Scalable Manufacturing of Compound A for CNS Drug Development via Optimized Sonogashira Coupling
The pharmaceutical industry's relentless pursuit of effective treatments for central nervous system (CNS) disorders has placed significant emphasis on the development of robust synthetic routes for complex heterocyclic compounds. Patent CN103857679A discloses a groundbreaking methodology for the preparation of 5-[2-[7-(trifluoromethyl)-5-[4-(trifluoromethyl)phenyl]pyrazolo[1,5-a]pyrimidin-3-yl]ethynyl]-2-pyridinamine, herein referred to as Compound A. This molecule functions as a potent mGluR2 antagonist, holding immense therapeutic potential for managing depression and other neurological conditions. The technical breakthrough presented in this intellectual property lies not merely in the molecular structure but in the engineering of a scalable, safe, and economically viable manufacturing process that overcomes the limitations of earlier synthetic attempts described in prior art such as WO2006/0999721. By leveraging advanced transition metal catalysis and innovative purification strategies, this process ensures the delivery of high-purity pharmaceutical intermediates essential for downstream drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex pyrazolo[1,5-a]pyrimidine derivatives has been plagued by inefficiencies that hinder commercial viability. Conventional routes often rely on multi-step sequences requiring the isolation of sensitive intermediates, which can lead to significant yield losses and increased operational costs. Previous methods described in the background art were deemed unsuitable for supporting clinical programs due to their inability to be scaled effectively in production facilities. These legacy processes frequently suffered from poor impurity profiles, particularly regarding residual heavy metals from catalytic steps, which pose severe regulatory hurdles for CNS active pharmaceutical ingredients. Furthermore, the reliance on harsh reaction conditions or unstable reagents often compromised the safety profile of the manufacturing environment, creating bottlenecks in the supply chain for critical drug candidates.
The Novel Approach
The novel approach detailed in the patent introduces a streamlined synthetic strategy that fundamentally reshapes the production landscape for Compound A. Central to this innovation is the utilization of optimized Sonogashira coupling reactions that allow for the efficient construction of the carbon-carbon triple bond linkage between the pyrimidine core and the pyridine moiety. As illustrated in the reaction schemes, the process offers flexibility by accommodating both protected and unprotected alkyne precursors, enabling chemists to select the most cost-effective pathway based on raw material availability. 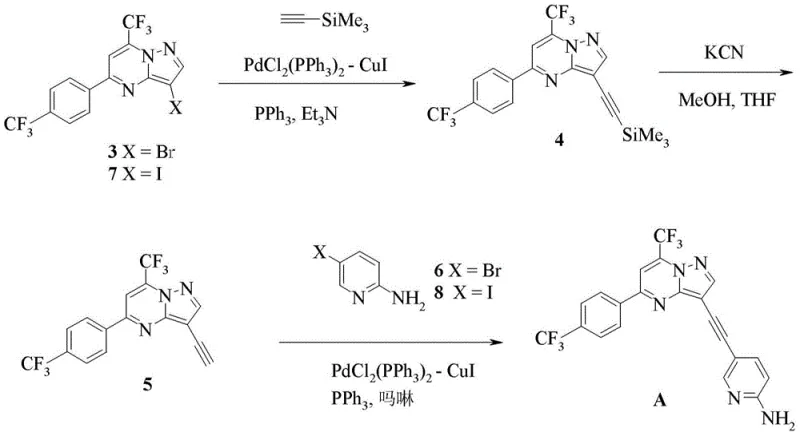 . Moreover, the patent describes a highly efficient one-pot synthesis for key intermediates, such as Compound 7, starting from readily available acetophenones.
. Moreover, the patent describes a highly efficient one-pot synthesis for key intermediates, such as Compound 7, starting from readily available acetophenones. 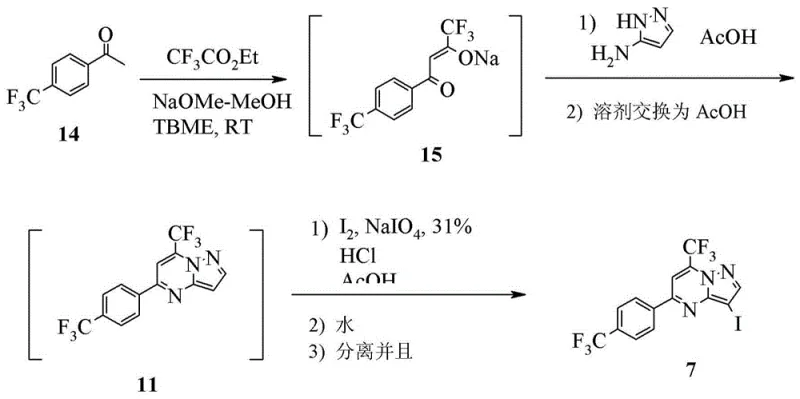 . This telescoping of reactions eliminates intermediate workups, drastically reducing solvent usage and processing time, which translates directly into substantial cost reduction in pharmaceutical intermediate manufacturing.
. This telescoping of reactions eliminates intermediate workups, drastically reducing solvent usage and processing time, which translates directly into substantial cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Pd-Cu Catalyzed Sonogashira Coupling
The core of this synthetic technology relies on the precise orchestration of palladium and copper catalysis to facilitate the cross-coupling reaction. The mechanism involves the oxidative addition of the aryl halide (Compound 3 or 7) to the zero-valent palladium species, forming an organopalladium intermediate. Simultaneously, the copper(I) co-catalyst activates the terminal alkyne by forming a copper acetylide species. The transmetallation step transfers the alkynyl group to the palladium center, followed by reductive elimination to forge the desired C-C bond and regenerate the active catalyst. The patent specifies the use of robust catalyst systems such as PdCl2(PPh3)2 combined with CuI, which have been proven to maintain high activity even under the demanding conditions required for large-scale batches. The choice of base, typically triethylamine or diisopropylamine, is critical for neutralizing the hydrogen halide byproduct and maintaining the catalytic cycle's integrity throughout the reaction duration.
Beyond the coupling chemistry, the patent places extraordinary emphasis on the purification mechanism to ensure product quality. A major challenge in transition metal-catalyzed reactions is the removal of trace palladium and copper, which are toxic and strictly regulated in final drug substances. The disclosed method employs a sophisticated scavenging strategy using tri-n-butylphosphine, which coordinates strongly with residual metal ions to form soluble complexes that can be separated from the product. Following metal scavenging, the process utilizes a salt formation step with methanesulfonic acid to precipitate the compound as a mesylate salt. 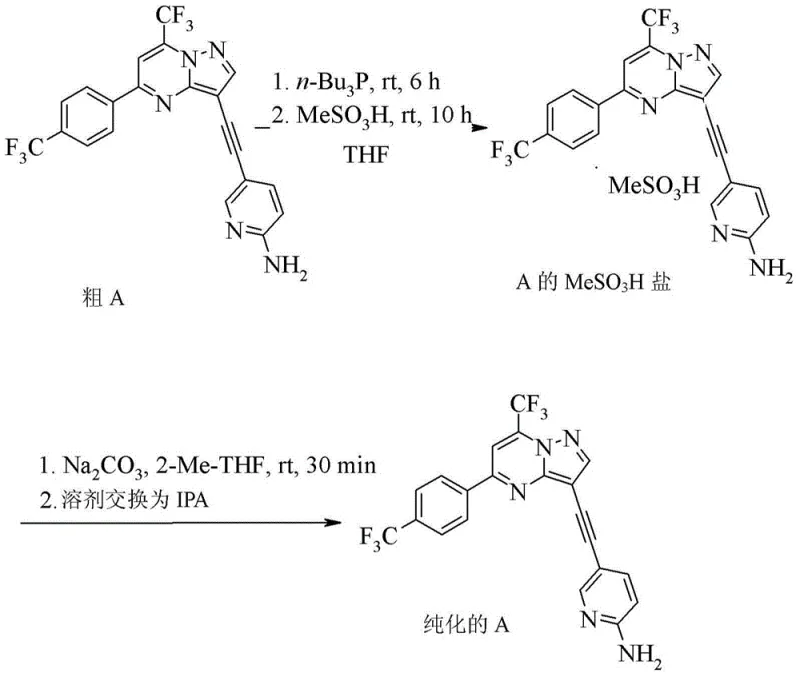 . This salt formation acts as a powerful purification tool, excluding organic impurities that do not form salts. The final conversion to the free base and crystallization from isopropanol yields a product with exceptional purity, addressing the stringent杂质谱 requirements demanded by R&D directors for CNS drug candidates.
. This salt formation acts as a powerful purification tool, excluding organic impurities that do not form salts. The final conversion to the free base and crystallization from isopropanol yields a product with exceptional purity, addressing the stringent杂质谱 requirements demanded by R&D directors for CNS drug candidates.
How to Synthesize Compound A Efficiently
The synthesis of Compound A is designed to be operationally simple yet chemically rigorous, making it ideal for transfer from laboratory bench to pilot plant and eventually to commercial manufacturing. The process begins with the preparation of the halogenated pyrazolo[1,5-a]pyrimidine core, which can be achieved via a condensation reaction followed by halogenation, or through the preferred one-pot method that minimizes handling. The subsequent coupling with the ethynyl pyridine fragment requires careful control of temperature and stoichiometry to maximize yield while minimizing side reactions such as homocoupling of the alkyne. The detailed standardized synthesis steps below outline the critical parameters for achieving consistent results, including specific solvent exchanges and crystallization protocols that are vital for controlling particle size and polymorphic form.
- Perform a Sonogashira coupling reaction between a halogenated pyrazolo[1,5-a]pyrimidine precursor (Compound 3 or 7) and a protected or unprotected ethynyl pyridine derivative in an inert solvent using a palladium and copper catalyst system.
- If a protected alkyne is used, conduct a desilylation step using fluoride sources or alkaline conditions to generate the terminal alkyne intermediate prior to the final coupling.
- Purify the crude product by treating with tri-n-butylphosphine to scavenge metal catalysts, followed by salt formation with methanesulfonic acid and subsequent recrystallization from isopropanol to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the technical advancements in this patent translate into tangible business value and risk mitigation. The primary advantage lies in the significant simplification of the manufacturing workflow, which directly correlates to reduced production costs and enhanced supply reliability. By eliminating multiple isolation steps and utilizing a one-pot approach for key intermediates, the process reduces the consumption of expensive solvents and minimizes waste generation, aligning with modern green chemistry principles and environmental compliance standards. This efficiency gain allows for a more competitive pricing structure without compromising on the quality of the high-purity pharmaceutical intermediates supplied to downstream partners.
- Cost Reduction in Manufacturing: The streamlined synthetic route significantly lowers the cost of goods sold by reducing the number of unit operations and the associated labor and utility costs. The ability to perform telescoped reactions means less equipment occupancy time and lower capital expenditure requirements for production facilities. Furthermore, the use of commercially available starting materials and standard catalysts avoids the need for bespoke, expensive reagents, ensuring that cost reduction in pharmaceutical intermediate manufacturing is sustainable over the long term.
- Enhanced Supply Chain Reliability: The robustness of the described chemistry ensures consistent batch-to-batch quality, which is crucial for maintaining uninterrupted supply chains for clinical trials and commercial launches. The process is designed to be scalable from kilogram to multi-ton quantities, providing the flexibility needed to respond to fluctuating market demands. By securing a reliable pharmaceutical intermediate supplier who utilizes this patented technology, companies can mitigate the risk of production delays caused by complex purification bottlenecks or low-yielding steps common in older methodologies.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability, having been validated for use in production facilities with standard reactor configurations. The reduction in solvent usage and the efficient recovery of materials contribute to a lower environmental footprint, facilitating easier regulatory approval and compliance with increasingly strict environmental regulations. The purification method effectively removes heavy metals, ensuring that the final product meets global safety standards without the need for additional, costly remediation steps, thus supporting the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Compound A. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on the feasibility and advantages of this manufacturing route. Understanding these details is essential for stakeholders evaluating the integration of this intermediate into their drug development pipelines.
Q: How does this process address heavy metal contamination in pharmaceutical intermediates?
A: The patented method employs a specific purification protocol using tri-n-butylphosphine to effectively scavenge palladium and copper residues, followed by a mesylate salt precipitation step that ensures the final API intermediate meets stringent purity specifications required for CNS drug development.
Q: What are the scalability advantages of the one-pot synthesis described in the patent?
A: The one-pot methodology eliminates the isolation of unstable or difficult-to-handle intermediates, significantly reducing solvent consumption and processing time. This streamlined approach enhances operational efficiency and reduces the overall environmental footprint of the manufacturing process.
Q: Is this synthetic route suitable for commercial-scale production of mGluR2 antagonists?
A: Yes, the patent explicitly details conditions optimized for large-scale production facilities, utilizing robust reagents and standard industrial solvents like THF, DMF, and isopropanol, ensuring reliable supply chain continuity for clinical and commercial programs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Compound A Supplier
As the demand for advanced CNS therapeutics continues to grow, partnering with a manufacturer who possesses deep technical expertise in complex heterocyclic synthesis is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this field, leveraging the innovations described in patent CN103857679A to deliver superior quality intermediates. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of any project phase. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of Compound A meets the highest standards of quality and consistency required by global regulatory bodies.
We invite you to collaborate with us to optimize your supply chain and accelerate your drug development timelines. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how our optimized processes can reduce your overall manufacturing expenses. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for high-purity pharmaceutical intermediates and commercial scale-up of complex pharmaceutical intermediates.
