Scalable Enantioselective Synthesis of Non-Steroidal Antiestrogen Intermediates for Commercial Production
The pharmaceutical industry continuously seeks robust manufacturing pathways for complex active pharmaceutical ingredients, particularly those requiring high stereochemical fidelity. Patent CN1322200A presents a groundbreaking advancement in the enantioselective synthesis of non-steroidal antiestrogen compounds, specifically targeting the efficient production of Formula I and Formula XIV intermediates. This intellectual property outlines a short, practical commercial process that overcomes the significant limitations of previous laboratory-scale methods, which often relied on costly and inefficient purification techniques. By integrating novel reaction conditions and catalytic systems, this technology enables the production of chemically and enantiomerically pure intermediates suitable for large-scale industrial application. The strategic design of this synthetic route addresses critical pain points in modern drug manufacturing, including impurity control, solvent safety, and overall process economics. For stakeholders evaluating potential partnerships for high-purity Pharmaceutical Intermediates, understanding the technical nuances of this patent provides a clear competitive advantage in supply chain planning.
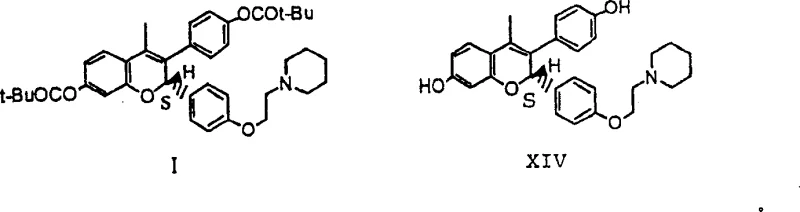
The transition from discovery chemistry to commercial manufacturing often reveals hidden bottlenecks that can jeopardize project timelines and budgets. Conventional methods for synthesizing these specific chromene-based antiestrogen antagonists typically involve multiple steps with low overall yields and rely heavily on chromatographic purification to achieve necessary purity levels. These traditional approaches frequently utilize hazardous solvents like dichloromethane and require expensive chiral stationary phases for separation, which are impractical for multi-kilogram or ton-scale production. Furthermore, prior art methods often struggle with the control of geometric isomers, leading to complex mixtures that are difficult to separate and result in significant material loss. The reliance on fractional crystallization without effective racemization loops means that unwanted enantiomers are discarded as waste, drastically increasing the cost of goods sold. For a reliable Pharmaceutical Intermediates supplier, adopting such inefficient processes would compromise the ability to offer competitive pricing and consistent supply continuity to global partners.
In stark contrast, the novel approach detailed in CN1322200A introduces a series of optimized reaction conditions that fundamentally shift the thermodynamic equilibrium towards the desired products. A key innovation lies in the use of sterically hindered organic amine bases, such as DBU or DBN, during the cyclization steps. This specific choice of reagents promotes the isomerization of unwanted cis-isomers and phenyl styryl ketones into the desired trans-chromanone structure with high selectivity. By driving the reaction equilibrium through crystallization-induced precipitation, the process eliminates the need for chromatographic separation at this critical stage. Additionally, the method employs a dynamic kinetic resolution strategy that allows for the recycling of unwanted diastereomers back into the productive cycle. This holistic view of the synthesis pathway ensures that cost reduction in Pharmaceutical Intermediates manufacturing is achieved not just through cheaper reagents, but through a fundamental redesign of the material flow to minimize waste and maximize yield.
Mechanistic Insights into DBU-Catalyzed Isomerization and Dynamic Resolution
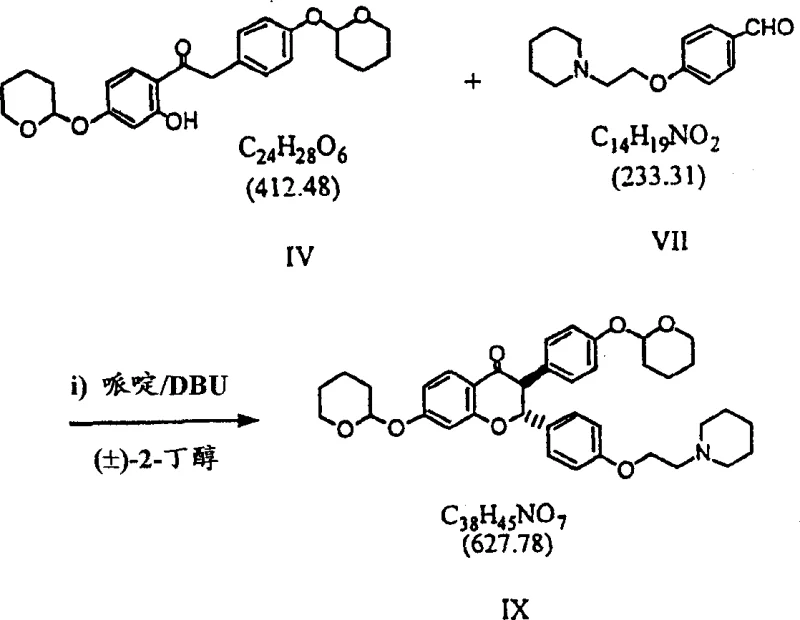
The core chemical innovation of this process resides in the precise control of stereochemistry during the formation of the chromene ring system. The reaction begins with a Knoevenagel condensation between a protected ketone and an aldehyde, followed by a base-catalyzed cyclization. The use of 2-butanol as a solvent in conjunction with DBU creates a unique environment where the equilibrium between the open-chain phenyl styryl ketone (Formula VIII) and the cyclic chromanone (Formula IX) is heavily favored towards the latter. This is crucial because the cyclic form is the necessary precursor for subsequent methylation and resolution steps. The mechanism involves the deprotonation of the acidic alpha-proton, allowing for bond rotation and re-closure into the thermodynamically more stable trans-configuration. This step effectively purifies the intermediate through crystallization, ensuring that downstream reactions proceed with minimal interference from geometric isomers. Such mechanistic precision is vital for R&D teams focusing on the commercial scale-up of complex Pharmaceutical Intermediates.
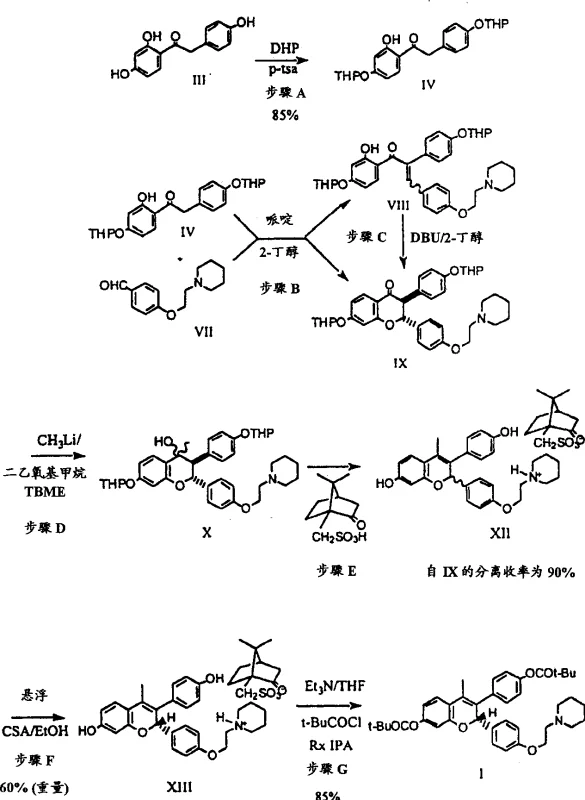
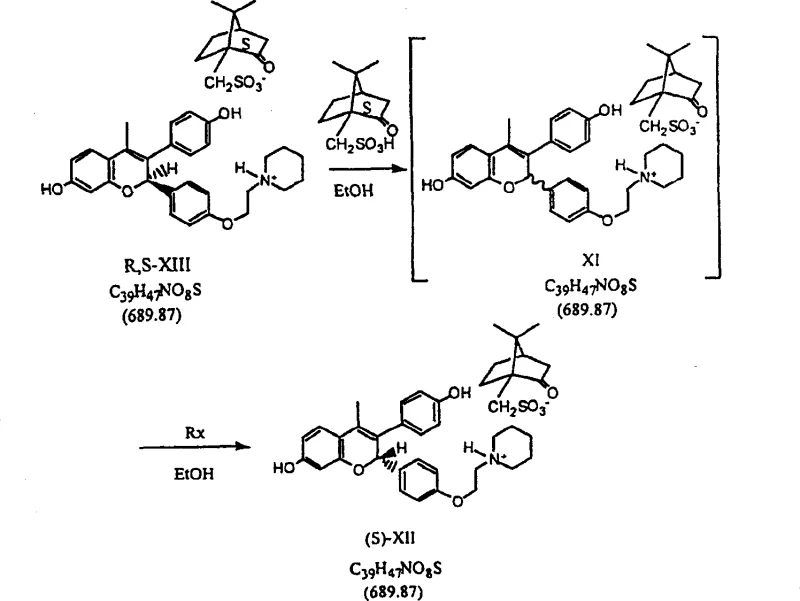
Following the cyclization, the process employs a highly selective methylation step using methyllithium in specific ether solvents like t-butyl methyl ether (TBME). This choice is critical to suppress the formation of Michael addition byproducts (Formula Q), which are common when using Grignard reagents or THF solvents. The subsequent steps involve a tandem dehydration and deprotection sequence that converts the intermediate directly into a racemic camphorsulfonic acid salt. The true brilliance of the patent, however, is manifested in the dynamic kinetic resolution step. By heating the mixture of diastereomeric salts in the presence of a catalytic amount of chiral acid, the unwanted isomer racemizes in solution while the desired isomer crystallizes out. This continuous conversion ensures that the theoretical yield can exceed 50%, traditionally the limit for classical resolutions. This level of impurity control and yield optimization demonstrates a deep understanding of physical organic chemistry applied to industrial problems.
How to Synthesize Antiestrogen Intermediate Efficiently
Implementing this synthesis requires strict adherence to the specified reaction parameters to ensure reproducibility and safety on a large scale. The process begins with the protection of phenolic hydroxyl groups followed by the condensation and cyclization sequence described previously. Operators must maintain precise temperature controls during the methylation step to prevent exothermic runaways and ensure selectivity. The resolution step requires careful monitoring of the diastereomeric ratio via HPLC to determine the optimal endpoint for crystallization. While the general chemistry is robust, the specific workup procedures, such as the avoidance of aqueous washes that might emulsify or hydrolyze sensitive intermediates, are critical for success. Detailed standard operating procedures regarding solvent swaps, drying temperatures, and filtration rates are essential to translate this patent data into a validated manufacturing process. For comprehensive technical guidance, the detailed standardized synthesis steps are outlined in the guide below.
- Perform Knoevenagel condensation of protected ketone and aldehyde using piperidine and DBU in 2-butanol to form trans-chromanone.
- Execute methylation using methyllithium in t-butyl methyl ether to minimize byproduct formation.
- Conduct dynamic kinetic resolution using (S)-camphorsulfonic acid to isolate the desired S,S-diastereomer salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the technological advancements encoded in CN1322200A translate directly into tangible business benefits that extend beyond simple chemical yield. The elimination of chromatographic purification steps represents a massive reduction in both consumable costs and processing time. Chromatography is notoriously difficult to scale, requiring large volumes of silica gel and solvents, along with significant downtime for column packing and regeneration. By replacing this unit operation with crystallization, the process becomes inherently more scalable and predictable. This shift allows for the use of standard stainless steel reactors and filtration equipment, which are readily available in most multipurpose chemical plants. Consequently, this leads to substantial cost savings in capital expenditure and operational overhead, making the final intermediate more price-competitive in the global market.
- Cost Reduction in Manufacturing: The process logic dictates significant economic advantages through the minimization of waste and the optimization of raw material usage. By employing a dynamic kinetic resolution, the method theoretically allows for the conversion of all starting material into the desired enantiomer, rather than discarding half as waste. Furthermore, the avoidance of expensive chiral chromatography columns and the reduction in solvent consumption due to streamlined workups contribute to a lower cost base. The use of common, commercially available reagents like DBU and camphorsulfonic acid ensures that raw material costs remain stable and predictable. These factors combined create a manufacturing profile that supports aggressive pricing strategies without compromising margin integrity.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex processes that have multiple failure points or rely on niche reagents. This synthetic route enhances reliability by utilizing robust chemistry that tolerates minor variations in scale without losing performance. The ability to recycle mother liquors containing unwanted isomers back into the process acts as a buffer against raw material fluctuations, ensuring that overall yield remains high even if individual batch efficiencies vary slightly. Additionally, the reduction in hazardous solvent usage simplifies logistics and storage requirements, reducing the risk of supply disruptions related to regulatory changes or transportation restrictions. This stability is crucial for partners seeking reducing lead time for high-purity Pharmaceutical Intermediates.
- Scalability and Environmental Compliance: Modern manufacturing mandates strict adherence to environmental, health, and safety (EHS) standards. This process aligns perfectly with green chemistry principles by eliminating chlorinated solvents like dichloromethane from the critical resolution and acylation steps. The replacement with alcoholic solvents and ethers reduces the environmental footprint and simplifies waste treatment protocols. The high purity achieved through crystallization reduces the need for energy-intensive distillation or reprocessing. As regulatory pressures increase globally, having a process that is inherently cleaner and safer provides a strategic advantage, ensuring long-term viability and compliance with evolving international standards for Pharmaceutical Intermediates production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and claims within CN1322200A, providing a factual basis for decision-making. Understanding these specifics helps stakeholders evaluate the feasibility of integrating this technology into their existing supply chains. The focus is on clarity regarding purity profiles, scalability limits, and the specific advantages over legacy methods. This transparency is intended to facilitate smoother technical transfers and more informed procurement discussions between suppliers and multinational corporations.
Q: How does this process improve optical purity compared to prior art?
A: The process utilizes a dynamic kinetic resolution step with (S)-camphorsulfonic acid, which allows for the racemization of the unwanted diastereomer in situ, driving the equilibrium towards the desired S,S-diastereomer and achieving over 98% de without chromatography.
Q: What solvents are avoided in this commercial scale-up method?
A: The method specifically avoids the use of dichloromethane (DCM) and dimethylformamide (DMF) during the resolution and acylation steps, replacing them with safer alcoholic solvents and t-butyl methyl ether to enhance environmental compliance and operator safety.
Q: Can the unwanted isomers be recycled to improve yield?
A: Yes, the mother liquor containing the unwanted R,S-diastereomer can be subjected to acid-catalyzed racemization conditions, converting it back into a mixture that can be re-subjected to resolution, thereby significantly reducing material waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Antiestrogen Intermediate Supplier
The technical potential of CN1322200A represents a significant opportunity for optimizing the production of next-generation antiestrogen therapies. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this sophisticated chemistry to life. Our facility is equipped with state-of-the-art reactors capable of handling the specific temperature and inert atmosphere conditions demanded by the methylation and resolution steps. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch meets the exacting standards required for pharmaceutical applications. Our team of process chemists is adept at translating patent literature into validated, GMP-compliant manufacturing processes, ensuring that the theoretical benefits of this route are fully realized in commercial output.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this route for your projects. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments tailored to your volume requirements. Let us demonstrate how our expertise in enantioselective synthesis can drive efficiency and reliability in your pharmaceutical development pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
