Advanced Synthesis of Allyl Quinone Derivatives for High-Purity Pharmaceutical Intermediates
Advanced Synthesis of Allyl Quinone Derivatives for High-Purity Pharmaceutical Intermediates
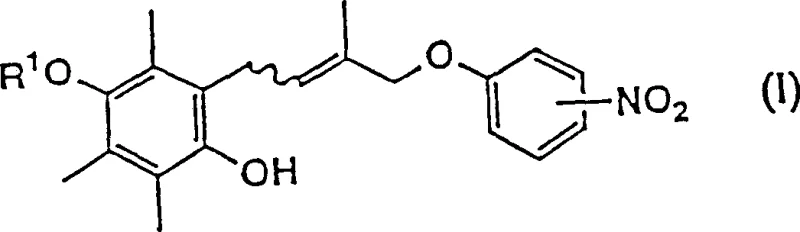
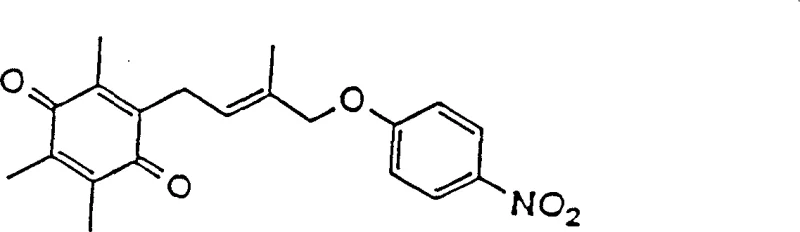
The pharmaceutical industry continuously seeks robust synthetic routes for complex intermediates that serve as the backbone for next-generation therapeutics. Patent CN1150159C introduces a groundbreaking methodology for the preparation of allyl quinone derivatives, specifically targeting the synthesis of 2-[4'-(p-nitro-phenoxy)-3'-methyl-2'-butenyl]-3,5,6-trimethyl-1,4-benzoquinone. This compound is a critical precursor for chroman derivatives such as CS-045, a potent hypoglycemic agent. The core innovation lies in the strategic replacement of highly reactive and unstable allyl halides with stable allyl alcohol derivatives, fundamentally altering the reaction landscape to favor higher yields and superior purity profiles. By leveraging this patented approach, manufacturers can overcome the historical limitations associated with Friedel-Crafts alkylations involving sensitive olefinic halides, thereby establishing a more reliable supply chain for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in EP-543,346, relied heavily on the use of 1-chloro-2-methyl-3-butene-2-alcohol as the alkylating agent. While theoretically viable, this allyl halide possesses inherent chemical instability and extreme reactivity that poses significant challenges for industrial scale-up. The high reactivity leads to uncontrolled side reactions, generating a complex mixture of degradation products and structural isomers that are notoriously difficult to separate from the desired product. Furthermore, the necessity for in-situ preparation of the allyl halide due to its instability introduces severe operational hazards, including exposure to pungent vapors and the risk of thermal runaway. These factors collectively result in low isolated yields and inconsistent batch-to-batch quality, rendering the conventional route economically unviable for large-scale commercial production of high-purity API intermediates.
The Novel Approach
The novel approach detailed in CN1150159C circumvents these pitfalls by utilizing stable uncle's allyl alcohol derivatives, specifically compounds of formula (V) or (VI), as the alkylating agents. This strategic shift allows for the direct condensation with trimethylhydroquinone derivatives under controlled Friedel-Crafts conditions without the formation of hazardous halide byproducts. The stability of the alcohol precursors ensures a cleaner reaction profile, significantly minimizing the generation of isomeric impurities and simplifying downstream purification processes. This method not only enhances the safety profile of the manufacturing operation but also delivers substantially improved yields, making it an industrially suitable method for the reliable production of complex quinone derivatives required for modern medicine synthesis.
Mechanistic Insights into Friedel-Crafts Alkylation and Oxidative Dehydrogenation
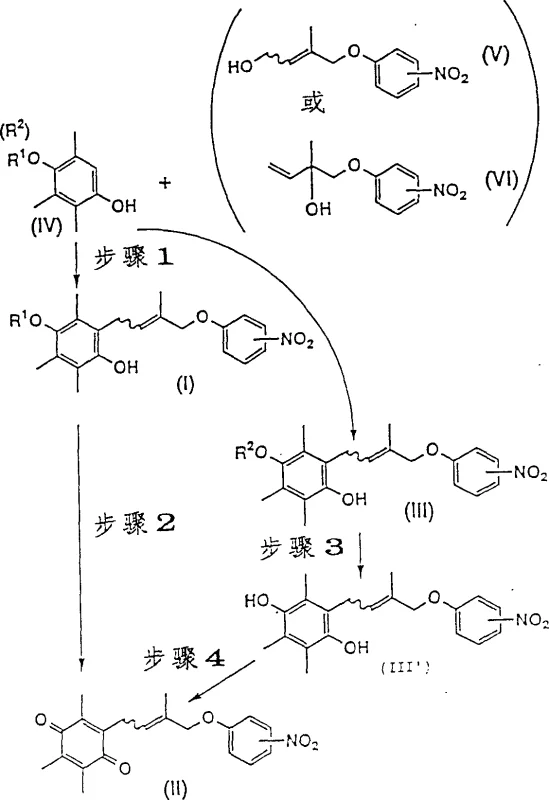
The core of this synthesis relies on a Lewis acid-catalyzed Friedel-Crafts alkylation where the allyl alcohol derivative acts as the electrophile source. In the presence of catalysts such as zinc chloride, aluminum chloride, or boron trifluoride-ether complexes, the hydroxyl group of the allyl alcohol is activated, facilitating the formation of a carbocation-like species that attacks the electron-rich aromatic ring of the trimethylhydroquinone derivative. The inclusion of silica gel in the reaction matrix has been observed to further enhance yields, likely by adsorbing water produced during the condensation and shifting the equilibrium towards product formation. This mechanistic pathway avoids the harsh conditions typically required for halide activation, preserving the integrity of the sensitive olefinic double bond and preventing polymerization or rearrangement side reactions that plague traditional methods.
Following the alkylation step, the resulting hydroquinone derivative must be oxidized to the corresponding quinone. The patent elucidates multiple viable oxidation mechanisms, ranging from stoichiometric oxidants like manganese dioxide and ceric ammonium nitrate to catalytic systems utilizing air. In the catalytic air oxidation protocol, the combination of cupric chloride and lithium chloride serves as a redox mediator, efficiently transferring electrons from the hydroquinone to molecular oxygen. This biomimetic approach is particularly advantageous for green chemistry initiatives, as it replaces toxic heavy metal oxidants with benign atmospheric oxygen. The ability to tune the oxidation potential allows for precise control over the reaction, ensuring complete conversion to the quinone without over-oxidation of the sensitive nitro-phenoxy side chain, thus maintaining the structural fidelity required for subsequent pharmaceutical transformations.
How to Synthesize Allyl Quinone Derivatives Efficiently
The synthesis of these high-value intermediates requires precise control over reaction parameters to maximize yield and purity. The process begins with the careful selection of the allyl alcohol precursor, followed by a Lewis acid-mediated coupling with the hydroquinone core. Subsequent oxidation steps convert the coupled product into the final quinone derivative. Detailed operational procedures, including specific solvent choices, temperature gradients, and workup protocols, are critical for reproducing the high yields reported in the patent literature. For a comprehensive guide on executing this synthesis with industrial precision, please refer to the standardized protocol outlined below.
- Perform Friedel-Crafts alkylation between trimethylhydroquinone derivatives and stable allyl alcohol derivatives using Lewis acid catalysts like zinc chloride.
- Execute oxidative dehydrogenation of the resulting hydroquinone intermediate using manganese dioxide or catalytic air oxidation systems.
- Purify the final quinone derivative through recrystallization or column chromatography to achieve high pharmaceutical grade purity.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this novel synthetic route offers profound commercial benefits for procurement and supply chain stakeholders, primarily driven by the enhanced stability of raw materials and the simplification of processing steps. The elimination of unstable allyl halides removes the need for complex in-situ generation units and specialized containment systems, leading to a drastic reduction in capital expenditure and operational complexity. Furthermore, the cleaner reaction profile translates to reduced solvent consumption during purification and lower waste disposal costs, contributing to a more sustainable and cost-effective manufacturing footprint. These efficiencies collectively strengthen the supply chain resilience by minimizing the risk of batch failures and ensuring consistent availability of critical intermediates.
- Cost Reduction in Manufacturing: The transition to stable allyl alcohol derivatives eliminates the expensive and hazardous handling requirements associated with reactive allyl halides. By avoiding the need for in-situ preparation and reducing the formation of difficult-to-remove isomers, the overall process mass intensity is significantly lowered. This streamlined workflow reduces the consumption of auxiliary chemicals and solvents, driving down the variable cost per kilogram of the final intermediate while simultaneously improving the overall process economics through higher effective throughput.
- Enhanced Supply Chain Reliability: The use of commercially stable starting materials mitigates the risk of supply disruptions caused by the short shelf-life of traditional reagents. Manufacturers can maintain larger inventories of key precursors without degradation concerns, ensuring continuous production capability even during fluctuations in raw material availability. Additionally, the robustness of the reaction conditions reduces the likelihood of unplanned downtime due to safety incidents or off-spec batches, providing a more predictable and reliable delivery schedule for downstream API producers.
- Scalability and Environmental Compliance: The compatibility of this method with catalytic air oxidation systems offers a clear pathway for green manufacturing at scale. Replacing stoichiometric heavy metal oxidants with atmospheric oxygen significantly reduces the generation of hazardous metal waste, simplifying effluent treatment and ensuring compliance with increasingly stringent environmental regulations. This environmental advantage not only lowers disposal costs but also enhances the corporate sustainability profile, making the supply chain more attractive to eco-conscious pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. Understanding these nuances is essential for R&D teams evaluating the feasibility of technology transfer and for quality assurance personnel establishing control strategies. The answers provided are derived directly from the experimental data and comparative analysis presented in the patent documentation, ensuring accuracy and relevance for industrial application.
Q: Why does this patent avoid using allyl halides like 1-chloro-2-methyl-3-butene-2-alcohol?
A: Conventional allyl halides exhibit extremely high reactivity and instability, leading to significant formation of degradation products and structural isomers that are difficult to isolate. This new method utilizes stable allyl alcohol derivatives, drastically reducing byproduct formation and improving operational safety by eliminating pungent and unstable reagents.
Q: What oxidation systems are compatible with this synthesis route?
A: The process supports versatile oxidation protocols including stoichiometric manganese dioxide, ceric ammonium nitrate, and highly scalable catalytic systems utilizing air, lithium chloride, and cupric chloride. This flexibility allows manufacturers to choose between high-yield batch processes or cost-effective continuous air oxidation methods.
Q: How does this method improve the impurity profile for API production?
A: By replacing unstable halide precursors with stable alcohol derivatives, the generation of complex structural isomers is minimized. Furthermore, the use of mild oxidation conditions prevents over-oxidation and ring degradation, resulting in a cleaner crude product that requires less intensive purification to meet stringent pharmaceutical specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allyl Quinone Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN1150159C can be seamlessly translated from the laboratory to the plant. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of allyl quinone derivative meets the exacting standards required for pharmaceutical synthesis, providing our partners with the confidence needed to advance their drug development pipelines.
We invite you to collaborate with us to optimize your supply chain for these essential building blocks. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals in the competitive pharmaceutical market.
