Advanced Synthesis of Tirofiban Hydrochloride: A Cost-Effective and Scalable API Intermediate Strategy
The pharmaceutical industry continuously seeks robust manufacturing processes for critical cardiovascular medications, and the synthesis of Tirofiban Hydrochloride represents a significant area of innovation. As detailed in patent CN115448871A, a novel preparation method has been developed that fundamentally restructures the synthetic pathway to enhance safety, purity, and cost-efficiency. Tirofiban, a potent glycoprotein IIb/IIIa inhibitor, requires stringent control over impurities, particularly heavy metals, to meet global regulatory standards. The traditional reliance on L-Tyrosine derivatives often introduces complex purification challenges late in the synthesis. This new approach pivots to a Boc-protected piperidine starting material, allowing for the strategic placement of catalytic steps early in the sequence. By addressing the inherent limitations of prior art, this technology offers a compelling value proposition for manufacturers aiming to optimize their supply chains for high-purity pharmaceutical intermediates.
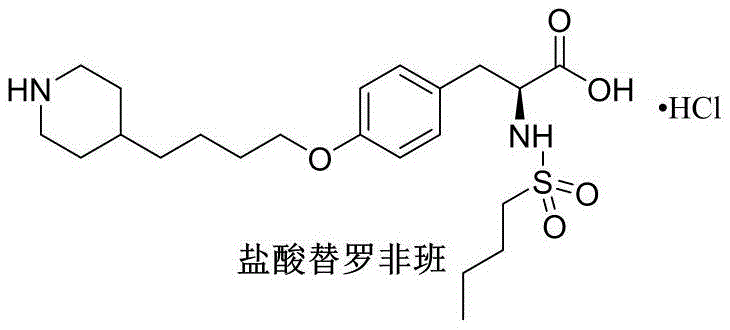
The structural complexity of Tirofiban Hydrochloride demands a synthesis strategy that balances stereochemical integrity with process simplicity. The molecule features a chiral tyrosine core linked via an ether bond to a piperidine-containing alkyl chain. Conventional methods often struggle to assemble this architecture without introducing difficult-to-remove contaminants. The breakthrough described in the patent lies in the disconnection of the molecule at the ether linkage, utilizing a pre-functionalized piperidine chain that is synthesized independently before coupling. This modular approach not only simplifies the reaction landscape but also isolates potential impurity sources, ensuring that the final active pharmaceutical ingredient (API) meets the rigorous quality specifications required by international health authorities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Tirofiban has relied heavily on routes originating from L-Tyrosine, which, while chemically logical, present substantial operational and economic drawbacks. One prevalent method, illustrated in Scheme 1 of the background art, involves a Mitsunobu reaction to form the critical ether bond. This reaction typically employs diethyl azodicarboxylate (DEAD) and triphenylphosphine, reagents that are not only expensive but also generate stoichiometric amounts of phosphine oxide byproducts that are notoriously difficult to separate from the product. Furthermore, this route positions the catalytic hydrogenation of a pyridine ring as the penultimate step. Performing Pd/C catalysis so late in the synthesis creates a severe risk of palladium contamination in the final drug substance, necessitating costly scavenging treatments and rigorous testing to ensure compliance with strict heavy metal limits.
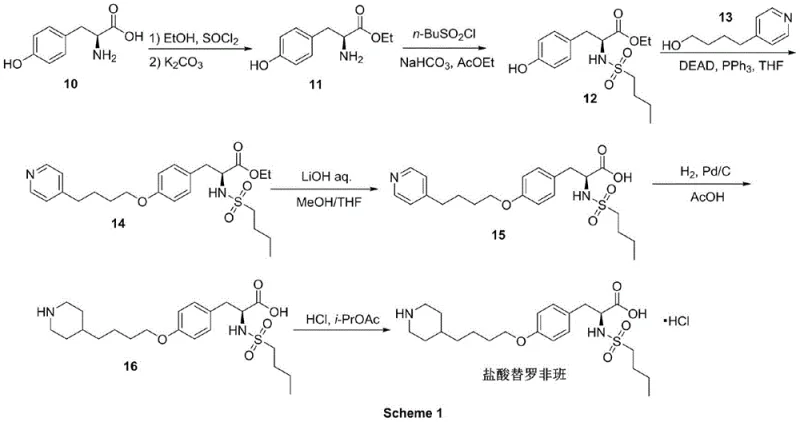
Another existing pathway, depicted in Scheme 2, attempts to shorten the sequence but introduces significant safety hazards. This method utilizes n-butyllithium to generate a nucleophile for coupling, a reagent that is pyrophoric and requires handling under inert atmospheres at cryogenic temperatures, increasing both capital expenditure for specialized equipment and operational risk. Additionally, the lack of ester protection on the tyrosine moiety in early stages leads to higher polarity intermediates, complicating extraction and purification processes. The persistence of late-stage hydrogenation in this route means that the issues of high-pressure equipment requirements and potential heavy metal residues remain unresolved, creating a bottleneck for efficient commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the novel synthesis route disclosed in CN115448871A offers a streamlined and safer alternative that fundamentally alters the process timeline. By initiating the synthesis with 4-(2-oxoethyl)piperidine-1-carboxylic acid tert-butyl ester, the process builds the alkyl chain first, allowing the catalytic hydrogenation step to occur at the second stage of the synthesis rather than the end. This strategic shift means that the palladium catalyst is introduced and subsequently removed via simple filtration when the molecule is still a simple intermediate. Consequently, the downstream chemistry proceeds in a metal-free environment, drastically reducing the risk of heavy metal carryover into the final API. The route avoids hazardous reagents like n-butyllithium and explosive azo compounds, relying instead on robust transformations such as Wittig olefination and nucleophilic substitution under mild thermal conditions.
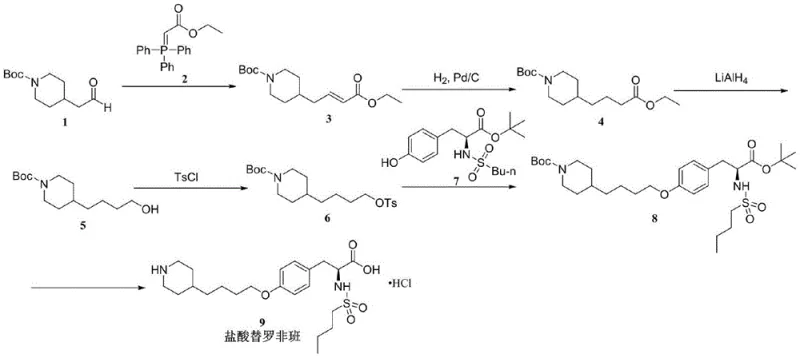
The efficiency of this new approach is further evidenced by its operational simplicity and high yield profile. The reaction conditions are温和 (mild), typically ranging from 25°C to 80°C, eliminating the need for energy-intensive cryogenic cooling or high-pressure reactors. The use of common organic solvents like dichloromethane, ethanol, and acetonitrile ensures that the process is compatible with standard multipurpose chemical manufacturing facilities. By decoupling the construction of the piperidine side chain from the tyrosine core until the later stages, the synthesis allows for better control over impurity profiles. The final deprotection step using hydrochloric acid cleanly removes both the Boc and t-butyl protecting groups simultaneously, delivering the target Tirofiban Hydrochloride with purity exceeding 99.8% and yields reaching up to 90% in the final isolation, demonstrating superior process robustness compared to traditional methods.
Mechanistic Insights into the Sequential Synthetic Strategy
The core of this innovative synthesis lies in the precise sequencing of functional group transformations to maximize orthogonality and minimize side reactions. The process begins with a Wittig reaction between the Boc-protected piperidine aldehyde and ethoxycarbonyl methylene triphenylphosphine. This step effectively extends the carbon chain by two carbons while installing an ester functionality, setting the stage for subsequent reduction. The resulting alpha,beta-unsaturated ester is then subjected to catalytic hydrogenation. Mechanistically, this saturation of the double bond is straightforward, but its placement is critical; by performing this reduction early, the palladium catalyst interacts only with a relatively simple substrate, minimizing the formation of complex organometallic species that could trap metal ions. Following filtration, the saturated ester undergoes reduction with lithium aluminum hydride to yield the primary alcohol, a transformation that proceeds with high chemoselectivity in the presence of the carbamate protecting group.
Subsequent activation of the primary alcohol via tosylation converts a poor leaving group into an excellent one, facilitating the crucial etherification step. The nucleophilic substitution between the tosylated piperidine derivative and the phenolic oxygen of the protected tyrosine intermediate is driven by a mild base such as potassium carbonate. This SN2-type displacement is highly efficient and avoids the racemization risks associated with harsher alkylation conditions. The final mechanistic event is the acid-mediated deprotection. Treatment with concentrated hydrochloric acid cleaves the tert-butyl carbamate (Boc) and the tert-butyl ester simultaneously. This dual deprotection is thermodynamically favorable and generates volatile byproducts (isobutylene and CO2), which drive the reaction to completion and simplify the workup, ensuring that the final salt formation occurs cleanly without the need for additional basification and re-acidification steps.
How to Synthesize Tirofiban Hydrochloride Efficiently
Implementing this synthesis requires careful attention to reaction monitoring and workup procedures to maintain the high purity standards demanded by the pharmaceutical sector. The process is divided into six distinct operational units, starting from the Wittig coupling and ending with the final crystallization of the hydrochloride salt. Each step has been optimized to balance reaction rate with impurity control, utilizing standard laboratory and plant equipment. The following guide outlines the critical operational parameters derived from the patent examples, providing a roadmap for technical teams to replicate this high-efficiency route. For the detailed standardized synthesis steps, please refer to the guide below.
- Perform a Wittig reaction between Boc-piperidine acetaldehyde and ethoxycarbonyl methylene triphenylphosphine to form the alpha,beta-unsaturated ester intermediate.
- Execute catalytic hydrogenation using Pd/C at ambient temperature to saturate the double bond, followed by filtration to remove palladium early in the sequence.
- Reduce the ester group to a primary alcohol using lithium aluminum hydride, then activate the alcohol via tosylation for subsequent etherification.
- Couple the tosylated intermediate with the protected tyrosine derivative under alkaline conditions to form the ether linkage.
- Remove all protecting groups (Boc and t-butyl ester) simultaneously using hydrochloric acid to yield the final Tirofiban Hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates directly into tangible operational improvements and risk mitigation. The primary value driver is the significant reduction in processing complexity, which inherently lowers the cost of goods sold (COGS). By eliminating the need for expensive and hazardous reagents like DEAD and n-butyllithium, the raw material costs are stabilized, and the safety infrastructure requirements are reduced. Furthermore, the early removal of the palladium catalyst removes a major bottleneck in the purification workflow. In traditional late-stage hydrogenation processes, extensive and costly metal scavenging resins are often required to meet regulatory limits, adding both material cost and processing time. This new route bypasses that need entirely, as the metal is removed when the batch volume is smaller and the chemistry is simpler.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the simplification of the unit operations and the elimination of high-cost reagents. Traditional Mitsunobu reactions generate large amounts of phosphine oxide waste, the disposal of which adds to the environmental compliance costs. By replacing this with a tosylation-substitution sequence, the waste profile is significantly improved, leading to lower waste treatment expenses. Additionally, the avoidance of cryogenic conditions reduces energy consumption, as the reactions proceed efficiently at ambient or slightly elevated temperatures. The high yields reported in the patent examples suggest a more atom-economical process, meaning less raw material is wasted, directly contributing to substantial cost savings in pharmaceutical intermediates manufacturing without compromising on quality.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on specialized or hazardous reagents that may face shipping restrictions or availability fluctuations. This new method utilizes commodity chemicals such as potassium carbonate, triethylamine, and common solvents, which are readily available from multiple global suppliers. The robustness of the chemistry also means that the process is less sensitive to minor variations in raw material quality, reducing the risk of batch failures. By simplifying the synthesis to avoid high-pressure hydrogenation in the final steps, the dependency on specialized high-pressure reactor vessels is minimized, allowing for production in a wider range of manufacturing facilities, thereby diversifying the supply base and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this route is designed with green chemistry principles in mind. The absence of pyrophoric reagents and the use of mild reaction conditions make the technology inherently safer for large-scale production, reducing the insurance and safety compliance burden on the manufacturer. The solvent systems used are largely recyclable, and the reduction in heavy metal usage aligns with increasingly stringent environmental regulations regarding metal discharge. The process generates fewer toxic byproducts compared to the azo-compound waste from Mitsunobu reactions, facilitating easier waste management and supporting the company's sustainability goals while ensuring the commercial scale-up of complex pharmaceutical intermediates remains viable and compliant.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis method is crucial for stakeholders evaluating its integration into their supply chains. The following questions address common concerns regarding quality control, safety, and scalability, based on the data provided in the patent documentation. These insights are intended to clarify how this process overcomes the specific pain points associated with legacy manufacturing technologies, offering a clearer path toward efficient and compliant production.
Q: How does this new synthesis route address heavy metal residue concerns in Tirofiban production?
A: Unlike conventional methods that perform catalytic hydrogenation in the penultimate step, this novel process conducts Pd/C hydrogenation at the second step. The palladium catalyst is filtered off immediately, ensuring that Intermediate 6 contains less than 10ppm of Pd before the final coupling and deprotection stages, thereby eliminating the need for expensive heavy metal scavenging in the final API.
Q: What are the safety advantages of this method compared to traditional L-Tyrosine routes?
A: Traditional routes often rely on hazardous reagents such as n-butyllithium (pyrophoric) or Mitsunobu reagents like DEAD (potentially explosive). This new route utilizes stable, commercially available starting materials and operates under mild conditions (25°C to 80°C) without the need for high-pressure hydrogenation equipment in the final stages, significantly enhancing operational safety.
Q: Is this synthesis method suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. It avoids cryogenic conditions and high-pressure reactions, uses common organic solvents like dichloromethane and ethanol, and achieves high yields (up to 90% in the final step) with purity exceeding 99.8%, making it highly viable for industrial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tirofiban Hydrochloride Supplier
The technological advancements detailed in patent CN115448871A represent a significant leap forward in the manufacturing of cardiovascular API intermediates, yet realizing this potential requires a partner with deep technical expertise and scalable infrastructure. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that ensure every batch meets the highest international standards. We understand that transitioning to a new synthetic route involves careful validation, and our team is equipped to manage this process seamlessly, ensuring supply continuity and product excellence for our global partners.
We invite procurement leaders and R&D directors to explore how this optimized synthesis can benefit their specific projects. By partnering with us, you gain access not just to a product, but to a comprehensive Customized Cost-Saving Analysis that quantifies the economic advantages of this new route for your organization. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your volume requirements. Let us collaborate to drive efficiency and innovation in your supply chain, ensuring that you have a reliable source for high-quality Tirofiban Hydrochloride that supports your mission to deliver life-saving medications to patients worldwide.
