Optimizing Chloroquine Phosphate Production: A Technical Breakthrough in High-Purity API Manufacturing
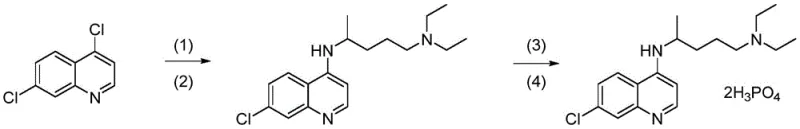
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with economic viability, particularly for essential antimalarial agents. Patent CN112300071A introduces a transformative synthetic method for high-purity chloroquine phosphate, addressing critical limitations found in traditional manufacturing processes. This innovation leverages a controlled micro-pressurization environment combined with a dual-catalyst system comprising phenol and dialkylamines to drive the condensation of 4,7-dichloroquinoline and 2-amino-5-diethylaminopentane. By shifting away from the harsh thermal conditions of prior art, this methodology not only enhances atom utilization but also drastically reduces the formation of difficult-to-remove impurities. For global supply chain stakeholders, this represents a pivotal shift towards more sustainable and reliable API intermediate production, ensuring consistent quality without compromising on throughput or environmental compliance standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chloroquine phosphate has been plagued by inefficient reaction conditions that necessitate excessive energy input and result in suboptimal yields. Previous techniques, such as those disclosed in earlier patents, often rely on atmospheric pressure conditions which force operators to elevate reaction temperatures significantly to achieve acceptable conversion rates. This thermal stress frequently triggers unwanted side reactions, specifically the decomposition of the diethylamino side chain, leading to the formation of deethylchloroquine impurities that are notoriously difficult to separate. Furthermore, the lack of effective catalytic systems in these older methods results in poor atom economy, where a significant portion of valuable starting materials is wasted or converted into solid waste streams. These inefficiencies create bottlenecks in large-scale manufacturing, driving up production costs and complicating the purification workflow required to meet regulatory purity specifications for pharmaceutical-grade ingredients.
The Novel Approach
The novel approach detailed in the patent data fundamentally reengineers the reaction landscape by introducing a micro-pressurized system operating between 1.4 to 1.7 atm. This slight increase in pressure allows the condensation reaction to proceed vigorously at a much milder temperature range of 70 to 100°C, effectively suppressing the thermal degradation pathways that compromise product integrity. The strategic inclusion of phenol and dialkylamines acts as a synergistic catalytic system, accelerating the nucleophilic substitution without requiring extreme thermal energy. This methodological shift ensures that the reaction kinetics favor the formation of the desired chloroquine base while minimizing byproduct generation. Consequently, the downstream purification burden is significantly lightened, allowing for a more streamlined process flow that is inherently safer and more economically attractive for industrial-scale implementation compared to legacy atmospheric protocols.
Mechanistic Insights into Phenol-Assisted Micro-Pressurized Condensation
The core chemical innovation lies in the precise manipulation of reaction thermodynamics through pressure control and catalytic assistance. In this system, phenol serves not merely as a solvent but as a proton donor that facilitates the activation of the quinoline ring, making the chlorine atom at the 4-position more susceptible to nucleophilic attack by the diamine side chain. Simultaneously, the dialkylamine, preferably diethylamine, stabilizes the transition state and helps scavenge acidic byproducts, maintaining an optimal pH environment within the reaction matrix. The application of 1.4 to 1.7 atm pressure increases the collision frequency of reactant molecules without raising the kinetic energy to levels that would break the C-N bonds in the side chain. This delicate balance ensures that the activation energy barrier for the main reaction is lowered while the barrier for decomposition remains high, resulting in a highly selective transformation that preserves the structural integrity of the complex amine moiety throughout the synthesis.
Impurity control is further enhanced by the specific sequence of workup and purification steps designed to isolate the product from unreacted starting materials and catalysts. Following the condensation, the reaction mixture is treated with alkali metal carbonate or hydroxide solutions to adjust the pH to 8-9, which ensures that the basic chloroquine product remains in the organic phase while acidic impurities and salts are partitioned into the aqueous layer. The subsequent recrystallization of the crude chloroquine using non-polar solvents like petroleum ether exploits the solubility differences between the target molecule and lipophilic impurities. This intermediate purification step is critical, as it prevents the carryover of side products into the final salification stage, thereby guaranteeing that the final chloroquine phosphate crystal lattice is formed from high-purity precursors, ultimately yielding a product with HPLC purity exceeding 98 percent.
How to Synthesize Chloroquine Phosphate Efficiently
Implementing this synthesis route requires strict adherence to the defined parameters regarding pressure, temperature, and stoichiometry to maximize yield and purity. The process begins with the precise mixing of 4,7-dichloroquinoline and phenol, followed by the controlled addition of the amine reactants under the specified micro-pressure conditions. Operators must monitor the temperature closely to maintain the 70 to 100°C window, ensuring that the exothermic nature of the reaction does not lead to thermal runaway. Following the reaction, the separation of the organic phase and the subsequent recrystallization steps must be performed with high-purity solvents to avoid introducing new contaminants. The detailed standardized synthesis steps, including specific workup procedures and solvent recovery protocols, are outlined in the technical guide below for engineering teams to review and adapt for their specific facility capabilities.
- Condense 4,7-dichloroquinoline with 2-amino-5-diethylaminopentane using phenol and dialkylamine catalysts at 70-100°C under 1.4-1.7 atm pressure.
- Refine the crude chloroquine intermediate via recrystallization in non-polar solvents like petroleum ether to remove impurities.
- React purified chloroquine with phosphoric acid in ethanol, followed by final recrystallization to obtain high-purity chloroquine phosphate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers profound advantages that directly address the pain points of procurement managers and supply chain directors seeking stability and cost efficiency. The ability to operate at lower temperatures translates directly into reduced energy consumption for heating and cooling systems, lowering the overall utility costs associated with batch production. Moreover, the design of the process explicitly incorporates the recovery and recycling of key reagents such as phenol and dialkylamines from the aqueous waste streams. This circular approach to material usage significantly diminishes the volume of raw materials required per kilogram of finished product, insulating the manufacturing process from volatility in raw material pricing markets. By minimizing waste generation and maximizing resource utilization, the process aligns with modern green chemistry principles, reducing the environmental footprint and potential regulatory liabilities associated with hazardous waste disposal.
- Cost Reduction in Manufacturing: The elimination of high-temperature requirements and the implementation of catalyst recycling mechanisms lead to substantial operational expenditure savings. By recovering valuable amines and phenols that would otherwise be lost in the aqueous phase, the net consumption of expensive reagents is drastically reduced. This efficiency gain allows for a more competitive cost structure without sacrificing product quality, providing a buffer against market fluctuations in chemical feedstock prices. Additionally, the simplified purification train reduces the need for extensive chromatography or complex distillation setups, further lowering capital and maintenance costs for the production facility.
- Enhanced Supply Chain Reliability: The reliance on commercially available and easily sourced raw materials ensures that production schedules are not disrupted by niche supply shortages. The robustness of the reaction conditions, which tolerate slight variations without catastrophic failure, enhances the predictability of batch outcomes. This consistency is crucial for maintaining continuous supply lines to downstream pharmaceutical formulators. The ability to scale this process from laboratory benchmarks to multi-ton production runs without significant re-engineering provides supply chain heads with the confidence to commit to long-term contracts, knowing that the manufacturing partner can reliably meet demand surges.
- Scalability and Environmental Compliance: The process generates significantly less solid waste compared to traditional methods, simplifying the effluent treatment requirements and reducing the burden on wastewater treatment plants. The use of recyclable solvents and the minimization of hazardous byproducts facilitate easier compliance with increasingly stringent environmental regulations. This environmental stewardship not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the supply chain. The straightforward scale-up potential means that production capacity can be expanded rapidly to meet global health needs, ensuring that essential medications remain accessible without compromising on ecological standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this high-purity synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial manufacturing. The answers reflect the specific advantages in terms of purity control, yield optimization, and material recovery that distinguish this method from conventional alternatives.
Q: How does the micro-pressurization technique improve yield compared to atmospheric methods?
A: Operating at 1.4 to 1.7 atm allows the reaction to proceed efficiently at lower temperatures (70-100°C), significantly inhibiting the thermal decomposition of the side chain that typically generates deethylchloroquine impurities.
Q: What is the achieved purity level of the final Chloroquine Phosphate product?
A: Through a dual-recrystallization strategy involving both the free base and the phosphate salt, the process consistently achieves HPLC purity levels exceeding 98%, meeting stringent pharmacopeial standards.
Q: Can the catalysts and solvents be recovered to reduce production costs?
A: Yes, the process design includes specific recovery steps for phenol, dialkylamine, and excess amine reactants from the aqueous and mother liquor phases, enabling material circulation and substantial cost optimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chloroquine Phosphate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced synthetic technologies to meet the rigorous demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the one described in CN112300071A can be successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch of Chloroquine Phosphate meets the highest international standards. Our commitment to technical excellence ensures that we can deliver high-purity API intermediates that support the efficacy and safety of final drug products, providing our partners with a secure and high-quality supply foundation.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized manufacturing capabilities, we can help you reduce total landed costs while securing a stable supply of critical materials. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us collaborate to enhance your supply chain resilience and drive value through superior chemical manufacturing solutions, ensuring that your production timelines are met with precision and reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
