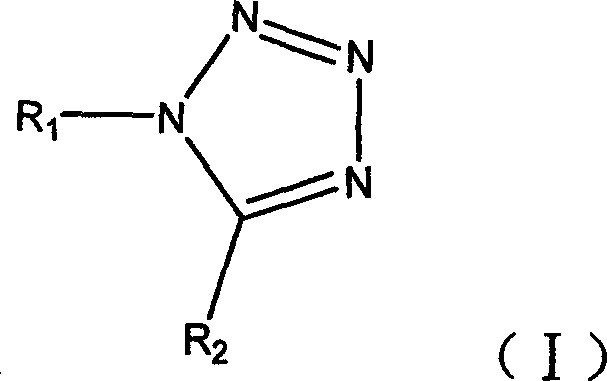
Advanced Tetrazole Synthesis: Leveraging Ytterbium Triflate for Commercial Scale-Up
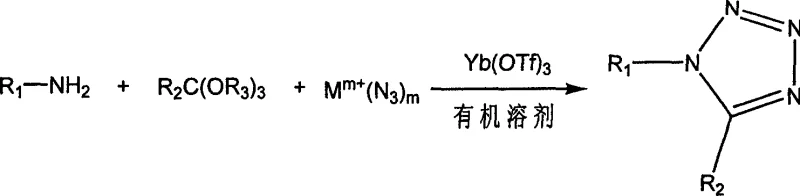
The pharmaceutical and fine chemical industries are constantly seeking more efficient, safer, and environmentally sustainable pathways for synthesizing critical heterocyclic scaffolds. Patent CN1775764A introduces a transformative chemical synthesis method for tetrazole compounds, utilizing ytterbium trifluoromethanesulfonate (Yb(OTf)3) as a highly effective Lewis acid catalyst. This innovation addresses long-standing challenges in tetrazole formation, specifically replacing harsh Brønsted acid conditions with a milder, recyclable catalytic system. By enabling the reaction of amines, crude carboxylic esters (orthoesters), and metal azides in organic solvents at temperatures ranging from 20°C to 150°C, this technology delivers exceptional reaction yields often exceeding 80%. For a reliable pharmaceutical intermediate supplier, adopting such a robust methodology is crucial for maintaining competitive advantage in the global market. The process not only simplifies the operational workflow by avoiding the preparation of amine hydrochlorides but also drastically reduces the environmental footprint by minimizing hazardous waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the development of this ytterbium-catalyzed protocol, the standard industrial practice for synthesizing tetrazoles relied heavily on traditional Brønsted acids such as acetic acid or trifluoroacetic acid, as exemplified by older patents like US3,767,667. These conventional routes typically required the conversion of amines into their corresponding hydrochloride salts before reaction, adding unnecessary synthetic steps and operational complexity. Furthermore, the use of strong acidic media in the presence of sodium azide posed severe safety hazards due to the potential evolution of highly toxic and explosive hydrazoic acid gas. From a supply chain perspective, the difficulty in recovering and purifying excess acetic acid from the reaction mixture created significant downstream processing bottlenecks. The resulting wastewater streams were often laden with acidic residues, necessitating costly neutralization and treatment procedures that inflated the overall production cost in API manufacturing. These inherent inefficiencies made the traditional method less attractive for modern, large-scale commercial operations focused on green chemistry principles.
The Novel Approach
The novel approach detailed in CN1775764A fundamentally shifts the paradigm by employing Yb(OTf)3, a stable and water-tolerant Lewis acid, to drive the three-component coupling reaction. This method allows for the direct use of free amines rather than hydrochloride salts, streamlining the raw material input and reducing pre-reaction preparation time. The reaction proceeds smoothly in a variety of common organic solvents, including chloroform, ethanol, and ethyl acetate, providing flexibility for process optimization. Crucially, the catalytic system operates under much milder conditions, typically between 50°C and 80°C, which enhances energy efficiency and reduces thermal stress on sensitive functional groups. The ability to recover the catalyst from the aqueous phase with a recovery rate of over 95% represents a monumental leap in resource efficiency. This technological advancement supports the commercial scale-up of complex heterocycles by offering a cleaner, safer, and more economically viable pathway that aligns with stringent environmental regulations.
Mechanistic Insights into Yb(OTf)3-Catalyzed Cyclization
The efficacy of this synthesis lies in the unique Lewis acidic properties of ytterbium triflate, which acts as a potent activator for the orthoester component. In the catalytic cycle, the Yb3+ ion coordinates with the oxygen atoms of the triethyl orthoformate, increasing the electrophilicity of the central carbon atom. This activation facilitates the nucleophilic attack by the amine, leading to the formation of an amidine intermediate. Subsequently, the azide ion, supplied by the metal azide salt such as sodium azide or potassium azide, attacks the activated imine carbon. The Lewis acid stabilizes the transition states throughout this cascade, lowering the activation energy barrier and allowing the cyclization to proceed rapidly even at moderate temperatures. Unlike protonic acids which can lead to indiscriminate protonation and side reactions, the specific coordination geometry of the lanthanide catalyst ensures high regioselectivity. This precise mechanistic control is vital for producing high-purity tetrazole derivatives required for sensitive biological applications, ensuring that the final product meets rigorous quality standards without extensive purification.
Impurity control is another critical aspect where this catalytic system excels, particularly when compared to acid-catalyzed methods that often generate polymeric byproducts or degradation products. The mild nature of Yb(OTf)3 prevents the decomposition of the azide reagent into hazardous hydrazoic acid, thereby eliminating a major source of safety risk and impurity formation. Experimental data from the patent indicates that product purities consistently reach levels above 99%, with some examples achieving 99.7% purity after simple recrystallization. This high level of chemical fidelity suggests that the catalyst does not promote unwanted side reactions such as over-alkylation or ring-opening. For R&D teams, this implies a simplified downstream processing workflow where extensive chromatographic purification may be unnecessary. The robustness of the catalyst across a wide range of substrates, including those with electron-withdrawing nitro groups or electron-donating methoxy groups, further demonstrates its versatility in managing diverse impurity profiles effectively.
How to Synthesize 1-Substituted Tetrazoles Efficiently
To implement this synthesis effectively, operators should follow a standardized protocol that maximizes catalyst turnover and product isolation. The process begins with the precise weighing of amine, orthoester, and metal azide in the optimized molar ratios described in the patent, typically around 1:1.5:1.5. These components are dissolved in a suitable solvent like chloroform or ethanol along with a catalytic amount of Yb(OTf)3, usually between 0.02 and 0.05 equivalents. The detailed standardized synthesis steps see the guide below.
- Combine amine, orthoester, metal azide, and Yb(OTf)3 catalyst in an organic solvent such as chloroform or ethanol at room temperature.
- Heat the reaction mixture to 50-80°C and maintain for 2-10 hours while monitoring progress via HPLC.
- Remove solvent, quench with ice water, extract with ethyl acetate, and recrystallize from ethanol to obtain high-purity tetrazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ytterbium-catalyzed tetrazole synthesis offers profound strategic benefits that extend beyond simple yield improvements. The primary value proposition lies in the drastic simplification of the supply chain for raw materials and the reduction of hazardous waste disposal costs. By eliminating the need for amine hydrochlorides and strong mineral acids, the process reduces the number of SKUs required for production and minimizes the regulatory burden associated with handling corrosive substances. Furthermore, the high recovery rate of the expensive ytterbium catalyst means that the effective cost per kilogram of catalyst consumption is negligible over time. This creates a more predictable cost structure, shielding the manufacturing operation from volatility in rare earth metal prices. The ability to run the reaction in common, easily recoverable solvents also enhances the overall economic efficiency of the plant.
- Cost Reduction in Manufacturing: The elimination of stoichiometric amounts of strong acids and the subsequent neutralization steps significantly lowers the consumption of auxiliary chemicals. Since the catalyst is recyclable with high efficiency, the long-term operational expenditure on catalytic materials is substantially reduced compared to single-use acid catalysts. Additionally, the high reaction yields minimize the loss of valuable starting materials, directly improving the material balance and reducing the cost of goods sold. The simplified workup procedure, which avoids complex distillation setups for acid recovery, further decreases energy consumption and labor hours required per batch.
- Enhanced Supply Chain Reliability: Utilizing commercially available and stable reagents such as triethyl orthoformate and sodium azide ensures a steady supply of inputs without reliance on specialized custom synthesis. The robustness of the reaction conditions allows for flexible scheduling, as the process is less sensitive to minor fluctuations in temperature or moisture compared to moisture-sensitive acid chloride methods. This reliability translates to shorter lead times for high-purity pharmaceutical intermediates, enabling manufacturers to respond more agilely to market demand. The reduced safety risks associated with hydrazoic acid generation also mean fewer unplanned shutdowns due to safety incidents or regulatory inspections.
- Scalability and Environmental Compliance: The process generates minimal "three wastes" (waste water, waste gas, waste residue), aligning perfectly with increasingly strict global environmental regulations. The absence of toxic gas evolution simplifies the engineering controls required for scale-up, allowing for larger reactor volumes without proportional increases in scrubbing capacity. The aqueous workup allows for easy separation of the organic product from the catalyst, which remains in the water phase, facilitating a clean separation that is ideal for continuous flow processing. This environmental compatibility enhances the corporate sustainability profile, a key factor for multinational clients evaluating potential partners.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this tetrazole synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these nuances is essential for process engineers and quality assurance teams looking to integrate this method into their existing production lines.
Q: What are the advantages of using Ytterbium Triflate over traditional acetic acid catalysts?
A: Ytterbium Triflate allows for milder reaction conditions, eliminates the generation of toxic hydrazoic acid gas associated with strong acid methods, and enables catalyst recovery rates exceeding 95%, significantly reducing waste.
Q: Can the catalyst be reused in this tetrazole synthesis process?
A: Yes, the patent demonstrates that Yb(OTf)3 can be recovered from the aqueous layer after reaction, dehydrated under vacuum at 190°C, and reused with recovery rates consistently above 95% without significant loss of activity.
Q: What is the typical purity achieved with this synthetic route?
A: Experimental data indicates that the resulting tetrazole compounds consistently achieve purity levels greater than 99%, often reaching 99.7%, making them suitable for direct use in sensitive pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrazole Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to deliver superior chemical solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of tetrazole intermediate meets the highest international standards. Our capability to handle complex heterocyclic synthesis positions us as a strategic partner for companies seeking to optimize their API supply chains.
We invite you to contact our technical procurement team to discuss how this advanced Yb(OTf)3 catalyzed route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this greener synthesis method. We are ready to provide specific COA data and route feasibility assessments to support your R&D and procurement decision-making processes. Let us collaborate to drive innovation and efficiency in your chemical manufacturing operations.
