Optimizing Lorlatinib Production: A High-Efficiency TBTU-Mediated Cyclization Strategy
The global demand for third-generation ALK inhibitors continues to surge, driven by the critical need for effective treatments in non-small cell lung cancer (NSCLC). At the forefront of this therapeutic landscape is Lorlatinib, a potent molecule whose complex macrocyclic structure presents significant synthetic challenges for manufacturers. Patent CN112457293A introduces a transformative approach to the final cyclization step, addressing long-standing inefficiencies in yield and reagent consumption that have plagued previous production methods. This technical disclosure outlines a robust protocol utilizing O-benzotriazole-N,N,N',N'-tetramethyluronium tetrafluoroborate (TBTU) in conjunction with 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU), delivering a commercially viable pathway that aligns with the rigorous standards of modern pharmaceutical supply chains.
For R&D directors and process chemists, the implications of this patent extend beyond simple yield improvements; it represents a fundamental optimization of the amide bond formation kinetics essential for closing the macrocyclic ring. By shifting away from expensive and often inefficient coupling agents like HATU or COMU, this method offers a streamlined route that minimizes waste generation and simplifies post-reaction workup. As a reliable pharmaceutical intermediates supplier, understanding these mechanistic nuances is crucial for scaling production from laboratory benchtop to multi-ton commercial manufacturing without compromising on the stringent purity specifications required for oncology drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing Lorlatinib have historically struggled with poor atom economy and inconsistent reaction outcomes, creating bottlenecks in the supply chain. Traditional protocols often rely on HATU (Hexafluorophosphate Azabenzotriazole Tetramethyl Uronium) paired with DIPEA as a base, which, while effective for some couplings, proves suboptimal for this specific macrocyclization. Data indicates that low-temperature variations of this method result in abysmal yields of merely 29%, forcing manufacturers to either accept massive material losses or resort to heating the reaction to 60-70°C. Even under heated conditions, yields plateau around 78%, leaving over 20% of valuable starting material lost to degradation or oligomerization side reactions.
Furthermore, alternative approaches utilizing COMU as a condensing agent with triethylamine (TEA) have demonstrated yields lower than 60%, accompanied by significant difficulties in purification. The excessive use of condensing agents and bases in these legacy processes not only inflates the raw material costs but also complicates the downstream isolation of the API. The presence of residual urea byproducts and unreacted coupling reagents necessitates extensive chromatographic purification or multiple recrystallization steps, which drastically increases the processing time and solvent consumption. For a procurement manager focused on cost reduction in pharmaceutical intermediates manufacturing, these inefficiencies translate directly into higher unit costs and longer lead times, making the adoption of more efficient catalytic systems an economic imperative.
The Novel Approach
The innovative strategy detailed in CN112457293A overcomes these hurdles by employing a specific combination of TBTU and DBU, which synergistically enhances the cyclization efficiency. This novel approach operates effectively at moderate temperatures ranging from 40°C to 60°C, striking an optimal balance between reaction kinetics and thermal stability of the sensitive intermediates. Experimental data from the patent reveals that this method consistently achieves yields exceeding 87%, with optimized examples reaching up to 89.4% and purity levels surpassing 98%. This represents a substantial leap forward compared to the comparative examples, where yields hovered between 58% and 77% under similar conditions using different reagent combinations.
Beyond the impressive yield metrics, the new method significantly reduces the molar equivalents of reagents required. While older methods often demanded large excesses of coupling agents to drive the reaction to completion, the TBTU/DBU system functions efficiently with near-stoichiometric ratios, typically around 1:0.5-1.5 for TBTU and 0.1-1 for DBU relative to the substrate. This reduction in reagent loading not only lowers the direct material costs but also simplifies the quenching and extraction phases. The ability to use standard solvents like DMF or toluene, followed by a straightforward ammonium chloride quench and ethyl acetate extraction, streamlines the entire workflow. For supply chain stakeholders, this translates to a more predictable and scalable process capable of supporting the commercial scale-up of complex pharmaceutical intermediates with greater reliability.
Mechanistic Insights into TBTU-Mediated Macrocyclization
The success of this synthesis hinges on the precise activation of the carboxylic acid moiety within the precursor molecule, known as Compound A. In this mechanism, TBTU acts as a highly efficient uronium-type coupling reagent that reacts with the carboxyl group to form an activated O-acylisourea intermediate. This activated species is significantly more electrophilic than the parent acid, facilitating the subsequent nucleophilic attack by the proximal amine group to close the macrocyclic ring. The choice of DBU as the base is critical; unlike weaker amines such as triethylamine, DBU is a strong, non-nucleophilic base that effectively deprotonates the amine nucleophile without competing in the coupling reaction itself. This ensures that the activation energy barrier for cyclization is lowered while minimizing the formation of N-acylurea byproducts that often plague urea-based coupling chemistries.
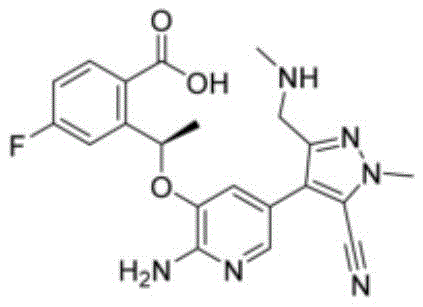
Furthermore, the steric and electronic properties of the TBTU reagent contribute to the high stereochemical integrity of the final product. The tetramethyluronium structure provides sufficient bulk to prevent racemization at the chiral center adjacent to the carbonyl, a common issue in peptide and macrocycle synthesis. The reaction proceeds through a concerted pathway where the base facilitates the rapid collapse of the tetrahedral intermediate, releasing the tetramethylurea byproduct and forming the stable amide bond of the Lorlatinib core. Understanding this mechanistic pathway allows process engineers to fine-tune parameters such as addition rates and temperature ramps to further suppress trace impurities. The result is a crude product with exceptional purity profiles, often exceeding 97-98% before any major purification, which validates the robustness of this catalytic system for high-purity pharmaceutical intermediates.
Impurity control is another vital aspect where this mechanism excels. In conventional HATU-mediated reactions, the formation of guanidine-type byproducts and epimerization can lead to difficult-to-remove impurities that require aggressive purification strategies. The TBTU/DBU system mitigates these risks by maintaining a cleaner reaction profile throughout the 2 to 3-hour reaction window. The rapid consumption of the activated intermediate prevents prolonged exposure to conditions that might promote hydrolysis or rearrangement. Consequently, the impurity spectrum is simplified, consisting mainly of unreacted starting material which is easily removed during the aqueous workup. This level of control is essential for meeting the rigorous regulatory standards imposed on oncology APIs, ensuring that the final drug substance is free from genotoxic or mutagenic contaminants.
How to Synthesize Lorlatinib Efficiently
The implementation of this TBTU-mediated cyclization requires careful attention to solvent selection and stoichiometry to replicate the high yields reported in the patent literature. The process begins with the dissolution of the open-chain precursor, Compound A, in a polar aprotic solvent, with DMF being the preferred medium due to its ability to solubilize both the organic substrate and the ionic coupling reagents. Once the solution is homogenized, the precise addition of TBTU and DBU initiates the activation sequence. The reaction mixture is then gently heated to the target range of 40-60°C, where it is maintained under agitation to ensure uniform heat transfer and mass transport. Monitoring the reaction progress via TLC or HPLC is recommended to determine the exact endpoint, typically occurring within 2 to 3 hours, after which the system is cooled and quenched.
- Dissolve Compound A in an organic solvent such as DMF and add the condensing agent TBTU along with the alkali promoter DBU.
- Heat the reaction mixture to a temperature range of 40-60°C and maintain stirring for 2 to 3 hours to ensure complete cyclization.
- Quench the reaction with ammonium chloride solution, extract the product with ethyl acetate, and purify via concentration and washing to obtain high-purity Lorlatinib.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this optimized synthetic route offers compelling economic and operational benefits that extend far beyond the laboratory. The primary advantage lies in the drastic reduction of raw material costs associated with the coupling agents. By replacing expensive reagents like HATU with the more cost-effective TBTU, and by reducing the required molar equivalents of the base, the overall bill of materials for the final step is significantly lowered. Additionally, the higher reaction yield means that less starting material is wasted, effectively increasing the throughput of the manufacturing facility without the need for additional capital investment in reactors or equipment. This efficiency gain is critical for maintaining competitive pricing in the global market for API intermediates.
- Cost Reduction in Manufacturing: The elimination of excessive reagent usage and the avoidance of low-yield pathways directly translate to substantial cost savings per kilogram of produced API. Since the process generates fewer byproducts and utilizes cheaper reagents, the expenditure on raw materials is optimized. Furthermore, the simplified workup procedure reduces the consumption of solvents and energy required for distillation and purification, contributing to a leaner manufacturing cost structure. These cumulative savings allow for more aggressive pricing strategies while maintaining healthy profit margins, a key factor for long-term commercial viability.
- Enhanced Supply Chain Reliability: The robustness of the TBTU/DBU chemistry ensures consistent batch-to-batch performance, which is vital for securing long-term supply contracts with major pharmaceutical companies. The use of readily available and stable reagents minimizes the risk of supply disruptions caused by the scarcity of specialized catalysts. Moreover, the shorter reaction times and streamlined isolation steps enable faster turnover of production batches, effectively reducing lead time for high-purity pharmaceutical intermediates. This agility allows suppliers to respond more rapidly to fluctuations in market demand, ensuring a steady flow of material to downstream formulation partners.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this method aligns well with green chemistry principles by reducing the E-factor (mass of waste per mass of product). The lower quantity of chemical waste generated simplifies effluent treatment and reduces the environmental footprint of the manufacturing site. The process is inherently scalable, having been demonstrated to work efficiently in standard organic solvents without requiring exotic conditions or cryogenic temperatures. This ease of scale-up facilitates the transition from pilot plant trials to full-scale commercial production, ensuring that supply commitments can be met reliably as the drug moves through various stages of clinical and commercial lifecycle.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method compares to established industry standards. Understanding these details is essential for technical teams evaluating the feasibility of adopting this process for their own manufacturing lines or for procurement specialists assessing the quality and reliability of potential suppliers.
Q: Why is the TBTU/DBU system superior to HATU/DIPEA for Lorlatinib synthesis?
A: The TBTU/DBU system described in patent CN112457293A achieves significantly higher yields (up to 89.4%) compared to traditional HATU methods (which range from 29% to 78%), while operating under milder conditions and requiring fewer equivalents of reagents.
Q: What are the critical reaction parameters for this cyclization?
A: Critical parameters include maintaining the reaction temperature between 40-60°C, using DMF as the preferred solvent, and ensuring a molar ratio of Compound A to TBTU to DBU of approximately 1:1:1 to maximize conversion efficiency.
Q: How does this method impact downstream purification costs?
A: By achieving crude purities exceeding 97% and minimizing side products through the use of specific base promoters like DBU, this method drastically reduces the burden on chromatographic purification, leading to substantial overall process cost savings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lorlatinib Supplier
As the pharmaceutical industry continues to evolve, the ability to deliver complex molecules like Lorlatinib with high efficiency and purity is paramount. NINGBO INNO PHARMCHEM stands ready to leverage this advanced TBTU-mediated technology to support your production needs. With our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we possess the technical expertise and infrastructure necessary to bring this optimized process to life. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, ensuring that every batch of intermediate meets the exacting standards required for oncology drug development.
We invite you to collaborate with us to explore the full potential of this synthetic route for your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your Lorlatinib manufacturing operations.
