Advanced Optical Resolution of Piperidine Intermediates for Commercial API Production
Advanced Optical Resolution of Piperidine Intermediates for Commercial API Production
The pharmaceutical industry continuously demands higher purity standards for chiral intermediates, particularly those serving as critical building blocks for complex therapeutic agents. Patent CN101952252A introduces a robust and industrially viable method for the optical resolution of alkyl piperidin-3-ylcarbamates, addressing the longstanding challenge of obtaining high optical purity without resorting to prohibitively expensive chromatographic techniques. This technology leverages the formation of diastereomeric salts using optically active tartaric acid, a strategy that significantly enhances the feasibility of large-scale manufacturing for diabetes treatment drug intermediates. By utilizing this specific resolution pathway, manufacturers can achieve optical purities exceeding 90% ee, ensuring that the final active pharmaceutical ingredients meet the stringent regulatory requirements for stereochemical integrity. The process is designed to be adaptable to various alkyl groups, providing flexibility in synthesizing a range of piperidine derivatives essential for modern medicinal chemistry pipelines.
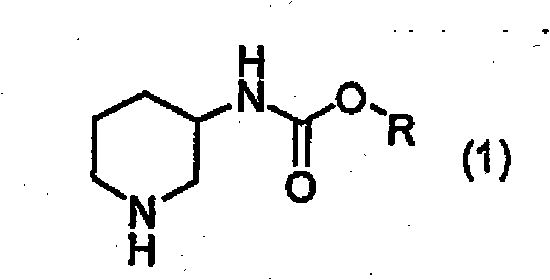
Furthermore, the methodology outlined in this patent provides a clear pathway for converting these resolved carbamates into optically active 3-aminopiperidines, which are versatile scaffolds in drug discovery. The ability to control the stereochemistry at the 3-position of the piperidine ring is crucial for the biological activity of the resulting compounds, as enantiomers often exhibit vastly different pharmacological profiles. This resolution technique not only improves the quality of the intermediate but also streamlines the downstream synthesis processes by reducing the burden of impurity removal in later stages. For a reliable pharmaceutical intermediate supplier, adopting such a proven resolution strategy means offering clients a more consistent and high-quality product that reduces their overall development risk. The integration of this technology into existing production lines represents a significant step forward in the manufacturing of high-purity piperidine derivatives, aligning with the industry's shift towards more efficient and sustainable chiral synthesis methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for obtaining optically active 3-aminopiperidine often rely on direct resolution of the amine itself, which can present significant technical hurdles in an industrial setting. One major drawback of conventional approaches is the difficulty in achieving satisfactory optical purity on a large scale, as the physical properties of the enantiomers of the free amine can be too similar to allow for efficient separation via crystallization. Additionally, many older methods require the use of expensive chiral resolving agents that are not easily recovered or recycled, leading to increased material costs and waste generation. The handling of free amines can also be problematic due to their potential instability or reactivity under certain conditions, which may lead to racemization or the formation of unwanted by-products during the purification process. These limitations often result in lower overall yields and higher production costs, making it challenging to maintain a competitive edge in the market for cost reduction in API manufacturing. Consequently, manufacturers are frequently forced to rely on multi-step synthetic routes or preparative chiral HPLC, both of which are resource-intensive and difficult to scale effectively for commercial supply.
The Novel Approach
In contrast, the novel approach described in the patent utilizes a protected intermediate, specifically the alkyl piperidin-3-ylcarbamate, which exhibits more favorable physical properties for resolution. By introducing the carbamate protecting group prior to the resolution step, the method enhances the crystallinity and stability of the intermediate, facilitating the formation of well-defined diastereomeric salts with tartaric acid. This strategic modification allows for a much more efficient separation of enantiomers through selective crystallization, significantly improving the optical purity of the isolated product. The use of tartaric acid, a naturally occurring and cost-effective chiral pool material, further enhances the economic viability of the process by reducing the reliance on synthetic chiral auxiliaries. Moreover, the conditions required for this resolution are mild and compatible with standard chemical processing equipment, enabling the commercial scale-up of complex chiral intermediates without the need for specialized infrastructure. This approach effectively bypasses the bottlenecks associated with direct amine resolution, offering a streamlined pathway that maximizes yield and purity while minimizing operational complexity and environmental impact.
Mechanistic Insights into Tartaric Acid-Mediated Diastereomeric Salt Formation
The core mechanism driving this resolution process is the formation of diastereomeric salts between the racemic alkyl piperidin-3-ylcarbamate and optically active tartaric acid. When the RS mixture of the carbamate interacts with a single enantiomer of tartaric acid, such as L-tartaric acid, two distinct diastereomeric salts are formed in situ. These salts possess different physical properties, most notably solubility, due to the distinct spatial arrangements of the molecules in the crystal lattice. By carefully controlling the solvent composition and temperature, one diastereomer can be selectively crystallized out of the solution while the other remains dissolved. For instance, using L-tartaric acid preferentially precipitates the salt containing the R-enantiomer of the carbamate, allowing for its physical separation via filtration. This thermodynamic control over the crystallization process is critical for achieving high enantiomeric excess, as it leverages the inherent energy differences between the diastereomeric pairs to drive the purification.
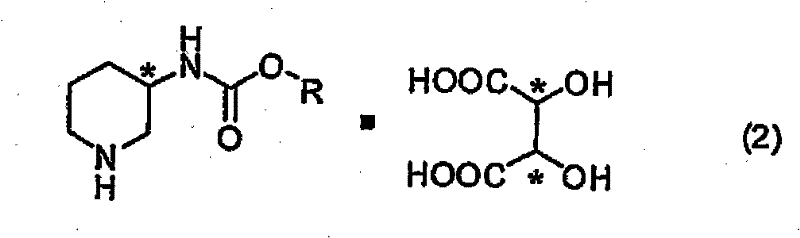
Following the isolation of the diastereomeric salt, the optically active carbamate is regenerated through a simple acid-base treatment. This step involves breaking the ionic bond between the carbamate and the tartaric acid, typically by adding a base such as sodium carbonate or an acid stronger than tartaric acid. The choice of reagent and conditions during this liberation step is crucial to prevent racemization, ensuring that the high optical purity achieved during crystallization is maintained in the final product. The patent specifies that treating the salt at temperatures below 60°C favors the recovery of the carbamate, while higher temperatures can lead to deprotection and the formation of 3-aminopiperidine directly. This mechanistic flexibility allows manufacturers to tailor the process output based on their specific downstream requirements, whether they need the protected intermediate for further functionalization or the free amine for final coupling reactions. The ability to recycle the tartaric acid from the mother liquor further enhances the sustainability of the process, aligning with green chemistry principles.
How to Synthesize Alkyl Piperidin-3-ylcarbamate Efficiently
Implementing this synthesis route requires precise control over reaction parameters to ensure consistent quality and yield. The process begins with the preparation of the racemic carbamate, followed by the critical resolution step where solvent selection plays a pivotal role. Detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios, temperature profiles, and workup procedures necessary to replicate the high purity results reported in the patent. Adhering to these protocols is essential for maintaining the integrity of the chiral center and maximizing the efficiency of the crystallization process.
- Prepare a solution of the racemic alkyl piperidin-3-ylcarbamate RS mixture in a suitable alcohol solvent such as methanol or ethanol.
- Add optically active tartaric acid, preferably L-tartaric acid for R-type isolation, to the solution to form diastereomeric salts.
- Crystallize the diastereomeric salt by cooling, filter the solid, and treat with acid or base to recover the optically active carbamate or 3-aminopiperidine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this resolution technology offers substantial strategic benefits that extend beyond mere technical performance. The process is designed to utilize readily available raw materials, such as tartaric acid and common alcohol solvents, which mitigates the risk of supply chain disruptions associated with specialized or scarce reagents. This reliance on commodity chemicals ensures a more stable and predictable sourcing environment, reducing the vulnerability of the production schedule to market fluctuations. Furthermore, the simplicity of the unit operations involved, primarily crystallization and filtration, allows for easier technology transfer and faster ramp-up times at manufacturing sites. This operational efficiency translates into a more responsive supply chain capable of meeting tight deadlines and adapting to changing demand volumes without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of expensive chiral chromatography steps and the use of recyclable resolving agents lead to significant cost savings in the overall production budget. By avoiding the need for high-consumption stationary phases and specialized equipment, the process reduces both capital expenditure and operating costs. The high yield of the resolution step minimizes material waste, ensuring that a larger proportion of the starting material is converted into valuable product. Additionally, the ability to recover and reuse tartaric acid from the process streams further lowers the effective cost of goods sold, making the final intermediate more competitive in the global market. These economic advantages are critical for maintaining profitability in the face of increasing pressure for cost reduction in API manufacturing.
- Enhanced Supply Chain Reliability: The robustness of this chemical process contributes to a more reliable supply of high-purity intermediates. Since the method does not depend on fragile biocatalysts or sensitive reagents that require strict cold-chain logistics, the storage and transportation of materials are simplified. The scalability of the crystallization process means that production volumes can be increased seamlessly to accommodate large orders, ensuring continuity of supply for key customers. This reliability is essential for pharmaceutical companies that need to secure long-term sources of critical intermediates to support their clinical and commercial programs. Reducing lead time for high-purity pharmaceutical intermediates becomes achievable when the manufacturing process is stable and predictable.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard reactor and filtration equipment that is common in fine chemical manufacturing facilities. This compatibility simplifies the transition from pilot scale to full commercial production, reducing the time and investment required for process validation. From an environmental perspective, the use of alcohol solvents and the potential for solvent recovery systems align with modern sustainability goals. The reduction in waste generation, particularly through the recycling of the resolving agent, minimizes the environmental footprint of the manufacturing operation. This compliance with environmental regulations not only avoids potential fines but also enhances the corporate social responsibility profile of the supply chain, which is increasingly important for stakeholders and partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this optical resolution technology. These answers are derived directly from the patent data to provide accurate and actionable insights for decision-makers evaluating this synthesis route for their supply chains.
Q: What is the primary advantage of using tartaric acid for piperidine resolution?
A: Using optically active tartaric acid allows for the formation of diastereomeric salts with significantly different solubility properties, enabling the isolation of high optical purity intermediates (90% ee or higher) without requiring expensive chiral chromatography.
Q: Which solvents are most effective for this crystallization process?
A: Alcohol solvents are preferred, specifically methanol, ethanol, or mixed solvents like methanol and 1-butanol. These solvents optimize the solubility balance required for selective crystallization of the target diastereomer.
Q: Can this method be scaled for industrial API production?
A: Yes, the process utilizes common reagents and standard crystallization unit operations, making it highly suitable for commercial scale-up of complex chiral intermediates with consistent quality and reduced operational complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkyl Piperidin-3-ylcarbamate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality chiral intermediates in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that verify every batch against the highest industry standards. Our expertise in implementing the tartaric acid resolution method allows us to offer Alkyl Piperidin-3-ylcarbamate with consistent optical purity, providing a solid foundation for your downstream synthesis.
We invite you to collaborate with us to optimize your supply chain and reduce your overall development costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your commercial goals. Let us be your partner in bringing high-quality pharmaceutical intermediates to market efficiently.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
