Revolutionizing Biphenyl Synthesis: A Cost-Effective Pd-Catalyzed Decarboxylative Coupling Strategy for Industrial Scale-Up
Revolutionizing Biphenyl Synthesis: A Cost-Effective Pd-Catalyzed Decarboxylative Coupling Strategy for Industrial Scale-Up
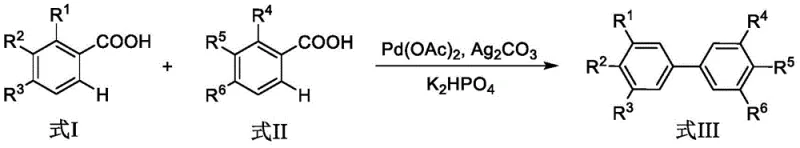
The landscape of organic synthesis for complex aromatic scaffolds is undergoing a significant transformation, driven by the urgent need for more sustainable and economically viable manufacturing processes. A pivotal advancement in this domain is detailed in patent CN107324964B, which discloses a highly efficient method for synthesizing biphenyl derivatives through a transition metal-catalyzed cross-dehydrogenation coupling and decarboxylation reaction. This innovative approach utilizes readily available aromatic carboxylic acids as direct coupling partners, bypassing the traditional requirement for pre-functionalized halogenated substrates. By leveraging palladium catalysis in conjunction with silver carbonate and phosphate bases, this technology enables the one-step construction of meta-substituted biphenyl cores with remarkable atom economy. For global procurement and R&D leaders, this represents a paradigm shift away from wasteful halogenation protocols towards a greener, more direct assembly of critical chemical building blocks used extensively in pharmaceuticals, agrochemicals, and functional materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biphenyl skeletons has relied heavily on organometallic-catalyzed cross-coupling reactions such as the Suzuki-Miyaura, Stille, Kumada, and Ullmann couplings. While these methods are robust, they suffer from inherent structural inefficiencies that impact both cost and environmental compliance in large-scale manufacturing. The most significant bottleneck is the prerequisite for pre-functionalized substrates, specifically aryl halides and organometallic reagents like boronic acids or organotin compounds. The synthesis of these starting materials often involves additional synthetic steps, including harsh halogenation processes that generate substantial quantities of corrosive and toxic halide waste. Furthermore, the use of organotin reagents in Stille couplings poses severe toxicity risks, requiring expensive removal protocols to meet stringent residual metal specifications for pharmaceutical intermediates. These multi-step sequences not only inflate the overall production cost but also extend lead times, creating supply chain vulnerabilities for high-volume pharmaceutical intermediate manufacturing.
The Novel Approach
In stark contrast, the methodology described in patent CN107324964B offers a streamlined alternative that directly utilizes aromatic carboxylic acids, which are abundant, stable, and inexpensive commodity chemicals. This decarboxylative cross-coupling strategy effectively merges the C-H activation and decarboxylation events into a single catalytic cycle, eliminating the need for prior halogenation of the aromatic ring. The reaction proceeds under relatively mild thermal conditions using ethylene glycol dimethyl ether as a solvent, with palladium acetate serving as the catalyst and silver carbonate acting as the oxidant. This direct approach significantly shortens the synthetic route, thereby reducing the cumulative yield losses associated with multi-step sequences. By avoiding the generation of stoichiometric halide byproducts, this process aligns perfectly with modern green chemistry principles, offering a compelling value proposition for companies seeking to reduce their environmental footprint while optimizing cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Pd-Catalyzed Cross-Dehydrogenation Coupling
The core of this technological breakthrough lies in the sophisticated interplay between the palladium catalyst and the carboxyl functionality on the aromatic ring. The reaction mechanism initiates with the coordination of the palladium species to the carboxyl group, facilitating a directed C-H activation at the ortho-position. This is followed by a decarboxylation event where carbon dioxide is extruded, generating a reactive aryl-palladium intermediate. Simultaneously, a second aromatic carboxylic acid molecule undergoes a similar activation process. The subsequent reductive elimination step forge the new carbon-carbon bond between the two aromatic rings, releasing the desired biphenyl derivative and regenerating the active palladium catalyst. The presence of silver carbonate is critical as it serves as a terminal oxidant to re-oxidize the palladium species, sustaining the catalytic cycle without the need for external oxidants that might degrade sensitive functional groups. This mechanistic pathway ensures high regioselectivity, predominantly yielding meta-substituted products which are often challenging to access via electrophilic aromatic substitution.
Understanding the impurity profile is paramount for R&D directors focused on purity specifications. The use of carboxylic acids minimizes the risk of homocoupling side reactions often seen with radical-based halogenation methods. However, the formation of symmetric biphenyl byproducts from the self-coupling of either starting acid is a potential pathway that must be managed. The patent data indicates that optimizing the molar ratios of the two different carboxylic acids, along with precise control of the silver carbonate and phosphate base loading, effectively suppresses these homocoupling impurities. The phosphate base plays a dual role, acting not only as a proton scavenger to facilitate the C-H activation step but also as a buffer to maintain the stability of the catalytic system. This fine-tuned balance allows for the synthesis of diverse derivatives, including those with electron-donating methyl and methoxy groups, as well as electron-withdrawing halogens, demonstrating the robustness of the catalytic system across a broad substrate scope.
How to Synthesize 3,3',5,5'-Tetramethylbiphenyl Efficiently
To illustrate the practical application of this technology, we examine the synthesis of 3,3',5,5'-tetramethylbiphenyl, a representative meta-substituted biphenyl derivative. The procedure involves charging a pressure reaction vessel with 2,4-dimethylbenzoic acid as the sole substrate for homocoupling or in combination with other substituted benzoic acids for cross-coupling. The reaction mixture includes palladium acetate (10-20 mol%), silver carbonate, and dipotassium hydrogen phosphate in ethylene glycol dimethyl ether. The system is sealed under an inert argon atmosphere to prevent oxidative degradation of the catalyst and heated to 150°C for 12 hours. Upon completion, the reaction mixture is cooled, and the crude product is isolated via filtration to remove inorganic salts and catalyst residues, followed by purification using silica gel column chromatography. This standardized protocol highlights the operational simplicity and reproducibility of the method.
- Mix aromatic carboxylic acids (Formula I and II) with palladium acetate catalyst, silver carbonate oxidant, and dipotassium hydrogen phosphate base in ethylene glycol dimethyl ether solvent.
- Maintain the reaction mixture under inert gas protection in a sealed pressure vessel at temperatures between 130°C and 160°C.
- Stir the reaction for 8 to 24 hours, then cool to room temperature and purify the resulting biphenyl derivative via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this decarboxylative coupling technology offers tangible strategic benefits beyond mere technical novelty. The primary driver for adoption is the substantial reduction in raw material costs. Aromatic carboxylic acids are produced on a massive industrial scale for various applications, making them significantly cheaper and more readily available than specialized aryl halides or organoboron reagents. This abundance ensures a stable supply chain, mitigating the risk of shortages that often plague niche halogenated intermediates. Furthermore, the elimination of halogenation steps simplifies the upstream supply chain, reducing the dependency on suppliers of hazardous halogenating agents and the associated logistics costs for handling dangerous goods.
- Cost Reduction in Manufacturing: The economic advantage of this process is derived from the convergence of several factors: lower raw material costs, reduced waste disposal fees, and shorter processing times. By removing the need for pre-functionalization, manufacturers save on the reagents, solvents, and energy required for those preliminary steps. Additionally, the avoidance of heavy metal contaminants like tin (from Stille reactions) reduces the cost of downstream purification and analytical testing. The overall process mass intensity (PMI) is improved, leading to a leaner manufacturing operation that delivers substantial cost savings without compromising on yield or quality.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like benzoic acids enhances supply security. Unlike complex organometallic reagents which may have limited suppliers and long lead times, aromatic acids are stocked by numerous global chemical distributors. This diversification of the supply base reduces the risk of production stoppages due to vendor issues. Moreover, the simplified workflow means fewer unit operations are required, which translates to faster turnaround times from order to delivery. This agility is crucial for responding to market demands for high-purity pharmaceutical intermediates in a just-in-time manufacturing environment.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this method is superior. It avoids the generation of large volumes of halide-containing wastewater, which is expensive to treat and subject to increasingly strict environmental regulations. The use of silver salts does require recovery protocols, but these are well-established in the industry. The reaction operates in a closed system under inert gas, minimizing VOC emissions. These factors make the process highly scalable, allowing for the commercial scale-up of complex biphenyl derivatives from pilot plant to multi-ton production with minimal regulatory friction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating its suitability for your specific project requirements. Understanding these details is essential for making informed decisions about process adoption and supplier qualification.
Q: What are the primary advantages of this decarboxylative coupling method over traditional Suzuki-Miyaura reactions?
A: This method eliminates the need for pre-halogenated substrates and organoboron reagents, significantly reducing raw material costs and avoiding the generation of hazardous halide waste streams associated with conventional cross-coupling protocols.
Q: Can this synthesis protocol be scaled for commercial production of pharmaceutical intermediates?
A: Yes, the process utilizes stable, commercially available aromatic carboxylic acids and standard transition metal catalysis, making it highly suitable for scale-up from laboratory to multi-ton annual production capacities with robust quality control.
Q: What represents the key regioselectivity outcome of this reaction?
A: The reaction specifically targets the ortho-position relative to the carboxyl group for decarboxylation and coupling, predominantly yielding meta-substituted biphenyl derivatives which are valuable scaffolds in medicinal chemistry.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biphenyl Derivatives Supplier
The technological potential of Pd-catalyzed decarboxylative coupling is immense, yet translating laboratory success into consistent commercial supply requires deep process engineering expertise. NINGBO INNO PHARMCHEM stands ready to bridge this gap as your trusted partner. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies demonstrated in patent CN107324964B are fully realized at an industrial level. Our facility is equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of biphenyl derivatives delivered meets the exacting standards required for API synthesis and advanced material applications.
We invite you to explore how this innovative synthesis route can optimize your supply chain and reduce your overall cost of goods. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your strategic sourcing goals for high-value chemical intermediates.
