Advanced Synthesis of Allyl Quinone Derivatives for Commercial Scale-Up of Complex Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic routes for critical intermediates, particularly those serving as precursors for hypoglycemic agents like CS-045. Patent CN1062549C discloses a groundbreaking industrial production method for specific quinone derivatives, addressing long-standing challenges in yield and stability. This technology represents a significant leap forward for any reliable pharmaceutical intermediate supplier aiming to optimize their portfolio. By shifting away from unstable allyl halides towards a controlled oxidation and hydrolysis sequence, the process ensures higher purity and operational safety. For R&D teams evaluating new pathways, this patent offers a validated framework for synthesizing 2-[4'-(p-nitro-phenoxy)-3'-methyl-2'-butenyl]-3,5,6-trimethylammonium-1,4-benzoquinones. The implications for cost reduction in API manufacturing are profound, as the streamlined workflow minimizes waste and simplifies downstream processing. Understanding this methodology is essential for stakeholders focused on securing a stable supply chain for complex organic syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those disclosed in EP-543,346, rely heavily on the condensation of trimethylhydroquinone with reactive allyl halides like 1-chloro-2-methyl-3-butene-2-alcohol. This traditional approach suffers from severe drawbacks, primarily due to the inherent instability of the allyl halide reagents. These compounds exhibit high reactivity that leads to the formation of numerous degradation products and structural isomers, making the isolation of the target product exceptionally difficult. Furthermore, the necessity for in-situ preparation of these unstable halides introduces significant operational complexities and security risks, including pungency and handling hazards. The resulting intermediates, such as the chloro-substituted quinhydrones, are also unstable, leading to low productive rates and inconsistent batch quality. Consequently, these conventional methods are often deemed unsuitable for large-scale industrial applications where consistency and safety are paramount.
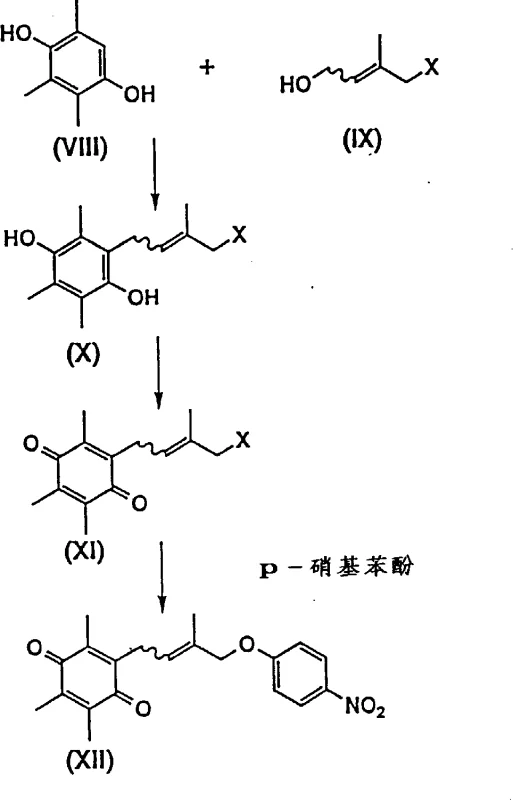
The Novel Approach
In stark contrast, the novel approach outlined in the patent data introduces a sophisticated multi-step strategy that circumvents the pitfalls of direct allyl halide usage. By utilizing stable hydroquinone derivatives and employing a sequence of oxidation and Claisen base hydrolysis, the process achieves high yields with superior safety profiles. This method effectively eliminates the generation of troublesome by-products associated with halide degradation, thereby simplifying the purification process. The use of specific oxidants like Manganese Dioxide or ceric ammonium nitrate allows for precise control over the oxidation state, ensuring the integrity of the molecular structure. For procurement managers, this translates to a more predictable production schedule and reduced risk of batch failures. The ability to produce high-purity quinone derivatives through this refined pathway underscores its value for commercial scale-up of complex pharmaceutical intermediates, offering a sustainable alternative to legacy chemistries.
Mechanistic Insights into Oxidation and Hydrolysis Cascade
The core of this technological advancement lies in its meticulously designed reaction cascade, which prioritizes stability and selectivity at every stage. The process begins with a Friedel-Crafts reaction between a Trimethylhydroquinone derivative and stable uncle's allyl alcohol derivatives, catalyzed by Lewis acids such as zinc chloride or aluminum chloride. This initial step is crucial as it establishes the carbon skeleton without introducing unstable halogenated species. Following this, a controlled oxidation step converts the hydroquinone intermediate into the corresponding quinone, utilizing reagents that minimize over-oxidation. The subsequent hydrolysis using Claisen base at low temperatures is particularly innovative, as it gently removes acyl protecting groups without damaging the sensitive quinone moiety. This mechanistic precision ensures that the final product retains the desired geometric isomerism, whether cis (Z) or trans (E), which is vital for downstream biological activity. Such detailed control over the reaction mechanism is what distinguishes this patent as a benchmark for high-purity OLED material and pharmaceutical intermediate synthesis.
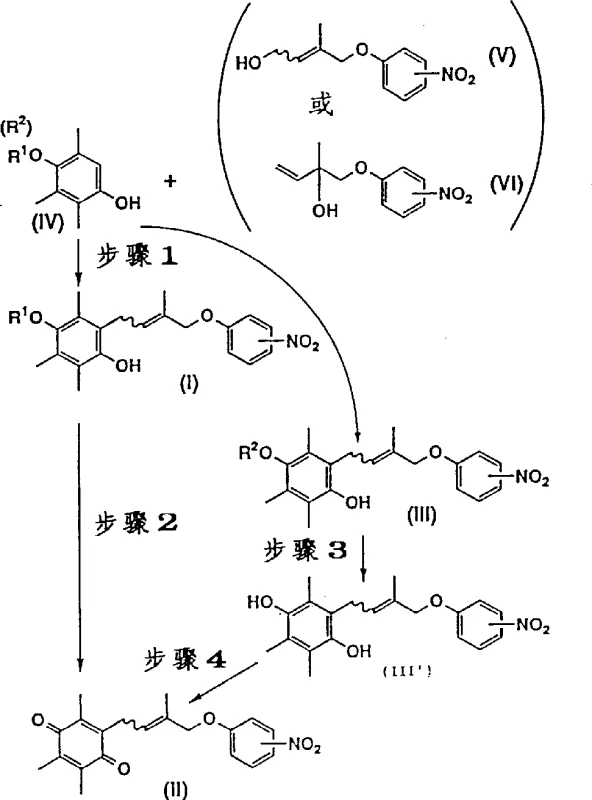
Impurity control is another critical aspect where this mechanism excels, directly addressing the concerns of R&D Directors regarding product specification. By avoiding the chaotic degradation pathways of allyl halides, the new method significantly reduces the burden on downstream purification units like silica gel column chromatography or HPLC. The specific selection of solvents, ranging from methylene dichloride to toluene, is optimized to maintain reagent solubility while facilitating easy separation of by-products. Moreover, the final oxidation step can be tuned using air or chemical oxidants to ensure complete conversion, leaving minimal residual starting material. This level of purity is essential when producing intermediates for potent hypoglycemic agents, where even trace impurities can affect efficacy. The robust nature of this mechanism supports reducing lead time for high-purity API intermediates by minimizing the need for repetitive recrystallization cycles. Ultimately, the chemical elegance of this process provides a solid foundation for consistent, high-quality manufacturing.
How to Synthesize Allyl Quinone Derivatives Efficiently
Implementing this synthesis route requires a clear understanding of the operational parameters defined in the patent to ensure optimal results. The process is divided into distinct stages, starting with the preparation of stable alcohol derivatives followed by condensation and oxidation. Operators must adhere to strict temperature controls, particularly during the hydrolysis step which is preferably carried out at 10°C or lower to prevent side reactions. The detailed standardized synthesis steps below outline the precise conditions for reagents, catalysts, and workup procedures necessary for successful execution. Following these guidelines ensures that the theoretical benefits of the patent are realized in practical production environments. For technical teams, having a clear roadmap is essential for translating laboratory success into plant-floor reality.
- Perform Friedel-Crafts reaction between Trimethylhydroquinone derivative and uncle's allyl alcohol derivatives using zinc chloride or aluminum chloride catalyst.
- Oxidize the resulting hydroquinone derivatives using Manganese Dioxide, ceric ammonium nitrate, or air with cupric chloride to form quinone intermediates.
- Conduct Claisen base hydrolysis on acyl group hydroquinone derivatives at low temperatures to prepare hydrolysates before final oxidation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial benefits that resonate deeply with procurement and supply chain leadership. The elimination of unstable and hazardous allyl halides drastically simplifies the raw material sourcing strategy, reducing dependency on specialty reagents that may have volatile availability. This shift not only enhances supply chain reliability but also mitigates the risks associated with storing and handling dangerous chemicals. For organizations focused on cost reduction in electronic chemical manufacturing or pharma, the streamlined process means fewer unit operations and lower energy consumption per kilogram of product. The high yield and purity achieved reduce the volume of waste generated, aligning with increasingly stringent environmental compliance standards. These factors collectively contribute to a more resilient and cost-effective supply chain, capable of meeting the demanding schedules of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The removal of expensive and unstable allyl halide reagents leads to significant raw material cost savings. By utilizing more stable and commercially available alcohol derivatives, the process avoids the premium pricing often associated with specialized halogenated compounds. Furthermore, the simplified purification requirements mean less solvent usage and reduced waste disposal costs, which are major components of overall manufacturing expenses. The high conversion rates ensure that raw materials are utilized efficiently, minimizing loss and maximizing output per batch. These qualitative improvements in process efficiency translate directly into a more competitive cost structure for the final API intermediate.
- Enhanced Supply Chain Reliability: Stability is the cornerstone of a reliable supply chain, and this process delivers by removing the bottleneck of in-situ reagent preparation. The use of shelf-stable intermediates allows for better inventory management and reduces the risk of production stoppages due to reagent degradation. This reliability is crucial for maintaining continuous supply to downstream manufacturers who depend on just-in-time delivery models. Additionally, the robustness of the reaction conditions means that the process is less susceptible to variations in ambient conditions, further stabilizing output. For supply chain heads, this predictability is invaluable for long-term planning and contract fulfillment.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing common industrial solvents and standard reaction equipment. The avoidance of heavy metal catalysts in certain steps and the use of air as an oxidant in others demonstrate a commitment to greener chemistry principles. This alignment with environmental goals facilitates easier regulatory approval and reduces the burden of effluent treatment. The ability to scale from laboratory to commercial production without fundamental changes to the chemistry ensures a smooth transition during technology transfer. Such scalability supports the growing demand for complex polymer additives and pharmaceutical intermediates without compromising on safety or quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. They are derived from the specific pain points identified in the background art and the beneficial effects highlighted in the patent description. These answers provide clarity on safety, purity, and scalability, helping stakeholders make informed decisions. Understanding these nuances is key to leveraging the full potential of this advanced manufacturing route.
Q: Why is the new synthesis method safer than the conventional Friedel-Crafts approach?
A: The conventional method relies on unstable allyl halides which degrade easily and pose security risks due to pungency and reactivity. The novel approach utilizes stable hydroquinone derivatives and controlled oxidation steps, significantly reducing operational hazards and degradation by-products.
Q: How does this process impact the purity of the final API intermediate?
A: By avoiding the formation of structural isomers common in prior art allyl halide reactions, this method ensures a cleaner reaction profile. The specific sequence of hydrolysis followed by oxidation allows for rigorous impurity control, resulting in high-purity quinone derivatives suitable for sensitive pharmaceutical applications.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process is designed for industrial suitability. It eliminates the need for in-situ preparation of unstable reagents and uses standard solvents like methylene dichloride and toluene. The steps are robust enough for commercial scale-up, ensuring consistent supply continuity for large-volume manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allyl Quinone Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the quinone derivative synthesis are seamlessly integrated into our operations. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that validate every batch against the highest industry standards. Our capability to handle complex chemistries allows us to offer a reliable Allyl Quinone Derivative Supplier partnership that you can trust for your most demanding projects. We understand that consistency and quality are non-negotiable for your downstream applications.
We invite you to engage with our technical procurement team to discuss how this technology can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this novel process. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your production requirements. Let us collaborate to optimize your manufacturing efficiency and secure a sustainable future for your product portfolio.
