Advanced Synthetic Route for Lenvatinib Intermediates Enhancing Purity and Scalability
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical oncology therapeutics, and the synthesis of Lenvatinib stands as a prime example of process evolution. As detailed in patent CN108623521B, a novel preparation method has been developed that fundamentally restructures the synthetic approach to this multi-kinase inhibitor. This technology addresses long-standing challenges in impurity management and operational safety, offering a streamlined alternative to legacy processes. By leveraging a strategic nitro-protection motif, the method achieves a remarkable total yield of approximately 84% and a final product purity of 99.7%. For R&D directors and procurement managers alike, this represents a significant opportunity to enhance the reliability of the supply chain for high-purity API intermediates while mitigating the environmental and safety risks associated with traditional chloroformate-based chemistries.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthesis routes, specifically designated as Route A and Route B in the technical literature, suffer from inherent chemical inefficiencies that complicate large-scale manufacturing. Route A involves the direct condensation of unprotected amine intermediates, a strategy that frequently leads to the generation of complex impurity profiles due to the high reactivity of the free amino group. Similarly, Route B necessitates the use of phenyl chloroformate, a reagent known for its significant toxicity and the production of phenol as a hazardous byproduct during subsequent urea formation. These legacy methods typically cap out at a total yield of around 70%, requiring extensive purification efforts to meet the stringent quality standards required for clinical-grade active pharmaceutical ingredients. The reliance on toxic reagents not only escalates waste treatment costs but also introduces substantial safety liabilities for production facilities handling these materials on a commercial scale.
The Novel Approach
The innovative methodology disclosed in the patent circumvents these issues by introducing a nitro-protected phenol building block early in the synthesis. This strategic modification prevents unwanted side reactions associated with free amines, thereby drastically simplifying the purification landscape. Instead of toxic chloroformates, the process utilizes safer coupling agents in the final steps, such as cyclopropyl isocyanate or carbamate derivatives. This shift not only improves the overall mass balance but also enhances the operational safety profile of the plant. The result is a more linear, predictable process flow that supports consistent batch-to-batch quality, making it an ideal candidate for cost reduction in pharmaceutical manufacturing where yield and purity are the primary drivers of margin.
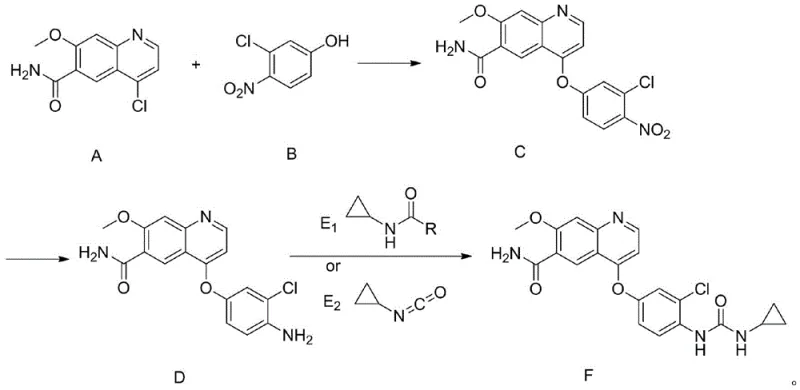
Mechanistic Insights into Nitro-Protection and Catalytic Reduction
The core of this technological advancement lies in the precise control of functional group reactivity through the nitro-protection strategy. The first stage involves a nucleophilic aromatic substitution where 4-chloro-7-methoxy-6-quinolinecarboxamide reacts with 3-chloro-4-nitrophenol. This reaction is facilitated by strong inorganic bases such as cesium carbonate or potassium carbonate in polar aprotic solvents like DMSO at temperatures ranging from 60°C to 100°C. The electron-withdrawing nature of the nitro group activates the phenol for coupling while simultaneously deactivating the ring towards oxidation, ensuring high conversion rates without the need for sensitive protecting group manipulations. This step establishes the critical ether linkage with exceptional efficiency, often exceeding 90% yield in isolated examples, setting a strong foundation for the subsequent transformations.
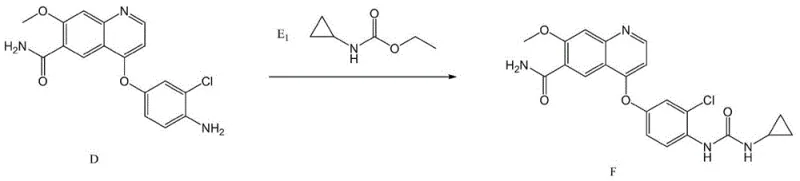
Following the etherification, the process employs a catalytic hydrogenation step to reveal the reactive amine functionality required for the final urea assembly. Using catalysts such as 10% Pd/C or Raney Nickel under hydrogen pressures of 0.1 MPa to 3 MPa, the nitro group is selectively reduced to an amino group. This transformation is remarkably clean, with patent data indicating yields as high as 99.5% for this specific step. The final coupling can proceed via two distinct pathways: reaction with a carbamate derivative (Compound E1) under basic conditions or direct reaction with cyclopropyl isocyanate (Compound E2). Both pathways effectively construct the urea moiety, but the isocyanate route offers particular advantages in atom economy. This mechanistic clarity allows for rigorous impurity control, ensuring that the final API meets the strict specifications demanded by global regulatory bodies.
How to Synthesize Lenvatinib Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly during the base-mediated etherification and the final urea formation steps. The protocol outlined in the patent provides a clear roadmap for transitioning from laboratory scale to pilot production, emphasizing the use of readily available solvents and catalysts. Operators should note that the choice of base in the first step significantly influences the reaction rate and impurity profile, with cesium carbonate showing superior performance in specific embodiments. Furthermore, the flexibility in the final coupling step allows manufacturers to select the most cost-effective reagent based on local supply chain availability. For a comprehensive breakdown of the specific molar ratios, temperature gradients, and workup procedures, please refer to the standardized guide below.
- Perform nucleophilic aromatic substitution between 4-chloro-7-methoxy-6-quinolinecarboxamide and 3-chloro-4-nitrophenol using a base like cesium carbonate in DMSO at 60-100°C.
- Reduce the nitro group of the intermediate to an amino group using catalytic hydrogenation with Pd/C or Raney Ni under 0.1-3 MPa pressure.
- React the resulting amine with either ethyl cyclopropylcarbamate or cyclopropyl isocyanate to form the final urea linkage of Lenvatinib.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers compelling value propositions for supply chain heads and procurement managers focused on long-term stability and cost efficiency. By eliminating the need for highly toxic phenyl chloroformate, the process removes a significant bottleneck related to hazardous material handling and disposal. This reduction in hazard classification translates directly into lower insurance premiums and simplified logistics for raw material transport. Additionally, the higher overall yield means that less starting material is required to produce the same amount of final API, effectively lowering the cost of goods sold without compromising on quality. The robustness of the chemistry also suggests a lower risk of batch failures, ensuring a more consistent supply of critical intermediates for downstream formulation.
- Cost Reduction in Manufacturing: The elimination of expensive and toxic reagents like phenyl chloroformate significantly lowers raw material costs and waste treatment expenses. The improved yield profile means less solvent and energy are consumed per kilogram of product, driving down the overall manufacturing footprint. Furthermore, the simplified purification requirements reduce the load on chromatography and crystallization units, allowing for faster throughput and lower utility costs.
- Enhanced Supply Chain Reliability: The starting materials for this route, including the nitro-phenol derivative and quinoline chloride, are commercially available from multiple global suppliers, reducing dependency on single-source vendors. The mild reaction conditions and use of standard catalysts like Pd/C ensure that the process can be easily replicated across different manufacturing sites, providing redundancy and security against regional supply disruptions.
- Scalability and Environmental Compliance: The process avoids the generation of phenolic waste streams, which are notoriously difficult and costly to treat in wastewater facilities. This aligns perfectly with modern green chemistry initiatives and strict environmental regulations. The straightforward workup procedures, primarily involving filtration and crystallization, are inherently scalable from 100 kgs to multi-ton production without requiring specialized high-pressure or cryogenic equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this novel Lenvatinib synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is crucial for technical teams evaluating the feasibility of adopting this route for commercial production. The answers highlight the specific advantages in terms of purity, safety, and operational simplicity that distinguish this method from prior art.
Q: How does the new synthesis route improve impurity control compared to conventional methods?
A: Conventional routes often utilize unprotected amines which lead to side reactions and difficult-to-remove impurities. The novel method employs a nitro-protected phenol intermediate, preventing premature reactions and ensuring a cleaner profile with purity reaching 99.7%.
Q: What are the safety advantages of avoiding phenyl chloroformate in this process?
A: Traditional Route B relies on phenyl chloroformate, a toxic reagent that generates hazardous phenol byproducts. The new method eliminates this requirement, significantly reducing toxicity risks and simplifying waste treatment protocols for safer industrial operation.
Q: What is the expected total yield for this optimized Lenvatinib preparation method?
A: According to the patent data, the optimized three-step sequence achieves a total yield of approximately 84%, which represents a substantial improvement over the roughly 70% yield reported for prior art methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lenvatinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to reactor is seamless and efficient. We are committed to delivering high-purity intermediates that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement complex chemistries, such as the nitro-protection strategy described here, underscores our position as a leader in custom synthesis and process development.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits specific to your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of technical confidence and commercial viability.
