Revolutionizing Lenvatinib Production: A Green, Low-Temperature Synthetic Route for Commercial Scale-Up
Introduction to Advanced Lenvatinib Synthesis
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for producing complex oncology therapeutics like Lenvatinib (E7080), a potent multi-receptor tyrosine kinase inhibitor. A pivotal advancement in this domain is detailed in patent CN110981800A, which discloses a novel preparation method that fundamentally reimagines the construction of the quinoline core. This intellectual property introduces a strategic shift by utilizing 4-nitro-2-chlorobenzonitrile as the initial raw material, leveraging the powerful electron-withdrawing nature of the nitro group to facilitate nucleophilic substitution under remarkably mild conditions. Unlike traditional methods that rely on harsh thermal energy and toxic reagents, this innovative approach integrates green chemistry principles, such as the use of Zinc Oxide (ZnO) as a recoverable Lewis acid catalyst. For R&D directors and procurement strategists, this patent represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates, ensuring both regulatory compliance and economic viability in the competitive landscape of cancer treatment manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Lenvatinib intermediates has been plagued by significant operational and environmental challenges that hinder efficient commercial scale-up. One prevalent prior art route, as illustrated in the accompanying diagram, initiates with 4-cyano-3-chloroaniline and necessitates reaction in high-boiling point solvents like N-methyl-2-pyrrolidone (NMP), which are notoriously difficult to remove and recycle. Furthermore, alternative schemes have relied on 4-aminosalicylic acid reacted with dimethyl sulfate, a reagent classified as highly toxic and carcinogenic, posing severe safety risks to personnel and requiring extensive waste treatment infrastructure. These conventional pathways often demand high-temperature cyclization steps in diphenyl ether, leading to increased energy consumption and the formation of complex impurity profiles that are difficult to purge. The reliance on such harsh conditions not only escalates production costs but also introduces substantial supply chain vulnerabilities due to the stringent handling requirements of hazardous materials.
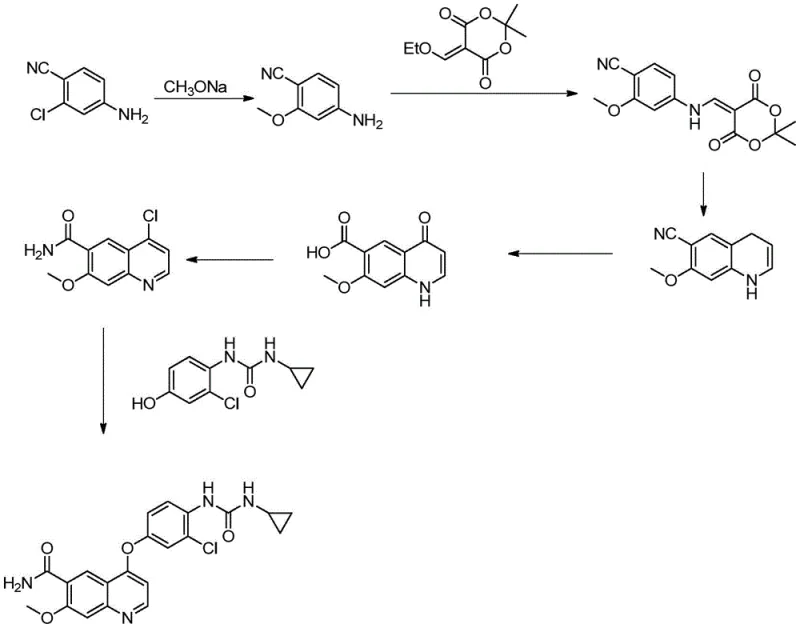
The Novel Approach
In stark contrast, the methodology disclosed in patent CN110981800A offers a streamlined and chemically elegant solution that bypasses these historical bottlenecks. By selecting 4-nitro-2-chlorobenzonitrile as the starting scaffold, the inventors exploit the electronic properties of the nitro group to drastically reduce the electron cloud density of the benzene ring. This activation allows for nucleophilic substitution with sodium methoxide to proceed rapidly at significantly lower temperatures, eliminating the need for energy-intensive heating. The subsequent steps involve a clever sequence of amino protection, acylation, and intramolecular alkylation that avoids the use of dangerous high-temperature environments. As depicted in the reaction scheme below, this novel route ensures a cleaner reaction profile with fewer byproducts, directly translating to higher yields and simplified downstream processing. This paradigm shift not only enhances the safety of the manufacturing process but also aligns perfectly with modern green chemistry mandates, making it an ideal candidate for reliable pharmaceutical intermediate supplier partnerships.
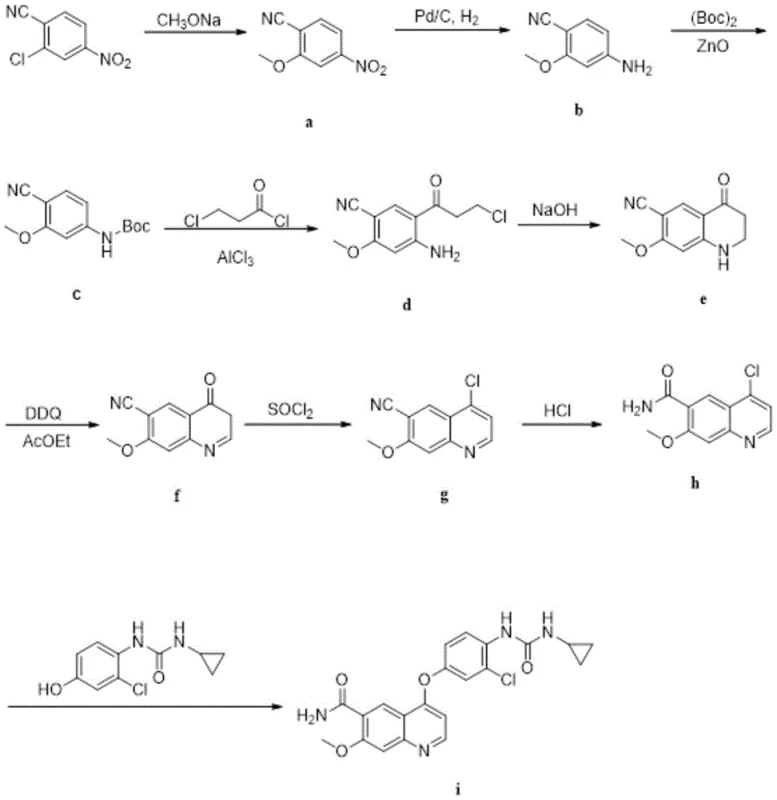
Mechanistic Insights into Nitro-Activated Substitution and Heterogeneous Catalysis
The core chemical innovation driving this process lies in the strategic manipulation of electronic effects and catalyst heterogeneity to control reaction kinetics and purity. In the initial step, the presence of the nitro group ortho to the chlorine atom creates a strong electron-deficient environment on the aromatic ring. This facilitates the attack of the methoxide nucleophile, lowering the activation energy required for the substitution reaction and preventing the formation of regio-isomers that often plague less activated systems. Following the reduction of the nitro group to an amine, the process employs Zinc Oxide (ZnO) as a Lewis acid catalyst for the Boc-protection step. Unlike homogeneous catalysts that remain dissolved and require complex aqueous workups to remove, ZnO operates as a heterogeneous solid. This physical state allows the catalyst to be easily separated from the reaction mixture via simple filtration, ensuring that no metal residues contaminate the final API, a critical factor for meeting stringent heavy metal specifications in oncology drugs.
Furthermore, the mechanism of the Friedel-Crafts acylation step demonstrates exceptional process efficiency through a tandem deprotection strategy. When the Boc-protected intermediate reacts with 3-chloropropionyl chloride in the presence of aluminum trichloride, the hydrochloric acid generated in situ serves a dual purpose: it catalyzes the acylation and simultaneously removes the amino protecting group. This "one-pot" functionality reduces the number of unit operations, minimizing solvent usage and material loss between steps. The subsequent intramolecular alkylation under alkaline conditions closes the quinoline ring with high fidelity. By avoiding the use of oxidizing agents until the very end (using DDQ for aromatization), the route preserves the integrity of sensitive functional groups throughout the synthesis. This meticulous control over reaction mechanisms ensures a robust impurity profile, providing R&D teams with a predictable and scalable pathway for producing high-purity pharmaceutical intermediates.
How to Synthesize 6-Carboxamido-7-methoxy-4-chloroquinoline Efficiently
The synthesis of the key Lenvatinib intermediate, 6-carboxamido-7-methoxy-4-chloroquinoline, via this novel route involves a precise sequence of nine chemical transformations that prioritize safety and yield. The process begins with the activation of the benzene ring through nitro-substitution, followed by selective reduction and protection strategies that set the stage for ring closure. Each step is optimized to minimize waste and maximize throughput, utilizing common reagents like sodium methoxide, palladium on carbon, and thionyl chloride under controlled conditions. The integration of heterogeneous catalysis and tandem reactions significantly reduces the operational complexity typically associated with quinoline synthesis. For technical teams looking to implement this methodology, the detailed standardized synthesis steps are provided in the guide below, ensuring reproducibility from the laboratory bench to the pilot plant.
- Perform nucleophilic substitution on 4-nitro-2-chlorobenzonitrile using sodium methoxide to introduce the methoxy group, leveraging the electron-withdrawing nitro group to lower reaction temperature.
- Execute catalytic hydrogenation to reduce the nitro group to an amine, followed by amino protection using di-tert-butoxycarbonyldicarbonate catalyzed by heterogeneous ZnO.
- Conduct Friedel-Crafts acylation with 3-chloropropionyl chloride and AlCl3, simultaneously removing the protecting group, followed by intramolecular alkylation and oxidative dehydrogenation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers compelling advantages that extend far beyond simple chemical yield improvements. The elimination of toxic reagents like dimethyl sulfate and high-boiling solvents like NMP directly addresses growing regulatory pressures regarding environmental, health, and safety (EHS) compliance. By shifting to a greener process profile, manufacturers can significantly reduce the costs associated with hazardous waste disposal and solvent recovery, leading to a leaner cost structure. Moreover, the use of readily available starting materials such as 4-nitro-2-chlorobenzonitrile ensures a stable supply base, mitigating the risk of raw material shortages that often disrupt production schedules. The mild reaction conditions also imply lower energy consumption, as the process does not require sustained high-temperature heating, further contributing to cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The implementation of this route drives down manufacturing expenses through several qualitative mechanisms. Firstly, the use of ZnO as a recoverable heterogeneous catalyst eliminates the need for expensive metal scavengers or complex purification steps to remove residual metals, which are costly and time-consuming. Secondly, the tandem deprotection-acylation step reduces the total number of reaction vessels and processing time required, effectively increasing facility throughput without capital expenditure. Finally, the avoidance of specialized high-temperature equipment lowers maintenance costs and extends the lifespan of reactor infrastructure, providing substantial long-term financial benefits.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the simplicity and robustness of the chemical transformations involved. Because the reaction conditions are mild and the reagents are commodity chemicals, the risk of batch failure due to thermal runaway or reagent instability is drastically minimized. This reliability translates to more consistent lead times for high-purity pharmaceutical intermediates, allowing downstream API manufacturers to plan their production schedules with greater confidence. Additionally, the simplified purification protocols mean that production bottlenecks are less likely to occur during the isolation and drying phases, ensuring a steady flow of material to meet market demand.
- Scalability and Environmental Compliance: Scaling complex organic syntheses often introduces unforeseen challenges, but this route is inherently designed for industrial expansion. The exothermic nature of the reactions is manageable under the described mild conditions, reducing the need for aggressive cooling systems that can be difficult to scale. From an environmental perspective, the reduction in toxic waste and the use of greener solvents align with global sustainability goals, making the final product more attractive to environmentally conscious partners. This compliance readiness ensures that the supply chain remains resilient against tightening environmental regulations, securing the long-term viability of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Lenvatinib synthesis pathway. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this technology for commercial production.
Q: How does the new synthesis route improve safety compared to traditional methods?
A: The novel route eliminates the use of highly toxic reagents like dimethyl sulfate and avoids high-boiling, difficult-to-remove solvents like NMP. By utilizing a nitro-activated starting material, the reaction temperatures are significantly lowered, reducing thermal hazards and equipment stress during commercial production.
Q: What is the advantage of using ZnO as a catalyst in this process?
A: Zinc oxide (ZnO) acts as a heterogeneous Lewis acid catalyst for the amino protection step. Unlike soluble bases or acids, ZnO is insoluble in the reaction system, allowing for simple filtration and effective recovery. This reusability contributes to a greener process profile and reduces waste generation.
Q: Is this synthetic route suitable for large-scale industrial manufacturing?
A: Yes, the process is specifically designed for industrial scalability. It features mild reaction conditions, avoids dangerous high-temperature steps, and utilizes readily available raw materials. The simplified purification steps, such as the easy removal of the heterogeneous catalyst, enhance operational efficiency for ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lenvatinib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in patent CN110981800A for the production of Lenvatinib intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry approach are fully realized in practice. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, including the management of heterogeneous catalysts and the precise temperature controls needed for the nitro-activation steps. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for oncology drug development, providing our partners with peace of mind regarding quality and consistency.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced technology for their supply chains. By partnering with our technical procurement team, you can gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this novel route for your organization. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in commercial scale-up of complex pharmaceutical intermediates can drive value and efficiency for your upcoming projects.
