Advanced Nickel-Catalyzed Synthesis of Tetramethylbiphenyl for High-Performance Polyimide Manufacturing
Advanced Nickel-Catalyzed Synthesis of Tetramethylbiphenyl for High-Performance Polyimide Manufacturing
The global demand for high-performance engineering plastics, particularly polyimides capable of withstanding temperatures exceeding 250°C, has driven intense research into efficient synthetic routes for their key monomers. Patent CN100465139C introduces a transformative methodology for the preparation of tetramethylbiphenyl, a critical intermediate in the production of biphenyl-type polyimides used extensively in aerospace and aviation sectors. This technology pivots away from traditional, waste-intensive zinc-mediated reductions, opting instead for a sophisticated nickel-catalyzed cross-coupling strategy utilizing novel hydroxy-imine spiro-nickel complexes. By leveraging the unique electronic properties of these organometallic catalysts, the process achieves superior selectivity and operational simplicity, addressing long-standing bottlenecks in the industrial scale-up of electronic chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of tetramethylbiphenyl has relied heavily on reductive coupling protocols that utilize halogenated xylenes reacted with excessive quantities of zinc powder. As illustrated in the traditional pathway, this method necessitates the formation of an organozinc intermediate prior to the coupling step, which is catalyzed by nickel complexes bearing triarylphosphine ligands. This conventional approach suffers from significant drawbacks, including the requirement for harsh reaction conditions and the generation of substantial amounts of zinc waste, which complicates downstream processing and environmental compliance. Furthermore, the catalysts employed in these legacy processes often require tedious pre-preparation steps under stringent conditions, adding unnecessary complexity and cost to the manufacturing workflow.
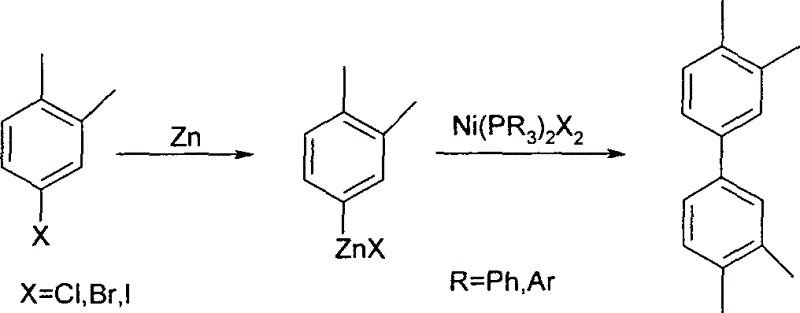
The Novel Approach
In stark contrast to the zinc-heavy legacy methods, the patented innovation employs a direct cross-coupling strategy between halogenated xylene and its corresponding Grignard reagent (xylene magnesium halide). This streamlined approach utilizes a newly developed class of organic nickel catalysts featuring hydroxy-imine ligands, which exhibit markedly higher catalytic efficiency. The reaction proceeds smoothly in common ether solvents such as tetrahydrofuran or diethyl ether, operating within a moderate temperature range from 25°C to reflux. By eliminating the need for stoichiometric zinc reduction and utilizing a more active catalyst system, this novel route not only simplifies the operational procedure but also significantly enhances the atom economy of the transformation, making it highly attractive for cost-sensitive fine chemical production.
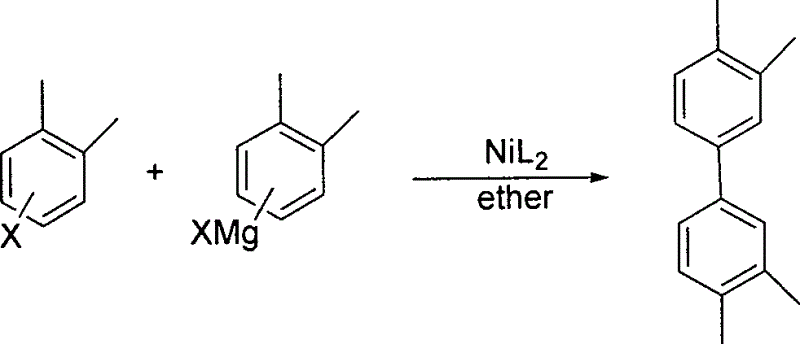
Mechanistic Insights into Hydroxy-Imine Spiro-Nickel Catalysis
The core breakthrough of this technology lies in the molecular architecture of the catalyst, specifically the hydroxy-imine spiro-nickel complexes. Unlike traditional triphenylphosphine ligands which can be sterically bulky and electronically saturated, the hydroxy-imine ligands coordinate to the nickel center through oxygen and nitrogen atoms. The presence of lone pair electrons on the nitrogen atom within the imine group, combined with the hydroxyl functionality, creates a coordination environment that facilitates easier ligand dissociation during the catalytic cycle. This lability is crucial for the oxidative addition and reductive elimination steps inherent to nickel-catalyzed coupling, resulting in catalytic activities that are several times higher than those observed with conventional phosphine-based systems.
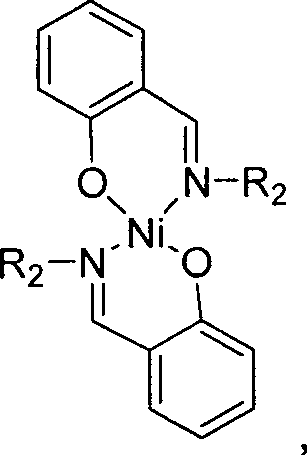
From an impurity control perspective, the enhanced selectivity of these spiro-nickel catalysts plays a pivotal role in ensuring product purity. The specific steric and electronic tuning provided by substituents such as the 2-fluorophenyl or tetrafluoro-4-chlorophenyl groups on the ligand framework helps suppress side reactions like homocoupling of the Grignard reagent or dehalogenation of the substrate. This intrinsic selectivity reduces the burden on purification units, allowing for the isolation of high-purity tetramethylbiphenyl suitable for polymerization into high-grade polyimides without extensive chromatographic cleanup, thereby preserving yield and reducing solvent consumption.
How to Synthesize Tetramethylbiphenyl Efficiently
The synthesis protocol outlined in the patent describes a robust two-step sequence that is amenable to standard chemical manufacturing equipment. The process begins with the in situ generation of the Grignard reagent, followed by the catalytic coupling step. This modular approach allows for precise control over reaction exotherms and reagent stoichiometry, ensuring consistent batch-to-batch quality. The detailed standardized synthesis steps, including specific molar ratios and temperature profiles optimized for maximum yield, are provided in the technical guide below.
- Prepare the Grignard reagent by reacting halogenated xylene with magnesium chips in an ether solvent like THF at temperatures between 0°C and 80°C.
- In a separate vessel, dissolve halogenated xylene and the novel hydroxy-imine spiro-nickel catalyst in an ether solvent under inert atmosphere.
- Add the Grignard reagent to the catalyst mixture, heat to reflux (25°C to reflux temperature), and stir for 4 to 24 hours to complete the coupling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this nickel-catalyzed methodology offers compelling economic and logistical benefits over traditional zinc-mediated processes. The primary advantage stems from the drastic simplification of the raw material profile; by removing the requirement for excessive zinc powder, manufacturers eliminate a major source of solid waste and the associated costs of hazardous waste disposal and treatment. Additionally, the catalyst itself is synthesized under mild conditions using readily available precursors like nickel acetate and salicylaldehyde derivatives, avoiding the complex supply chains often associated with specialized phosphine ligands or precious metal catalysts like palladium.
- Cost Reduction in Manufacturing: The elimination of stoichiometric zinc usage directly translates to reduced raw material costs and lower waste management expenses. Furthermore, the higher catalytic activity allows for potentially lower catalyst loading or shorter reaction times, which improves throughput and reduces energy consumption per kilogram of product. The simplified workup procedure, necessitated by the cleaner reaction profile, also minimizes solvent usage and labor hours required for purification, contributing to substantial overall cost savings in the manufacturing of polyimide intermediates.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as magnesium, ether solvents, and basic nickel salts ensures a stable and resilient supply chain, less susceptible to the geopolitical fluctuations that often impact rare earth or precious metal markets. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent choices, provides manufacturing flexibility, allowing production to continue even if specific solvent grades face temporary shortages, thereby securing continuity of supply for critical downstream applications.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in multi-hundred gram batches with consistent yields, and the absence of heavy metal zinc waste simplifies environmental permitting and compliance reporting. The use of ether solvents, while requiring careful handling, is well-established in industrial settings with existing recovery infrastructure, facilitating a smoother transition from pilot scale to commercial production without the need for entirely new waste treatment facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this catalytic system, focusing on catalyst stability, substrate scope, and purification requirements. These insights are derived directly from the experimental data and mechanistic understanding provided in the patent documentation, aimed at clarifying the operational parameters for potential licensees or manufacturing partners.
Q: What are the advantages of the hydroxy-imine spiro-nickel catalyst over traditional triphenylphosphine ligands?
A: The hydroxy-imine ligands possess lone pair electrons on the nitrogen and hydroxyl groups that facilitate easier leaving during the reaction cycle. This structural feature results in significantly higher catalytic activity and selectivity compared to traditional triphenylphosphine nickel complexes, while also being simpler to synthesize at room temperature.
Q: Can this method utilize chlorinated substrates effectively?
A: Yes, the patent demonstrates successful coupling using both 4-bromo-o-xylene and 4-chloro-o-xylene. While brominated substrates typically yield higher conversion rates (e.g., 73.3% vs 61.9% in examples), the novel catalyst system maintains sufficient activity to process the less reactive chlorinated derivatives under reflux conditions.
Q: How does this process improve environmental compliance compared to zinc-mediated methods?
A: Traditional methods require excessive amounts of zinc powder, generating substantial heavy metal waste that requires complex disposal protocols. The new Grignard-based route eliminates the need for stoichiometric zinc reduction, thereby drastically reducing solid waste generation and simplifying the downstream purification and environmental treatment processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetramethylbiphenyl Supplier
As the electronics and aerospace industries continue to push the boundaries of material performance, the need for high-purity intermediates like tetramethylbiphenyl has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced catalytic technologies to deliver superior chemical solutions. With extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, our team possesses the technical expertise to optimize this novel nickel-coupling route for maximum efficiency. Our rigorous QC labs and stringent purity specifications ensure that every batch meets the exacting standards required for high-performance polyimide synthesis.
We invite R&D directors and procurement leaders to collaborate with us to evaluate the feasibility of integrating this cost-effective synthesis method into your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis, specific COA data, and route feasibility assessments tailored to your volume requirements. Let us partner with you to secure a sustainable and economical source of critical electronic chemical intermediates.
