Advanced Organozinc Catalysis for High-Purity DMP-266 Intermediate Production
Introduction to Patent CN1106389C: A Breakthrough in Chiral Synthesis
The pharmaceutical industry continuously seeks robust methodologies for constructing chiral centers, particularly for antiretroviral agents like the reverse transcriptase inhibitor DMP-266. Patent CN1106389C presents a transformative approach to synthesizing the key aminoalcohol intermediate required for this drug. Unlike conventional pathways that rely on cumbersome protection and deprotection sequences, this invention leverages efficient enantioselective addition reactions using organozinc reagents. By utilizing a chiral zinc complex mediated by specific amino alcohol auxiliaries, the process achieves direct asymmetric addition to prochiral ketones. This technological leap not only simplifies the synthetic route but also ensures exceptional stereochemical control, delivering intermediates with enantiomeric excess values exceeding 99 percent in optimized embodiments. For global supply chains, this represents a pivotal shift towards more sustainable and economically viable manufacturing of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for DMP-266 intermediates, as referenced in prior art such as USP 5,519,021, typically involve a multi-step sequence that begins with the protection of the aniline nitrogen. Commonly, a p-methoxybenzyl (PMB) group is installed to prevent side reactions during the subsequent nucleophilic addition. This protection step necessitates additional reagents, solvents, and purification stages, inherently increasing the cost of goods sold and the environmental footprint. Furthermore, the deprotection stage required later in the synthesis often involves harsh conditions or expensive oxidants, which can compromise the integrity of sensitive functional groups like the trifluoromethyl moiety. The cumulative effect of these extra steps is a longer lead time for high-purity pharmaceutical intermediates and a reduced overall yield due to material losses at each isolation point. Additionally, the use of lithium acetylides in conventional methods often demands cryogenic temperatures below minus 60 degrees Celsius, imposing significant energy burdens and safety risks on large-scale operations.
The Novel Approach
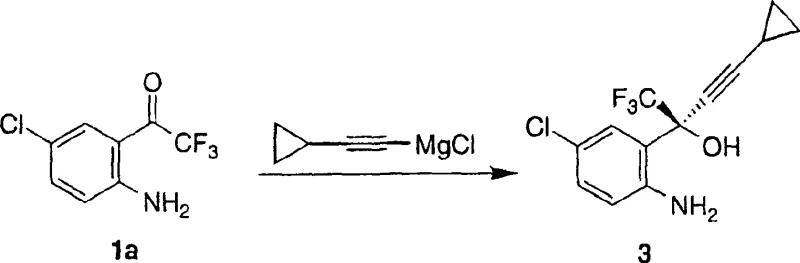
The methodology disclosed in CN1106389C circumvents these inefficiencies by enabling the direct chiral addition of cyclopropylacetylene to the unprotected 2-trifluoromethylcarbonyl-4-chloroaniline. This is achieved through the in situ formation of a highly reactive chiral organozinc complex. The process utilizes a dual-additive system comprising a primary chiral auxiliary, such as (1R,2S)-N-pyrrolidinyl norephedrine, and a secondary additive like neopentyl alcohol or trifluoroethanol. This combination modulates the reactivity of the dialkylzinc species, allowing for high enantioselectivity without the need for nitrogen protection. As illustrated in the reaction scheme below, this streamlined pathway collapses multiple operational units into a single pot operation, drastically reducing solvent consumption and waste generation. The ability to operate at milder temperatures, ranging from minus 20 degrees Celsius to 60 degrees Celsius, further enhances the practicality of this method for industrial application.
Mechanistic Insights into Enantioselective Organozinc Addition
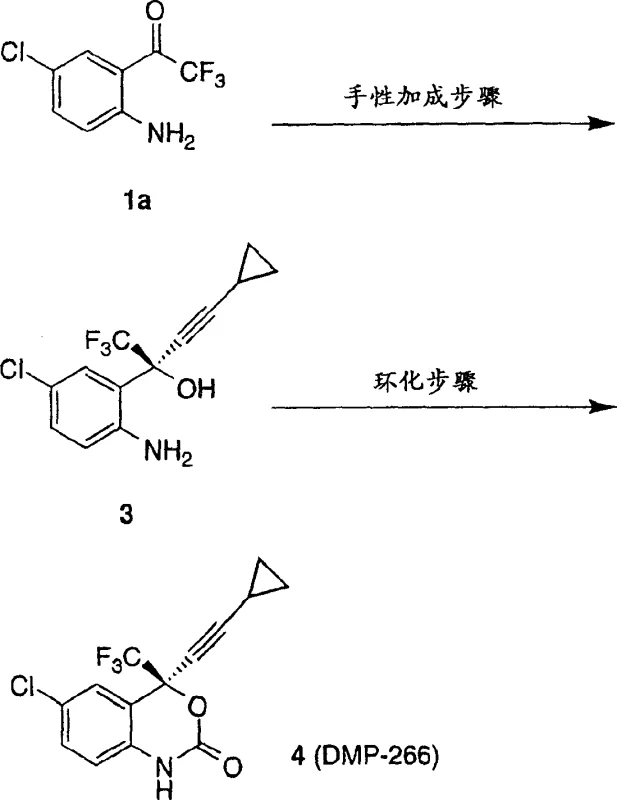
The core of this technology lies in the precise assembly of the chiral organozinc catalyst. The mechanism initiates with the reaction of a dialkylzinc reagent, preferably dimethylzinc or diethylzinc, with the chiral auxiliary in an aprotic solvent such as tetrahydrofuran or toluene. This step generates a chiral zinc alkoxide complex which serves as the stereodirecting environment. The introduction of a second additive, which can be an alcohol, carboxylic acid, or amine, further refines the coordination sphere around the zinc center, enhancing both reaction rate and stereocontrol. Subsequently, the nucleophile, generated separately as a magnesium chloride or lithium salt of cyclopropylacetylene, transmetallates with the chiral zinc complex. This forms the active chiral organozinc species that is poised for asymmetric induction. When the prochiral ketone is introduced, the bulky chiral ligand shields one face of the carbonyl group, forcing the nucleophilic attack to occur from the less hindered trajectory. This steric differentiation is critical for achieving the high optical purity required for API intermediates, ensuring that the resulting aminoalcohol possesses the correct absolute configuration for biological activity.
Impurity control is intrinsically built into this mechanistic design. By avoiding the use of strongly basic lithium reagents directly on the unprotected aniline, the process minimizes the risk of deprotonation at the nitrogen atom, which could lead to polymerization or tar formation. Furthermore, the moderate reaction temperatures prevent thermal degradation of the sensitive trifluoromethyl group. The patent data indicates that careful control of the stoichiometry, specifically using approximately 1.2 equivalents of the chiral auxiliary relative to the ketone, optimizes the balance between conversion and enantioselectivity. Post-reaction workup involving acidic quenching allows for the separation of the product from the zinc salts and facilitates the recovery of the valuable chiral auxiliary from the aqueous phase, thereby closing the loop on material usage and reducing the impurity profile of the final crude solid.
How to Synthesize DMP-266 Intermediate Efficiently
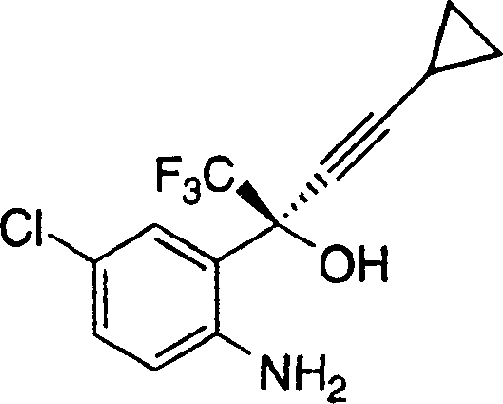
Implementing this synthesis requires strict adherence to inert atmosphere techniques and precise temperature control to maximize the potential of the chiral zinc catalyst. The process is designed to be scalable, moving seamlessly from gram-scale optimization to kilogram-level production without loss of fidelity. Operators must ensure that the formation of the organometallic nucleophile is complete before transmetallation to avoid competing non-selective background reactions. The detailed standardized synthesis steps for this high-efficiency route are provided in the guide below.
- Formation of Chiral Zinc Complex: Under inert atmosphere, mix dialkylzinc with a chiral auxiliary (e.g., N-pyrrolidinyl norephedrine) and a second additive (e.g., neopentyl alcohol) in solvent at -20°C to 50°C.
- Preparation of Nucleophile: Generate cyclopropylacetylide metal salt (MgCl or Li) separately and add to the chiral zinc complex to form the active chiral organozinc species.
- Asymmetric Addition: Add the prochiral ketone (2-trifluoromethylcarbonyl-4-chloroaniline) to the mixture at 0°C to 20°C, stir for extended periods to ensure high enantiomeric excess, then quench and isolate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this organozinc-based methodology offers substantial strategic benefits beyond mere technical elegance. The elimination of the protection-deprotection sequence translates directly into a significantly reduced number of processing steps, which lowers the overall consumption of raw materials, solvents, and utilities. This streamlining effect drives down the variable costs associated with manufacturing, making the final intermediate more price-competitive in the global market. Moreover, the ability to recover and reuse the chiral auxiliary, as demonstrated in the patent examples with recovery rates approaching ninety-four percent, mitigates the cost impact of using specialized ligands. This circular approach to reagent usage aligns with modern green chemistry principles and reduces the dependency on volatile raw material markets.
- Cost Reduction in Manufacturing: The removal of the PMB protection step eliminates the need for expensive protecting group reagents and the subsequent oxidative deprotection chemicals. This simplification reduces the total mass intensity of the process, leading to lower waste disposal costs and reduced solvent purchase requirements. Additionally, the reaction operates efficiently at higher temperatures compared to cryogenic lithiation methods, resulting in significant energy savings on cooling infrastructure. The high isolated yields reported, often exceeding ninety percent, ensure that maximum value is extracted from every kilogram of starting ketone, minimizing the financial loss associated with low-yielding transformations.
- Enhanced Supply Chain Reliability: The reagents required for this process, including dialkylzinc solutions and common Grignard reagents, are commercially available from multiple global suppliers, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions, which tolerate a broader temperature window than traditional methods, makes the process less susceptible to minor fluctuations in plant operating parameters. This reliability ensures consistent batch-to-batch quality and predictable delivery schedules. Furthermore, the simplified workflow reduces the total campaign time, allowing manufacturing facilities to increase throughput and respond more agilely to fluctuations in demand for the final API.
- Scalability and Environmental Compliance: The patent provides explicit data on kilogram-scale runs, demonstrating that the exotherm management and mixing requirements are manageable in standard glass-lined or stainless steel reactors. The use of less hazardous solvents like toluene and heptane, combined with the avoidance of pyrophoric lithium reagents in the main reaction vessel, enhances operational safety. From an environmental perspective, the reduction in step count inherently lowers the E-factor (mass of waste per mass of product), facilitating easier compliance with increasingly stringent environmental regulations. The ability to recycle the aqueous layer for auxiliary recovery further minimizes liquid effluent volume, supporting sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this enantioselective organozinc technology. These insights are derived directly from the experimental data and embodiments within the patent documentation to assist technical teams in evaluating feasibility.
Q: What is the primary advantage of the organozinc method over traditional lithiation routes for DMP-266 intermediates?
A: The organozinc method described in CN1106389C eliminates the need for protecting groups (like PMB) on the aniline nitrogen, significantly reducing step count and waste while maintaining high enantioselectivity (>99% ee).
Q: Can the chiral auxiliary be recovered in this process?
A: Yes, the patent demonstrates that the chiral auxiliary, such as (1R,2S)-N-pyrrolidinyl norephedrine, can be recovered from the aqueous layer after workup with high efficiency (up to 94% recovery), lowering material costs.
Q: What temperature range is critical for maintaining high enantiomeric excess?
A: While the reaction can proceed between -78°C and 70°C, optimal results with >99% ee are achieved by maintaining the addition and stirring phases between -10°C and 20°C, avoiding the extreme cryogenic conditions often required for lithium acetylides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DMP-266 Intermediate Supplier
The technological advancements detailed in CN1106389C highlight the complexity and precision required to produce high-value chiral intermediates for the pharmaceutical sector. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production necessary to bring such sophisticated chemistry to life. Our facility is equipped with state-of-the-art reactor trains capable of handling sensitive organometallic reactions under strict inert conditions, ensuring that the stringent purity specifications required for reverse transcriptase inhibitor intermediates are consistently met. Our rigorous QC labs employ advanced chiral HPLC and NMR spectroscopy to verify enantiomeric excess and chemical purity at every stage of the campaign, guaranteeing a product that meets the highest international standards.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific supply needs. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this streamlined organozinc method. We encourage you to contact us to request specific COA data from our pilot batches and comprehensive route feasibility assessments. Let us collaborate to secure a stable, cost-effective, and high-quality supply of this critical pharmaceutical building block for your global operations.
