Advanced Synthesis of Stable Garcinia Acid Derivatives for Commercial Oncology Applications
Advanced Synthesis of Stable Garcinia Acid Derivatives for Commercial Oncology Applications
The pharmaceutical industry continuously seeks novel intermediates that offer superior therapeutic indices compared to natural extracts, and Patent CN1563014A represents a significant breakthrough in this domain by detailing the modification of three key isomers of total garcinolic acid. This intellectual property outlines the preparation of new compounds derived from morellic acid, neogambogic acid, and allogambogic acid, specifically engineered to overcome the inherent limitations of the parent natural products. By strategically modifying functional groups while preserving the core pharmacophore, these derivatives achieve higher stability, improved solubility profiles, and reduced toxic side effects, making them ideal candidates for next-generation anticancer and broad-spectrum antimicrobial agents. For R&D teams focusing on oncology, this technology provides a robust pathway to develop drugs that selectively target cancer cells without compromising the normal hemopoietic system, addressing a critical pain point in current chemotherapy regimens.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional utilization of gamboge resin and its primary active constituent, morellic acid, has been historically plagued by significant physicochemical and pharmacological challenges that hinder widespread clinical adoption. While natural morellic acid demonstrates potent anticancer activity, it suffers from poor water solubility and extreme instability in alkaline environments, leading to rapid hydrolysis that compromises drug efficacy during formulation and storage. Furthermore, conventional extracts often exhibit considerable toxicity, causing local irritant effects on blood vessels and inducing superficial phlebitis when administered intravenously, which severely limits the dosage and duration of treatment protocols. The reliance on crude extraction methods also results in inconsistent purity levels, making it difficult to meet the stringent regulatory requirements for modern pharmaceutical ingredients, thereby creating supply chain bottlenecks for manufacturers aiming to produce standardized injectable formulations or oral solid dosages.
The Novel Approach
The innovative methodology described in the patent circumvents these issues through precise chemical derivatization that enhances the molecular properties of the garcinia acid scaffold without destroying its biological activity. By selectively reducing ketone carbonyl groups at positions 8C and 12C to hydroxyl groups, the new compounds gain substantial chemical stability while maintaining their ability to inhibit tumor growth effectively. Additionally, the conversion of the carboxyl group at 30C into esters or amides drastically improves solubility characteristics, allowing for better partition ratios between lipid and aqueous phases which is essential for bioavailability. This approach not only mitigates the vascular irritation associated with the parent compound but also enables the creation of pharmaceutically acceptable salts that are stable in neutral or slightly acidic aqueous solutions, thus opening new avenues for diverse drug delivery systems ranging from injections to complex tablets.
Mechanistic Insights into Selective Reduction and Functionalization
The core of this synthetic strategy lies in the controlled manipulation of the polyprenylated xanthone skeleton, where specific reagents are employed to target distinct functional groups with high chemoselectivity. For instance, the reduction of ketone carbonyls is achieved using mild reducing agents like sodium borohydride or lithium aluminum hydride in alcoholic solvents, where the stoichiometry of the reagent dictates whether single or double reduction occurs, allowing for fine-tuning of the final product profile. Simultaneously, the carboxyl group undergoes activation via coupling agents such as EDC and DMAP to form stable esters or amides, a process that protects the molecule from alkaline hydrolysis while introducing lipophilic or hydrophilic moieties as needed. This dual-modification strategy ensures that the resulting derivatives possess a balanced amphiphilic nature, crucial for crossing cell membranes and reaching intracellular targets, while the preservation of the conjugated double bond system maintains the electronic properties required for interaction with biological receptors.
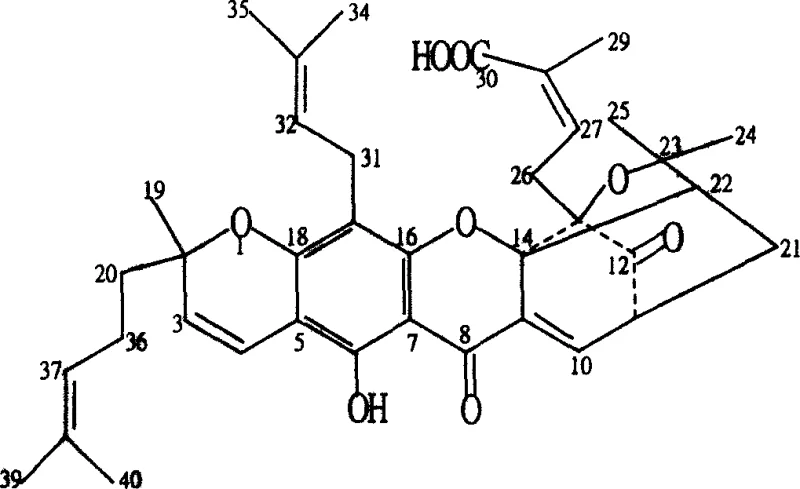
Furthermore, the patent details advanced halogenation and substitution reactions that introduce halogen atoms or amino groups onto the prenyl side chains, further diversifying the chemical space available for structure-activity relationship studies. These modifications are performed under controlled low-temperature conditions to prevent unwanted side reactions, ensuring that the stereochemistry of the chiral centers remains intact throughout the synthesis. The ability to substitute halogen atoms with nucleophiles like amines or sulfonates allows for the creation of water-soluble salts, which directly addresses the formulation challenges posed by the hydrophobic nature of the original natural product. This comprehensive mechanistic approach provides a versatile platform for generating a library of analogs, enabling medicinal chemists to optimize potency and safety profiles systematically before advancing candidates into preclinical development stages.
How to Synthesize Garcinia Acid Derivatives Efficiently
The synthesis of these high-value intermediates requires a disciplined approach to reaction conditions and purification to ensure the high purity necessary for pharmaceutical applications. The process begins with the isolation of highly purified total cambogic acid, followed by sequential modification steps that must be carefully monitored to control regioselectivity and yield. Detailed standardized synthesis steps see the guide below.
- Dissolve purified morellic acid in organic solvent and perform selective reduction of ketone carbonyls using sodium borohydride or lithium aluminum hydride.
- Conduct esterification or amidation at the C30 carboxyl group using DMAP and EDC coupling agents to enhance solubility.
- Perform halogen addition or substitution reactions on the double bonds to create stable halogenated derivatives with improved pharmacological profiles.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of these synthetic derivatives offers transformative advantages over relying on variable natural extracts, primarily by stabilizing the supply of critical oncology intermediates. The shift from extraction-dependent sourcing to semi-synthetic manufacturing reduces the risk of supply disruptions caused by agricultural variability or seasonal harvesting constraints, ensuring a consistent flow of materials for drug production. Moreover, the enhanced stability of the derivatives minimizes waste during storage and transportation, as the compounds are less prone to degradation under standard logistics conditions, thereby lowering the total cost of ownership for inventory management. This reliability is paramount for maintaining continuous manufacturing schedules in the highly regulated pharmaceutical sector, where batch failures due to原料 instability can result in significant financial losses and regulatory setbacks.
- Cost Reduction in Manufacturing: The synthetic routes described utilize widely available organic reagents and standard catalytic systems, avoiding the need for exotic or prohibitively expensive catalysts that often drive up the cost of goods sold. By streamlining the purification process through established techniques like silica gel chromatography and crystallization, manufacturers can achieve high purity levels without resorting to complex and costly preparative HPLC methods on a large scale. The elimination of instability-related losses means that overall process efficiency is significantly improved, as fewer batches are rejected due to degradation, leading to substantial cost savings in the long run. Additionally, the ability to create stable salts simplifies the final drug formulation process, potentially reducing the number of excipients required and shortening the development timeline for finished dosage forms.
- Enhanced Supply Chain Reliability: Transitioning to a semi-synthetic supply model decouples production from the geopolitical and environmental risks associated with sourcing raw plant resins from specific geographic regions. This localization of manufacturing capability allows for greater agility in responding to market demand surges, as production capacity can be scaled up in controlled industrial facilities rather than being limited by biomass availability. The robustness of the chemical intermediates ensures that they can be stockpiled safely for extended periods, providing a strategic buffer against unexpected supply chain shocks. This resilience is a critical factor for supply chain heads who prioritize business continuity and risk mitigation in their vendor selection criteria, especially for life-saving oncology medications.
- Scalability and Environmental Compliance: The reaction conditions specified in the patent, such as ambient temperature stirring and standard solvent systems, are inherently scalable from laboratory benchtop to multi-ton commercial production without requiring specialized high-pressure or cryogenic equipment. This ease of scale-up facilitates rapid technology transfer to contract manufacturing organizations, accelerating the time to market for new drug candidates. Furthermore, the processes avoid the generation of heavy metal waste often associated with traditional catalytic hydrogenation or cross-coupling reactions, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The use of recyclable solvents and straightforward workup procedures contributes to a greener manufacturing footprint, enhancing the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology in pharmaceutical development pipelines. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the value proposition for stakeholders.
Q: How do these derivatives improve upon natural Morellic acid stability?
A: The derivatives modify unstable functional groups, specifically reducing ketone carbonyls to hydroxyls and esterifying carboxyl groups, which prevents hydrolysis in alkaline conditions.
Q: What is the primary advantage for pharmaceutical formulation?
A: The new compounds exhibit significantly better water solubility and partition ratios between fat and aqueous phases, facilitating easier drug delivery system development.
Q: Are these intermediates suitable for large-scale production?
A: Yes, the synthesis utilizes standard organic reagents and purification methods like silica gel chromatography that are adaptable to industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Garcinia Acid Derivatives Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready supply is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for oncology intermediates, guaranteeing that every batch of Garcinia acid derivatives adheres to the highest quality standards. We understand the critical nature of these compounds in cancer therapy and are committed to providing a secure, compliant, and high-quality supply chain that supports your global clinical and commercial needs. Our technical team is ready to collaborate on process optimization to further enhance yield and reduce environmental impact, aligning with your sustainability objectives.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project requirements. By engaging with us early in your development cycle, you can leverage our expertise to conduct a Customized Cost-Saving Analysis that identifies opportunities for process intensification and raw material optimization. Let us be your strategic partner in bringing these promising anticancer agents from the patent lab to the patient bedside, ensuring reliability and excellence at every step of the journey.
