promoters facilitates the formation of reactive organozinc species that exhibit superior nucleophilicity and stereocontrol. Experimental data from the patent demonstrates that this approach can achieve diastereomeric ratios as high as 9:1 or even 10.3:1 favoring the desired (2R,3S/2S,3R) configuration. Moreover, the total isolated yield of all enantiomers, when converted to the hydrochloride salt, can reach up to 65%, representing a substantial improvement over the 50% ceiling of previous methods. This shift not only simplifies the thermal management of the reactor but also significantly enhances the throughput and economic efficiency of the manufacturing process.
Mechanistic Insights into Zinc-Mediated Stereoselective Addition
The core of this technological advancement lies in the unique activation of the zinc metal and the subsequent formation of the organozinc intermediate. The patent specifies that commercially available zinc powder can be activated by stirring in a solvent like tetrahydrofuran (THF), often in the presence of lead. The lead, whether present as an impurity or added intentionally as powder or salt, plays a crucial role in modifying the zinc surface potential, thereby enhancing its reactivity towards the alpha-haloalkylpyrimidine substrate. Simultaneously, the addition of iodine is suspected to generate zinc iodide in situ, which acts as a Lewis acid catalyst. This Lewis acid likely coordinates with the carbonyl oxygen of the ketone substrate, organizing the transition state in a manner that favors the attack of the organozinc species from a specific face. This coordination is pivotal for achieving the high levels of stereoselectivity observed, directing the formation of the (2R,3S/2S,3R) enantiomer pair over the thermodynamically less favorable or kinetically slower alternatives.
Furthermore, the choice of solvent and the rigorous exclusion of moisture are critical mechanistic factors. The reaction is conducted in aprotic organic solvents such as THF, toluene, or dichloromethane, which must be dried thoroughly to prevent the premature quenching of the sensitive organozinc reagent. The protocol involves adding the iodine and Lewis acid to the zinc slurry before introducing the substrates, ensuring that the active catalytic species are fully formed prior to the nucleophilic addition step. The reaction mixture is then allowed to warm to room temperature after the initial addition, promoting the completion of the transformation without triggering decomposition pathways. This controlled environment minimizes side reactions such as enolization or reduction, ensuring that the carbon-carbon bond formation proceeds with high fidelity. The resulting crude product contains a high proportion of the desired diastereomers, which can be further purified through standard crystallization techniques, leveraging the solubility differences of the hydrochloride salts to achieve pharmaceutical-grade purity.
How to Synthesize Triazole Alcohol Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for reproducing these high-yielding results in a laboratory or pilot plant setting. The process begins with the activation of zinc dust, often involving a reflux step in THF under nitrogen to remove surface oxides and ensure maximum reactivity. Once the zinc slurry is prepared and cooled, the iodine promoter is introduced to generate the active zinc iodide species. The substrates, specifically the triazole ketone and the bromo-pyrimidine derivative, are then added slowly to maintain thermal control. This careful addition profile is essential to manage the exotherm and maintain the optimal temperature window for stereoselectivity. Following the reaction period, the mixture is quenched with aqueous acetic acid, which safely destroys excess organometallic reagents and solubilizes zinc salts for removal. The detailed standardized synthesis steps for this specific transformation are provided in the guide below.
- Activate zinc dust with lead and iodine in anhydrous THF under inert atmosphere to generate the reactive organozinc species.
- Add the alpha-haloalkylpyrimidine and triazole ketone substrates slowly while maintaining temperature between -10°C and +15°C.
- Quench the reaction with acetic acid and water, followed by extraction and crystallization to isolate the hydrochloride salt with high diastereomeric ratio.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this zinc-mediated process offers tangible strategic benefits beyond mere chemical yield. The elimination of cryogenic requirements translates directly into reduced capital expenditure on specialized cooling infrastructure and lower operational energy costs. Traditional organolithium processes demand significant utilities to maintain temperatures below -50°C, whereas this new method operates near ambient or slightly cooled conditions, allowing for the utilization of standard jacketed reactors found in most multipurpose chemical plants. This compatibility with existing infrastructure drastically reduces the barrier to entry for contract manufacturing organizations (CMOs) and internal production teams, facilitating faster technology transfer and quicker time-to-market for new drug candidates. Additionally, the reagents employed—zinc dust, iodine, and lead—are commodity chemicals with stable global supply chains, mitigating the risk of raw material shortages that can plague more exotic organometallic reagents.
- Cost Reduction in Manufacturing: The process significantly lowers production costs by removing the need for expensive and hazardous organolithium reagents, which often require specialized handling and storage. By utilizing zinc dust, a low-cost and widely available metal, the direct material cost of the reaction is minimized. Furthermore, the improved stereoselectivity reduces the burden on downstream purification steps; since the crude product already possesses a high diastereomeric ratio, the need for extensive chromatographic separation is diminished or eliminated. This reduction in purification complexity leads to substantial savings in solvent consumption, silica gel usage, and labor hours, contributing to a leaner and more cost-effective manufacturing workflow that enhances overall margin potential for the final API.
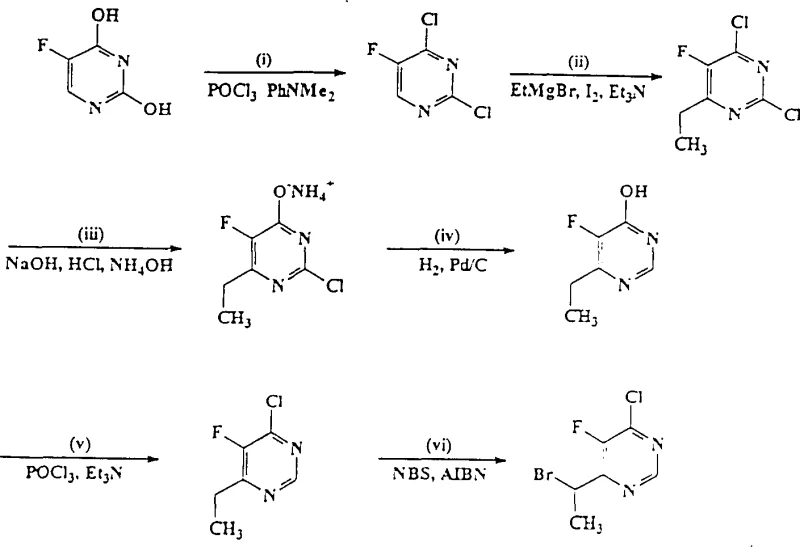
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, a critical factor for maintaining regulatory compliance and supply continuity. The tolerance of the process to slight variations in temperature and the use of stable intermediates mean that production schedules are less likely to be disrupted by equipment failures or environmental fluctuations. Moreover, the ability to synthesize the key alpha-haloalkylpyrimidine starting materials via established halogenation routes, as shown in the preparation examples, ensures a secure upstream supply. This vertical integration potential allows manufacturers to control the quality and availability of critical precursors, reducing dependency on external vendors and insulating the supply chain from market volatility.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the shift away from pyrophoric organolithium reagents to solid zinc dust significantly improves workplace safety and reduces the risk of fire hazards. The waste stream generated by the zinc process is primarily composed of zinc salts, which are easier to treat and dispose of compared to the complex lithium waste streams. The process also demonstrates excellent scalability, with examples showing successful execution on multi-kilogram scales without loss of selectivity or yield. This scalability ensures that the method can seamlessly transition from clinical trial material production to commercial tonnage manufacturing, supporting the long-term growth of the product lifecycle without the need for disruptive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this organozinc addition technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is essential for evaluating the feasibility of integrating this route into your existing manufacturing portfolio. The insights provided here cover aspects ranging from reagent specificity to downstream processing strategies.
Q: What is the primary advantage of the organozinc method over organolithium for this synthesis?
A: The organozinc method described in CN1076019C operates at significantly milder temperatures (-10°C to +15°C vs -70°C) and achieves superior stereoselectivity (up to 9:1 or higher dr) compared to the competitive enolization and low yields associated with organolithium reagents.
Q: How does the presence of lead and iodine affect the reaction outcome?
A: Lead acts as a promoter to activate the zinc surface, while iodine facilitates the formation of zinc iodide in situ, which serves as a Lewis acid catalyst to enhance both the reaction rate and the stereocontrol of the nucleophilic addition.
Q: Is this process suitable for large-scale manufacturing of antifungal intermediates?
A: Yes, the process eliminates the need for cryogenic conditions and uses stable, commercially available reagents like zinc dust and THF, making it highly amenable to kilogram-to-tonne scale production with simplified safety protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazole Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. Our team of expert chemists has extensively analyzed the methodologies described in CN1076019C and possesses the technical capability to implement this zinc-mediated process with precision. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move smoothly from development to full-scale manufacturing. Our facilities are equipped with state-of-the-art reactors capable of handling the specific thermal and mixing requirements of organometallic reactions, and our stringent purity specifications guarantee that every batch meets the rigorous demands of global regulatory agencies. With our rigorous QC labs, we ensure that the stereoselectivity and chemical purity of the triazole intermediates are consistently maintained at the highest levels.
We invite you to collaborate with us to leverage this advanced technology for your antifungal drug programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this process can optimize your bill of materials. Please contact us to request specific COA data from our pilot batches and comprehensive route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain partner committed to innovation, quality, and reliability, ensuring the uninterrupted availability of high-purity pharmaceutical intermediates for your global operations.
