Advanced Synthesis of Lasofoxifene Intermediate: A Scalable Commercial Route
Advanced Synthesis of Lasofoxifene Intermediate: A Scalable Commercial Route
The pharmaceutical landscape for osteoporosis treatment continues to evolve, driven by the demand for safer and more cost-effective manufacturing processes for key active ingredients. Patent CN102311406A introduces a groundbreaking methodology for the preparation of a critical Lasofoxifene intermediate, specifically 1-[2-[4-(6-methoxyl group-2-phenyl-3,4-dihydronaphthalene-1-yl) phenoxy] ethyl] tetramethyleneimine, also known as Nafoxidene or Compound IV. This molecule serves as a pivotal precursor in the synthesis of Lasofoxifene, a selective estrogen receptor modulator (SERM). The disclosed technology represents a significant departure from traditional synthetic pathways, offering a robust alternative that prioritizes operator safety, environmental compliance, and economic efficiency. By leveraging a streamlined three-step sequence involving Grignard addition, bromination, and Suzuki-Miyaura coupling, this innovation addresses the longstanding bottlenecks associated with cryogenic conditions and hazardous reagents. For global supply chain leaders, this patent signals a viable pathway toward reducing lead time for high-purity pharmaceutical intermediates while maintaining rigorous quality standards essential for regulatory approval.
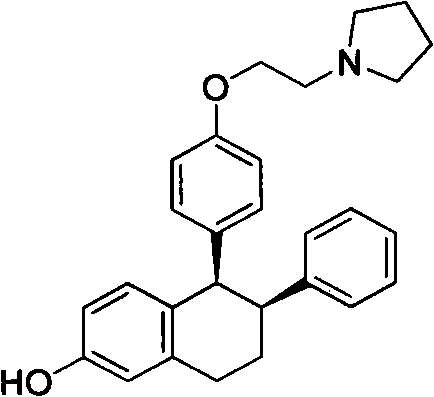
Understanding the structural complexity of the target molecule is essential for appreciating the synthetic challenges involved. The core architecture requires the precise assembly of a tetrahydronaphthalene system linked to a phenoxy-ethyl-pyrrolidine side chain. Achieving this connectivity with high regioselectivity and stereochemical integrity has historically been a formidable task for process chemists. The new method described in the patent simplifies this construction by utilizing readily available starting materials such as 6-methoxytetralone and brominated phenoxy derivatives. This strategic disconnection not only shortens the overall synthetic route but also enhances the atom economy of the process. As a reliable pharmaceutical intermediate supplier, recognizing the value of such process intensification is crucial for maintaining competitiveness in the generic drug market. The ability to produce Compound IV or its inorganic acid salts with improved yield and reduced impurity profiles directly translates to lower cost of goods sold (COGS) for the final API manufacturer.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
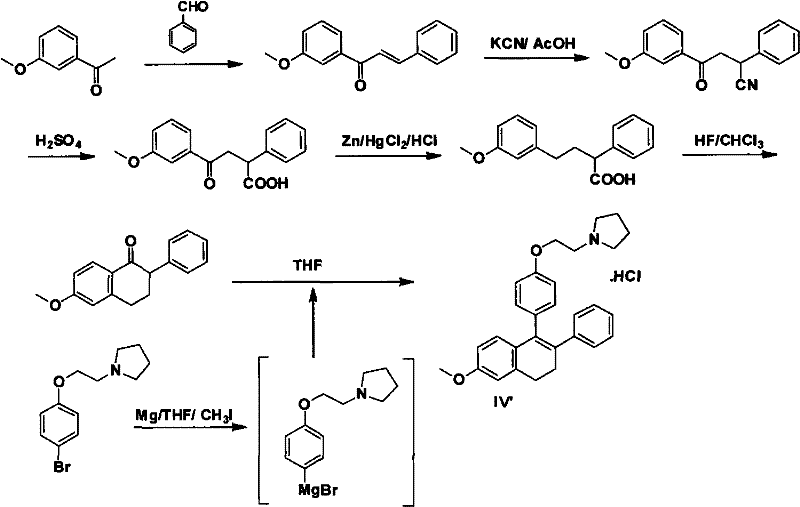
Prior to this innovation, the industry relied on synthetic routes that were fraught with significant operational hazards and economic inefficiencies. One prominent method, disclosed in U.S. Patent 3274213B1, necessitates the use of potassium cyanide, a highly toxic reagent that imposes severe safety protocols and waste disposal burdens on manufacturing facilities. Furthermore, this legacy route employs hydrogen fluoride, a severely corrosive substance that demands specialized equipment resistant to fluoridation, thereby inflating capital expenditure (CapEx). The chemical efficiency of this older pathway is also suboptimal, with the final Grignard reaction step yielding merely 24%, resulting in substantial material loss and increased production costs. Another approach, detailed in European Patent EP 0802910A1, attempts to mitigate some toxicity issues but introduces new challenges related to cost and scalability.
The European method relies heavily on anhydrous cerous compounds, which are not only expensive but also required in large stoichiometric quantities, driving up raw material costs significantly. Moreover, this process mandates reaction conditions at -78°C, requiring energy-intensive cryogenic cooling systems that are difficult to maintain on a large industrial scale. Such ultra-low temperature operations limit batch sizes and increase the risk of thermal runaway if cooling fails, posing a threat to supply chain continuity. The cumulative effect of these factors—high reagent costs, specialized equipment needs, and low overall yields—renders these conventional methods unsuitable for modern, cost-sensitive pharmaceutical manufacturing. For procurement managers, these legacy routes represent a supply chain vulnerability, where reliance on hazardous chemicals can lead to regulatory delays and unexpected production stoppages.
The Novel Approach
In stark contrast, the methodology presented in CN102311406A offers a streamlined and industrially friendly alternative that circumvents the pitfalls of prior art. The new route initiates with the formation of a Grignard reagent from Compound I using magnesium metal in tetrahydrofuran (THF), followed by nucleophilic addition to 6-methoxytetralone to generate Compound II. This step proceeds under mild conditions, either at normal temperature or under reflux, eliminating the need for cryogenic infrastructure. Subsequently, Compound II undergoes a regioselective bromination using pyridinium tribromide or elemental bromine to afford Compound III. The use of pyridinium tribromide is particularly advantageous as it is a solid reagent that is easier to handle and dose accurately compared to liquid bromine, enhancing process safety and reproducibility.
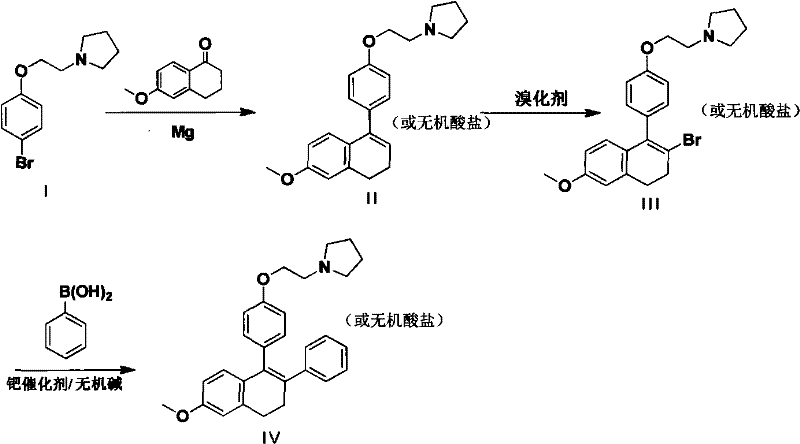
The final transformation involves a palladium-catalyzed Suzuki-Miyaura cross-coupling reaction between the bromo-intermediate Compound III and phenylboronic acid. This powerful carbon-carbon bond-forming reaction is conducted in the presence of a mineral alkali, such as sodium carbonate, and a palladium catalyst like tetrakis(triphenylphosphine)palladium. This step efficiently installs the phenyl ring at the desired position on the naphthalene core, completing the skeleton of the Lasofoxifene intermediate. The entire sequence is designed to be telescoped or performed with minimal purification between steps, as the intermediate salts can often be carried forward directly. This modular approach allows for significant flexibility in manufacturing, enabling producers to adapt quickly to market demands. By replacing toxic cyanides and corrosive fluorides with standard organometallic coupling chemistry, this novel approach aligns perfectly with the principles of green chemistry and sustainable manufacturing.
Mechanistic Insights into Grignard Addition and Suzuki Coupling
The success of this synthetic strategy hinges on the precise control of organometallic reactivity and transition metal catalysis. In the initial step, the generation of the Grignard reagent from the bromo-phenoxy precursor is facilitated by the activation of magnesium metal, often initiated by iodine or 1,2-dibromoethane. The presence of the pyrrolidine ether side chain requires careful optimization to prevent side reactions such as Wurtz coupling or deprotonation of the amine, although the ether linkage provides sufficient stability under the chosen conditions. The subsequent addition to 6-methoxytetralone is a classic nucleophilic attack on the carbonyl carbon, forming a tertiary alcohol intermediate which likely undergoes dehydration in situ or during workup to establish the dihydronaphthalene double bond. The inventors noted that the yield of this step exceeded expectations, suggesting that the specific solvent system and stoichiometry (1.0-1.5:1 molar ratio of ketone to Grignard) effectively suppress competing pathways.
Impurity control is further refined in the bromination and coupling stages. The bromination of the dihydronaphthalene system must occur selectively at the 2-position to enable the subsequent cross-coupling. The use of pyridinium tribromide allows for a controlled release of bromine, minimizing polybromination or oxidative degradation of the electron-rich aromatic rings. In the final Suzuki coupling, the choice of palladium catalyst is critical; tetrakis(triphenylphosphine)palladium is preferred for its high activity in coupling sterically hindered substrates. The reaction mechanism involves the oxidative addition of the aryl bromide to the Pd(0) species, transmetallation with the phenylboronate complex formed in the basic aqueous phase, and reductive elimination to release the coupled product. This catalytic cycle is highly efficient, and the use of a biphasic system (THF/water with carbonate base) ensures that inorganic byproducts are easily separated during the aqueous workup, leading to a high-purity final product suitable for downstream API synthesis.
How to Synthesize Nafoxidene Efficiently
The synthesis of Nafoxidene (Compound IV) via this patented route offers a clear blueprint for process chemists aiming to optimize production workflows. The protocol begins with the preparation of the Grignard reagent in THF, followed by the addition of 6-methoxytetralone to form the core naphthalene structure. Following isolation or direct processing, the intermediate is brominated to activate the coupling site, and finally, the phenyl group is introduced via palladium catalysis. Each step has been optimized for yield and ease of purification, with the final product often isolated as a stable hydrochloride salt to ensure long-term storage stability. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures, are outlined below to guide technical teams in replicating this high-efficiency process.
- Convert Compound I into a Grignard reagent using Magnesium metal in THF, then react with 6-methoxytetralone to form Compound II.
- Perform a regioselective bromination on Compound II using pyridinium tribromide to yield the bromo-intermediate Compound III.
- Execute a palladium-catalyzed Suzuki coupling between Compound III and phenylboronic acid to finalize the Lasofoxifene intermediate Compound IV.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of hazardous reagents like potassium cyanide and hydrogen fluoride removes the need for specialized containment systems and expensive waste treatment protocols, leading to substantial cost savings in both operational expenditure and regulatory compliance. Furthermore, the shift away from cryogenic conditions (-78°C) to ambient or reflux temperatures drastically reduces energy consumption and allows for the use of standard glass-lined or stainless steel reactors, lowering the barrier to entry for contract manufacturing organizations (CMOs). This process intensification means that production capacity can be scaled up rapidly without the need for significant capital investment in new infrastructure, ensuring a more resilient and responsive supply chain for critical osteoporosis medications.
- Cost Reduction in Manufacturing: The replacement of expensive cerium chloride and toxic cyanides with commodity chemicals like magnesium, bromine sources, and phenylboronic acid significantly lowers the raw material cost profile. Additionally, the higher overall yield of the new route compared to the 24% yield of legacy methods means less starting material is required per kilogram of final product, directly improving the margin structure. The simplified purification steps, often relying on acid-base extractions rather than complex chromatography, further reduce solvent usage and processing time, contributing to a leaner manufacturing cost base.
- Enhanced Supply Chain Reliability: By utilizing widely available reagents and avoiding supply-constrained specialty catalysts or dangerous gases, the risk of production delays due to raw material shortages is minimized. The robustness of the Suzuki coupling reaction, which tolerates a variety of functional groups and conditions, ensures consistent batch-to-batch quality, reducing the likelihood of failed batches that disrupt inventory levels. This reliability is crucial for maintaining continuous supply to API manufacturers who operate on tight just-in-time schedules, thereby strengthening the partnership between intermediate suppliers and downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory bench scale to multi-ton commercial production without the engineering challenges associated with heat transfer in cryogenic reactions. From an environmental perspective, the reduction in toxic waste streams aligns with increasingly stringent global environmental regulations, reducing the liability and disposal costs associated with hazardous waste. This 'green' credential not only improves the corporate sustainability profile but also facilitates faster regulatory approvals in markets with strict environmental oversight, accelerating time-to-market for the final drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Lasofoxifene intermediates using this patented methodology. These insights are derived directly from the experimental data and process descriptions provided in the patent literature, offering a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about sourcing strategies and process validation requirements.
Q: What are the key advantages of this new synthesis route over prior art?
A: This method eliminates the use of highly toxic potassium cyanide and corrosive hydrogen fluoride found in older routes. It also avoids the expensive cerium chloride and ultra-low temperature (-78°C) conditions required by European patents, significantly reducing operational costs and safety risks.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates under normal temperature or reflux conditions rather than cryogenic temperatures. The use of stable reagents like pyridinium tribromide and standard palladium catalysts makes it highly adaptable for multi-kilogram to ton-scale manufacturing.
Q: How is product purity controlled in the final coupling step?
A: Purity is managed through the precise stoichiometric control of phenylboronic acid (1.0-2.0:1 molar ratio) and the use of efficient extraction and acidification protocols. The formation of the inorganic acid salt (hydrochloride) further facilitates purification via crystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lasofoxifene Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development and commercialization of life-saving medications. Our team of expert process chemists has extensively evaluated the route disclosed in CN102311406A and possesses the technical expertise to implement this synthesis at scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Lasofoxifene intermediate meets the highest industry standards for impurity profiles and physicochemical properties.
We invite you to collaborate with us to optimize your supply chain for osteoporosis therapeutics. By leveraging our manufacturing capabilities, you can achieve significant efficiencies and secure a stable source of this key building block. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our production of Nafoxidene can enhance your project's economics and timeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
