Scalable Tetrazole Synthesis Using Recyclable Fe3O4/ZnS Nanocatalysts for Commercial API Production
Introduction to Advanced Tetrazole Manufacturing Technologies
The pharmaceutical and agrochemical industries rely heavily on nitrogen-containing heterocycles, with the tetrazole ring standing out as a critical bioisostere for carboxylic acids in drug design. A pivotal advancement in this domain is documented in patent CN101857577B, which discloses a highly efficient method for preparing tetrazole compounds utilizing core-shell magnetic nanoparticles as a catalyst. This technology addresses the longstanding challenges of catalyst recovery and metal contamination that have plagued traditional synthetic routes. By employing Fe3O4/ZnS magnetic nanocatalysts, the process enables the synthesis of diverse 5-substituted-1H-tetrazoles from readily available benzonitriles and sodium azide. The general structural framework of these high-value intermediates is illustrated below, highlighting the versatility of the R-group substitution which is crucial for tuning biological activity in final API candidates.
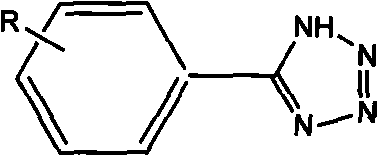
As a reliable pharmaceutical intermediate supplier, understanding the nuances of such catalytic systems is paramount for ensuring supply continuity. The innovation lies not just in the yield, but in the engineering of the catalyst itself; the core-shell architecture combines the magnetic separability of iron oxide with the catalytic activity of zinc sulfide. This dual functionality transforms a homogeneous-style reaction into a heterogeneous process that is far more amenable to large-scale industrial production. For R&D directors and procurement managers alike, this represents a shift towards greener, more cost-effective manufacturing paradigms that align with modern regulatory standards for residual metals in active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tetrazoles via [2+3] cycloaddition of nitriles and azides has relied heavily on Lewis acid catalysts or soluble transition metal salts. While methods developed by researchers like Sharpless utilizing zinc, iron, or copper salts demonstrated good catalytic activity, they suffered from significant downstream processing drawbacks. The primary issue is the difficulty in separating these homogeneous catalysts from the reaction mixture, often requiring extensive chromatography or complex extraction protocols to meet strict purity specifications. Furthermore, the use of stoichiometric amounts of certain reagents or expensive noble metals drives up the cost of goods sold (COGS) and generates substantial heavy metal waste, creating environmental compliance burdens for manufacturing facilities. These factors collectively hinder the scalability of traditional routes, making them less attractive for the commercial production of high-volume intermediates.
The Novel Approach
The novel approach described in the patent data overcomes these barriers by introducing a magnetically recoverable heterogeneous catalyst system. By utilizing core-shell Fe3O4/ZnS nanoparticles, the reaction maintains high catalytic efficiency while allowing for instantaneous separation of the catalyst simply by applying an external magnetic field. This eliminates the need for filtration or centrifugation steps that often result in product loss or catalyst degradation. The process operates under relatively mild conditions, typically around 120°C in DMF solvent, and demonstrates exceptional tolerance to various functional groups on the aromatic ring. This robustness ensures that sensitive substrates can be converted without decomposition, thereby expanding the accessible chemical space for medicinal chemists. The ability to reuse the catalyst for multiple cycles without significant loss of activity further amplifies the economic and environmental benefits of this methodology.
Mechanistic Insights into Fe3O4/ZnS Catalyzed Cycloaddition
The mechanistic pathway for this transformation involves the activation of the nitrile group by the Lewis acidic sites on the surface of the ZnS shell. The zinc species coordinate with the nitrogen atom of the nitrile, increasing the electrophilicity of the carbon atom and facilitating the nucleophilic attack by the azide ion. This initial interaction lowers the activation energy barrier for the [2+3] dipolar cycloaddition, which is the rate-determining step in tetrazole formation. The Fe3O4 core serves primarily as a magnetic handle, ensuring that the active ZnS sites remain accessible while providing the physical property necessary for rapid phase separation. The synergy between the magnetic core and the catalytic shell creates a stable nano-reactor environment that promotes high turnover frequencies. Understanding this mechanism is vital for process optimization, as it suggests that maintaining the integrity of the core-shell structure is key to long-term catalyst stability.
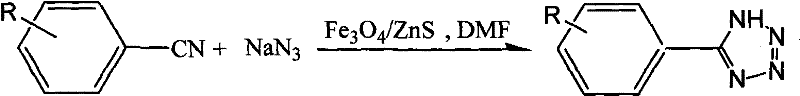
Regarding impurity control, the heterogeneous nature of the catalyst inherently minimizes the risk of metal leaching into the final product, a common concern with soluble metal salts. However, rigorous quality control is still necessary to ensure that no trace iron or zinc remains above acceptable thresholds for pharmaceutical applications. The reaction conditions, specifically the temperature range of 110-130°C and the use of DMF as a polar aprotic solvent, are optimized to maximize solubility of the ionic sodium azide while maintaining the stability of the organic nitrile substrates. Side reactions are minimal due to the specificity of the catalyst surface, leading to clean reaction profiles that simplify downstream purification. This level of control over the reaction trajectory is what distinguishes advanced nanocatalysis from traditional bulk chemical processes, offering a clearer path to GMP-compliant manufacturing.
How to Synthesize 5-Substituted-1H-Tetrazoles Efficiently
The synthesis protocol derived from the patent data outlines a straightforward procedure suitable for both laboratory scale-up and pilot plant operations. The process begins with the precise weighing of benzonitrile derivatives and sodium azide, typically in a molar ratio of 1:2 to 1:3 to drive the equilibrium towards completion. The core-shell magnetic catalyst is added at a loading of 20-30% relative to the total mass of reactants, suspended in dimethylformamide (DMF). The mixture is then heated to 120°C and stirred for approximately 24 hours to ensure full conversion. Following the reaction, the unique magnetic properties of the catalyst allow for its immediate retrieval, leaving a clear solution from which the product can be isolated via acidification and extraction. For detailed operational parameters and safety guidelines regarding azide handling, please refer to the standardized synthesis steps provided below.
- Combine benzonitrile or substituted benzonitrile with sodium azide (molar ratio 1: 2 to 1:3) and Fe3O4/ZnS catalyst (20-30% mass ratio) in DMF solvent.
- Heat the reaction mixture to 120°C under stirring for approximately 24 hours to ensure complete conversion to the tetrazole ring.
- Separate the magnetic catalyst using an external magnet, wash with ethyl acetate for reuse, and isolate the product via acidification and extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this magnetic nanocatalyst technology translates into tangible operational efficiencies and risk mitigation. The primary advantage lies in the drastic simplification of the workup procedure; the ability to remove the catalyst magnetically removes the need for expensive filtration equipment or disposable filter aids that can clog during large-scale processing. This streamlining of the unit operations directly correlates to reduced batch cycle times and lower labor costs per kilogram of product. Additionally, the reusability of the catalyst means that the effective cost of the catalytic material is amortized over many batches, significantly lowering the raw material cost contribution to the final price. This economic model supports more competitive pricing strategies for high-purity pharmaceutical intermediates in a global market.
- Cost Reduction in Manufacturing: The elimination of soluble heavy metal catalysts removes the necessity for costly scavenging resins or complex aqueous washes designed to reduce metal content to ppm levels. This reduction in downstream processing steps leads to substantial cost savings in terms of solvent consumption, waste disposal fees, and processing time. Furthermore, the high yields reported across various substrates, ranging from electron-deficient nitro compounds to electron-rich amino derivatives, ensure that raw material utilization is maximized. By minimizing the formation of byproducts and simplifying the purification train, the overall manufacturing cost structure is optimized, allowing for better margin protection even when raw material prices fluctuate.
- Enhanced Supply Chain Reliability: The robustness of the Fe3O4/ZnS catalyst system ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply to downstream API manufacturers. Since the catalyst can be reused multiple times without significant degradation, the supply chain is less vulnerable to disruptions in the sourcing of fresh catalytic materials. The use of commodity chemicals like benzonitriles and sodium azide as starting materials further secures the supply base, as these are widely available from multiple global vendors. This diversification of the raw material basket reduces dependency on single-source suppliers and mitigates the risk of shortages that could otherwise halt production lines.
- Scalability and Environmental Compliance: From an environmental perspective, the heterogeneous nature of the process aligns well with green chemistry principles by reducing waste generation and energy consumption. The magnetic separation technique is inherently scalable; magnetic separators are standard equipment in modern chemical plants, meaning the transition from gram-scale to ton-scale production does not require fundamental changes to the process physics. This ease of scale-up reduces the time and capital investment required for technology transfer. Moreover, the reduced heavy metal load in the effluent simplifies wastewater treatment requirements, ensuring compliance with increasingly stringent environmental regulations without the need for expensive tertiary treatment facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this tetrazole synthesis technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers cover aspects of catalyst lifecycle, substrate compatibility, and process safety, which are critical for a comprehensive risk assessment.
Q: Can the Fe3O4/ZnS catalyst be reused without loss of activity?
A: Yes, the core-shell magnetic structure allows for easy separation and the catalyst maintains high efficiency over at least 10 consecutive cycles with negligible yield drop.
Q: What is the substrate scope for this tetrazole synthesis method?
A: The method is highly versatile, accommodating various substituents including chloro, nitro, methyl, and amino groups on the benzene ring with yields ranging from 76% to 94%.
Q: How does this method improve supply chain reliability compared to traditional Lewis acid catalysis?
A: By eliminating the need for complex downstream purification to remove soluble heavy metal residues, this process significantly reduces production lead time and waste treatment costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrazole Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced catalytic technologies like the Fe3O4/ZnS system in securing a competitive edge in the fine chemicals market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of tetrazole intermediate meets the exacting standards required by global pharmaceutical clients. Our commitment to technical excellence allows us to offer customized solutions that balance performance with cost-efficiency.
We invite you to engage with our technical procurement team to discuss how this specific synthetic route can be adapted to your project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the potential economic benefits for your specific supply chain. We encourage you to reach out for specific COA data and route feasibility assessments to validate the suitability of this technology for your upcoming campaigns. Let us collaborate to optimize your intermediate sourcing strategy and drive value through scientific innovation.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
