Advanced Palladium-Catalyzed Synthesis and Optical Resolution of Piperidin-3-ylcarbamate Intermediates
The pharmaceutical industry constantly seeks robust, scalable, and cost-effective pathways for synthesizing complex heterocyclic intermediates, particularly those serving as core structures for therapeutic agents. Patent CN101977899A presents a significant technological advancement in the manufacturing of piperidin-3-ylcarbamate compounds, which are vital precursors for various bioactive molecules, including potential diabetes therapeutics. This intellectual property outlines a dual-innovation approach: first, replacing expensive noble metal catalysts with a more economical palladium system for the hydrogenation of pyridine rings, and second, establishing a highly efficient optical resolution protocol using chiral organic acids. For R&D directors and procurement specialists, this patent represents a pivotal shift towards sustainable manufacturing practices that do not compromise on the stringent purity requirements demanded by modern regulatory bodies. The detailed methodology provided ensures that the transition from pyridine-3-ylcarbamate to the saturated piperidine analog is not only chemically feasible but also industrially viable, addressing long-standing issues regarding catalyst cost and stereochemical control.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of piperidin-3-ylcarbamate compounds relied heavily on the use of rhodium-based catalysts for the hydrogenation of the corresponding pyridine derivatives. While rhodium catalysts are effective, they present substantial economic and supply chain vulnerabilities due to their extreme cost and scarcity in the global market. Furthermore, conventional methods often struggled with achieving consistent high yields without rigorous purification steps, and the optical resolution of the resulting racemic mixtures was frequently inefficient, leading to suboptimal optical purity that required additional, costly recrystallization cycles. These legacy processes imposed a heavy burden on manufacturing budgets and extended lead times, making the final API intermediates less competitive in a price-sensitive market. The reliance on such precious metals also introduced complexity in waste management and catalyst recovery, complicating the environmental compliance profile of the manufacturing site.
The Novel Approach
The methodology disclosed in CN101977899A fundamentally disrupts this status quo by demonstrating that palladium catalysts, specifically palladium on carbon (Pd/C), can effectively replace rhodium when the reaction environment is precisely engineered. The key breakthrough lies in the addition of carboxylic acids or phosphoric acid to the reaction mixture, which adjusts the aqueous layer pH to a range of 1 to 7, preferably between 2 and 6. This acidic environment dramatically enhances the reaction rate and yield, allowing for the conversion of pyridine-3-ylcarbamate to piperidin-3-ylcarbamate with exceptional efficiency. Additionally, the patent introduces a refined optical resolution strategy using optically active mandelic acid or tartaric acid, which facilitates the formation of diastereomeric salts that can be easily crystallized to achieve high enantiomeric excess. This novel approach not only slashes raw material costs but also simplifies the downstream processing workflow.
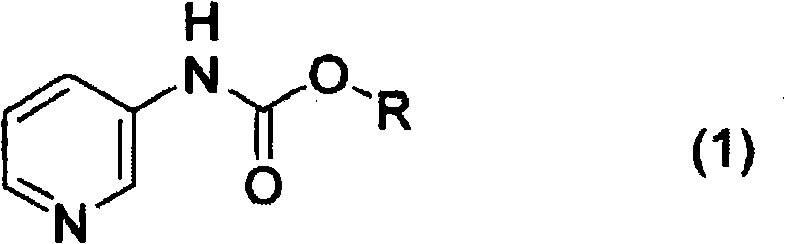
Mechanistic Insights into Pd-Catalyzed Hydrogenation and Acidic Control
The core chemical transformation involves the catalytic hydrogenation of the aromatic pyridine ring to a saturated piperidine ring, a reaction that is thermodynamically favorable but kinetically challenging without the correct catalyst and conditions. In this specific protocol, the palladium catalyst serves as the active site for hydrogen activation, but its efficacy is profoundly influenced by the protonation state of the substrate. By introducing acids such as acetic acid, the nitrogen atom of the pyridine ring becomes protonated, which alters the electronic density of the ring and potentially prevents catalyst poisoning by the basic amine functionality. This protonation is critical; without it, the basic nitrogen can coordinate strongly to the palladium surface, deactivating the catalyst and stalling the reaction. The patent data indicates that maintaining the pH within the specific window of 1 to 7 ensures that the substrate remains sufficiently activated for reduction while keeping the catalyst surface accessible for hydrogen transfer. This mechanistic understanding allows for the use of lower catalyst loadings and milder hydrogen pressures, typically around 0.1 to 5 MPa, further enhancing the safety and economic profile of the process.
Following the hydrogenation, the control of stereochemistry becomes the primary focus for ensuring the biological activity of the final drug substance. The optical resolution mechanism relies on the principle of diastereomeric salt formation, where the racemic piperidin-3-ylcarbamate reacts with a single enantiomer of a chiral acid, such as R-mandelic acid. Because the resulting salts are diastereomers, they possess different physical properties, most notably solubility in specific solvent systems. The patent elucidates that the choice of solvent is paramount; for instance, using ethanol or mixed solvents like methanol and 1-butanol creates a solubility differential that favors the crystallization of one diastereomer over the other. This selective crystallization effectively separates the desired enantiomer from the racemic mixture. The process is robust enough to handle various alkyl groups on the carbamate moiety, including ethyl, propyl, and tert-butyl groups, demonstrating the versatility of the resolution method across a range of structurally related intermediates.
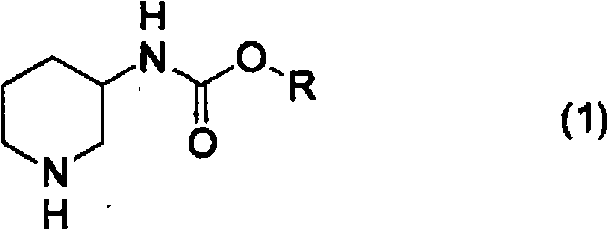
How to Synthesize Piperidin-3-ylcarbamate Efficiently
The synthesis pathway described in the patent offers a clear, step-by-step guide for producing high-quality piperidin-3-ylcarbamate intermediates suitable for pharmaceutical applications. The process begins with the protection of 3-aminopyridine via carbamation, followed by the critical palladium-catalyzed hydrogenation step under acidic conditions, and concludes with the optical resolution using chiral acids. Each stage has been optimized to maximize yield and purity while minimizing the use of hazardous reagents. The following sections detail the operational parameters derived directly from the experimental examples provided in the intellectual property documentation, serving as a foundational reference for process chemists looking to implement this technology.
- Perform carbamation of 3-aminopyridine using alkyl chloroformate or di-tert-butyl dicarbonate to form the pyridine-3-ylcarbamate precursor.
- Conduct catalytic hydrogenation using Palladium on Carbon (Pd/C) in the presence of a carboxylic acid or phosphoric acid to maintain pH between 1 and 7.
- Execute optical resolution by forming diastereomeric salts with optically active mandelic acid or tartaric acid in alcohol solvents, followed by crystallization and free-basing.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN101977899A offers tangible strategic benefits that extend beyond simple chemical yield. The most immediate impact is seen in the drastic reduction of raw material costs associated with the catalyst system. By shifting from rhodium to palladium, manufacturers can insulate their production budgets from the volatile pricing of precious metals, ensuring more stable long-term costing models. Furthermore, the use of common carboxylic acids like acetic acid as reaction additives eliminates the need for specialized, expensive ligands or promoters, simplifying the bill of materials. This simplification translates directly into cost reduction in pharmaceutical intermediates manufacturing, as the sourcing of reagents becomes more straightforward and less prone to supply disruptions. The ability to use water and common alcohols as solvents further reduces the environmental footprint and waste disposal costs, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The substitution of rhodium catalysts with palladium on carbon represents a significant economic advantage, as palladium is generally more abundant and less expensive per unit of catalytic activity in this specific application. The process eliminates the need for costly ligand systems often required for asymmetric hydrogenation, relying instead on classical resolution which, while theoretically limited to 50% yield, is highly practical and scalable when the unwanted enantiomer can be recycled. The high conversion rates observed under acidic conditions mean that less starting material is wasted, and the overall throughput of the reactor is maximized. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to lower utility costs per kilogram of product produced.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals such as acetic acid, ethanol, and standard Pd/C catalysts significantly de-risks the supply chain compared to processes dependent on proprietary or scarce rhodium complexes. These materials are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks that can halt production lines. The robustness of the reaction to varying pH levels within the specified range also provides a buffer against minor fluctuations in reagent quality, ensuring consistent batch-to-batch performance. This reliability is crucial for maintaining continuous supply to downstream API manufacturers, preventing delays that could impact clinical trial timelines or commercial product launches.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard hydrogenation equipment and filtration units found in most multipurpose chemical plants. The use of aqueous workups and common organic solvents simplifies the separation and purification stages, making technology transfer from lab to pilot to commercial scale more predictable. From an environmental perspective, the avoidance of heavy metal residues associated with homogeneous rhodium catalysts simplifies the purification of the final product to meet stringent residual metal specifications. The ability to recover and recycle the resolving agent, such as mandelic acid, from the mother liquors further minimizes waste generation, supporting sustainability goals and reducing the costs associated with hazardous waste treatment and disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific experimental conditions and advantages highlighted in the patent documentation, providing clarity on how this method compares to traditional approaches. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this process for their specific intermediate needs.
Q: Why is palladium preferred over rhodium for this hydrogenation?
A: Palladium catalysts, specifically Pd/C, are significantly more economical than rhodium catalysts while achieving comparable or superior yields when the reaction pH is strictly controlled between 1 and 7 using acids like acetic acid.
Q: What is the critical factor for achieving high optical purity?
A: The selection of the resolving agent is crucial; using optically active mandelic acid (specifically R-mandelic acid for the R-enantiomer) in specific alcohol solvents allows for the formation of diastereomeric salts with high enantiomeric excess, often exceeding 90% ee.
Q: Can this process be scaled for industrial production?
A: Yes, the process utilizes common solvents like water, ethanol, and acetic acid, and operates at moderate temperatures and hydrogen pressures, making it highly suitable for commercial scale-up without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Piperidin-3-ylcarbamate Supplier
The technological innovations detailed in patent CN101977899A underscore the complexity and precision required to produce high-quality piperidin-3-ylcarbamate intermediates efficiently. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate these patented methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require material for early-stage development or full-scale manufacturing, we can meet your volume requirements without compromising on quality. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to verify stringent purity specifications, including enantiomeric excess and residual solvent levels, guaranteeing that every batch meets the exacting standards of the global pharmaceutical industry.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project. By leveraging our capabilities, you can access a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how the switch to palladium catalysis and optimized resolution can improve your bottom line. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on real-world performance data rather than theoretical projections. Let us be your trusted partner in navigating the complexities of fine chemical synthesis and securing a reliable supply of critical pharmaceutical intermediates.
