Advanced Organozinc Addition for High-Purity DMP-266 Intermediate Commercialization
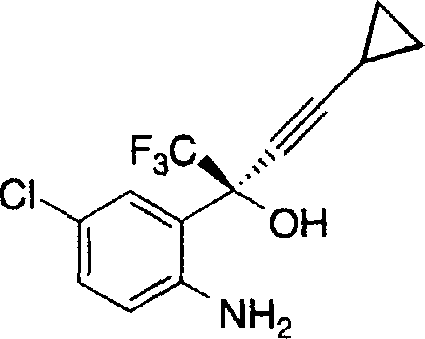
The pharmaceutical industry continuously seeks robust synthetic routes for critical antiviral agents, particularly reverse transcriptase inhibitors like DMP-266. Patent CN1255919A discloses a groundbreaking method for the preparation of key intermediates in the synthesis of (-)-6-chloro-4-cyclopropylethynyl-4-trifluoromethyl-1,4-dihydro-2H-3,1-benzoxazin-2-one. This technology leverages an efficient enantioselective addition reaction using organozinc reagents to transform prochiral ketones into high-value chiral amino alcohols. The significance of this innovation lies in its ability to bypass complex protection strategies that have historically plagued the manufacturing of such complex heterocyclic compounds. By utilizing a chiral zinc complex formed from dialkyl zinc and specific chiral auxiliaries, the process achieves remarkable stereocontrol. This technical advancement provides a reliable pharmaceutical intermediate supplier with a distinct competitive edge in producing high-purity materials essential for antiretroviral therapy formulations. The method's broad applicability to various prochiral ketones further enhances its utility across diverse medicinal chemistry programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzoxazinone derivatives like DMP-266 involved multi-step sequences that were both time-consuming and costly. Prior art, such as that described in USP 5,519,021 and related PCT applications, typically relied on the addition of high enantioselectivity acetylides followed by cyclization. A major drawback of these conventional pathways was the necessity for protecting group chemistry to mask reactive functional groups, specifically the aniline nitrogen, during the nucleophilic addition step. This requirement introduced additional unit operations, including protection, deprotection, and intermediate purification, which inherently lowered the overall yield and increased waste generation. Furthermore, the use of stoichiometric chiral reagents in older methods often resulted in significant material costs and complicated downstream processing. The cumulative effect of these inefficiencies created substantial bottlenecks in cost reduction in API manufacturing, making the final drug substance less accessible for global health initiatives. These legacy processes also posed challenges for commercial scale-up of complex pharmaceutical intermediates due to the sensitivity of protection steps to reaction conditions.
The Novel Approach
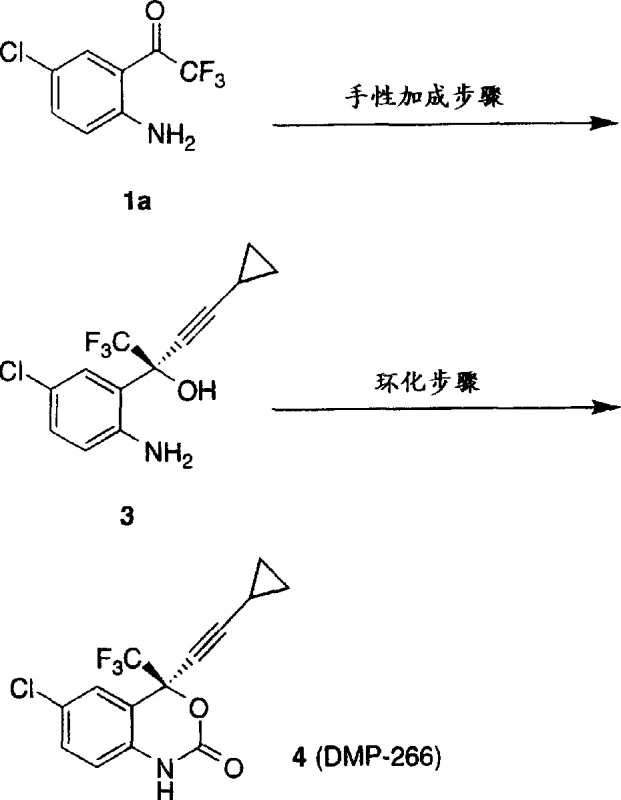
The methodology outlined in CN1255919A represents a paradigm shift by enabling a direct chiral addition to the ketone substrate without prior protection of the aniline moiety. This novel approach utilizes a chiral organozinc complex that exhibits high chemoselectivity, allowing the nucleophilic attack to occur exclusively at the carbonyl center while leaving the amine group intact. By eliminating the protection-deprotection cycle, the process drastically simplifies the synthetic route, reducing the number of isolation steps and solvent exchanges required. This streamlining not only accelerates the production timeline but also significantly reduces the consumption of raw materials and reagents. The ability to perform this transformation under relatively mild conditions, ranging from -20°C to 60°C, further enhances operational safety and equipment compatibility. Consequently, this innovation offers a pathway for reducing lead time for high-purity chiral intermediates, providing a strategic advantage for supply chain heads managing tight production schedules. The direct nature of the reaction ensures a more atom-economical process, aligning with modern green chemistry principles.
Mechanistic Insights into Organozinc-Catalyzed Enantioselective Addition
The core of this technology lies in the formation of a highly reactive and stereoselective chiral organozinc species. The mechanism initiates with the reaction of a dialkyl zinc reagent, such as dimethyl zinc or diethyl zinc, with a chiral auxiliary like (1R, 2S)-N-pyrrolidyl norephedrine. This interaction generates a chiral zinc complex that serves as the template for asymmetric induction. The addition of a secondary auxiliary agent, which can be an alcohol or acid, further modulates the reactivity and selectivity of the zinc center. Subsequent transmetallation with an organometallic reagent, such as cyclopropyl acetylene magnesium chloride, transfers the nucleophilic alkynyl group to the chiral zinc framework. This resulting chiral organozinc complex then attacks the prochiral ketone substrate with high facial selectivity. The precise coordination geometry around the zinc atom ensures that the nucleophile approaches the carbonyl group from a specific direction, thereby establishing the desired stereocenter with exceptional fidelity. This mechanistic understanding is crucial for R&D directors focusing on purity and impurity profiles, as it explains the high enantiomeric excess observed in the final product.
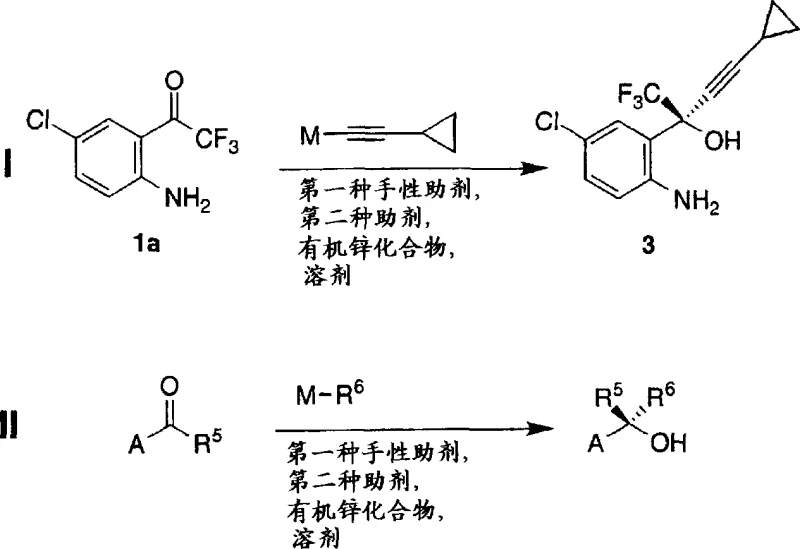
Controlling the impurity profile is paramount in pharmaceutical manufacturing, and this organozinc method offers inherent advantages in this regard. The high chemoselectivity of the chiral zinc reagent minimizes side reactions such as over-addition or attack on other electrophilic sites within the molecule. Furthermore, the use of specific chiral auxiliaries allows for fine-tuning of the reaction conditions to suppress the formation of diastereomers. The patent data indicates that by selecting appropriate secondary auxiliaries like neopentyl alcohol or trifluoroethanol, the enantiomeric excess can be pushed to levels exceeding 99%. This high level of stereocontrol reduces the burden on downstream purification processes, such as chiral chromatography or recrystallization, which are often costly and yield-losing. For quality assurance teams, this means a more consistent product quality with fewer batches rejected due to out-of-specification chiral purity. The robustness of the mechanism against minor variations in temperature and stoichiometry further ensures batch-to-batch reproducibility, a critical factor for regulatory compliance and commercial viability.
How to Synthesize DMP-266 Intermediate Efficiently
The synthesis of the key amino alcohol intermediate involves a sequence of precise steps designed to maximize yield and stereochemical integrity. The process begins with the preparation of the chiral zinc complex under an inert atmosphere, typically nitrogen or argon, to prevent oxidation of the sensitive organometallic species. Careful control of temperature during the addition of dialkyl zinc to the chiral auxiliary is essential to manage exotherms and ensure proper complexation. Once the active chiral species is generated, the organometallic nucleophile is introduced, followed by the slow addition of the ketone substrate to maintain reaction homogeneity. The detailed standardized synthesis steps see the guide below.
- Form a chiral zinc complex by reacting a chiral auxiliary like N-pyrrolidyl norephedrine with dialkyl zinc in an inert solvent under controlled temperature conditions.
- Generate the organometallic reagent, such as cyclopropyl acetylene magnesium chloride, and mix it with the chiral zinc complex to form the active chiral organozinc species.
- Add the prochiral ketone substrate to the chiral organozinc solution at low temperature to achieve high enantioselective addition, followed by workup and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this organozinc-based synthesis route offers transformative benefits for procurement and supply chain management. The elimination of protection and deprotection steps directly translates to a reduction in the number of raw materials required, simplifying the procurement landscape and reducing inventory holding costs. By removing the need for specific protecting group reagents and the associated quenching agents, the process becomes less dependent on specialized chemical supplies that may be subject to market volatility. This simplification enhances supply chain reliability by reducing the number of critical path items that could potentially cause production delays. Furthermore, the streamlined process flow reduces the overall manufacturing cycle time, allowing for faster response to market demand fluctuations. For procurement managers, this means a more resilient supply base capable of adapting to sudden increases in order volume without compromising on delivery schedules. The operational efficiency gains also contribute to substantial cost savings, making the final intermediate more price-competitive in the global marketplace.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the significant reduction in unit operations. By bypassing the protection-deprotection sequence, manufacturers save on the costs associated with additional reactors, filtration equipment, and drying cycles. The consumption of solvents is also drastically reduced, as fewer workup and purification steps are needed. Additionally, the high yield and enantioselectivity minimize the loss of valuable starting materials, ensuring that a greater proportion of input costs are converted into saleable product. The use of commercially available chiral auxiliaries and standard organozinc reagents avoids the premium pricing often associated with custom-synthesized catalysts. These factors combine to create a leaner manufacturing model that delivers significant economic value without sacrificing product quality.
- Enhanced Supply Chain Reliability: The reagents utilized in this synthesis, such as dialkyl zinc and common chiral amino alcohols, are widely available from multiple global suppliers. This multi-sourcing capability mitigates the risk of supply disruptions that can occur when relying on single-source specialty chemicals. The robustness of the reaction conditions also means that the process is less susceptible to variations in raw material quality, further stabilizing the supply chain. For supply chain heads, this translates to improved forecast accuracy and reduced safety stock requirements. The ability to scale the process from gram to kilogram levels, as demonstrated in the patent examples, ensures that the technology can grow with demand. This scalability provides a secure foundation for long-term supply agreements with pharmaceutical partners.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard industrial solvents like toluene and THF that are easily managed in large-scale facilities. The reduction in chemical waste generated by eliminating protection steps aligns with increasingly stringent environmental regulations. Lower waste volumes mean reduced costs for waste treatment and disposal, contributing to a smaller environmental footprint. The high atom economy of the direct addition reaction ensures efficient use of resources, a key metric for sustainable manufacturing. For organizations committed to green chemistry initiatives, this method offers a clear pathway to reducing environmental impact while maintaining commercial productivity. The simplified waste stream also facilitates easier regulatory compliance and permitting for new manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enantioselective organozinc addition technology. These answers are derived directly from the experimental data and process descriptions found within the patent documentation. They are intended to provide clarity on the feasibility, performance, and strategic value of adopting this synthetic route for pharmaceutical intermediate production. Understanding these details is essential for stakeholders evaluating the technology for integration into their existing manufacturing portfolios.
Q: How does this organozinc method improve upon conventional synthesis routes for DMP-266 intermediates?
A: Conventional methods often require multiple protection and deprotection steps to manage functional group compatibility during the addition reaction. This patented organozinc approach enables direct enantioselective addition to the ketone without protecting the aniline nitrogen, significantly simplifying the process flow and reducing material consumption.
Q: What level of enantiomeric excess can be achieved using this chiral auxiliary system?
A: The patent data demonstrates that by optimizing the chiral auxiliary and secondary auxiliary agents, enantiomeric excess values exceeding 99% can be consistently achieved. Specific examples utilizing N-pyrrolidyl norephedrine with neopentyl alcohol show exceptional stereocontrol suitable for strict pharmaceutical specifications.
Q: Is this synthesis method scalable for industrial commercial production?
A: Yes, the patent includes working examples conducted on a kilogram scale, demonstrating the feasibility of commercial scale-up. The use of readily available reagents like dialkyl zinc and standard solvents such as toluene and THF supports robust supply chain continuity and large-scale manufacturing capabilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DMP-266 Intermediate Supplier
The technical potential of the organozinc-mediated enantioselective addition described in CN1255919A is immense, offering a superior route for producing high-value antiviral intermediates. NINGBO INNO PHARMCHEM stands ready to leverage this advanced chemistry as part of our comprehensive CDMO service offerings. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required by global regulatory agencies. Our commitment to technical excellence ensures that the high enantiomeric excess and yield demonstrated in the patent are consistently replicated at commercial scale.
We invite pharmaceutical partners to engage with our technical procurement team to explore how this technology can optimize your supply chain. Request a Customized Cost-Saving Analysis to understand the specific economic benefits applicable to your project. We are prepared to provide specific COA data and route feasibility assessments to support your decision-making process. By collaborating with us, you gain access to a partner dedicated to driving efficiency and quality in the manufacturing of complex pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
