Advanced One-Pot Synthesis of Pregnenolone Derivatives for Scalable Pharmaceutical Manufacturing
Introduction to Next-Generation Steroid Synthesis
The pharmaceutical industry is constantly seeking robust, environmentally sustainable, and cost-effective pathways for the production of critical hormonal intermediates. Patent CN102286052B introduces a transformative methodology for synthesizing pregnenone alcohol compounds, specifically targeting the efficient conversion of pseudosteroidal sapogenins. This innovation represents a significant departure from legacy processes that have long relied on hazardous reagents and energy-intensive conditions. By leveraging a novel "one-pot" protocol involving Oxone oxidation followed by alkali elimination hydrolysis, this technology addresses the dual challenges of environmental compliance and process efficiency. For R&D teams and supply chain leaders, understanding this shift is crucial, as it offers a viable route to high-purity steroidal intermediates while mitigating the ecological footprint associated with traditional steroid degradation. The method not only streamlines the synthetic sequence but also enhances the overall atom economy, making it a compelling candidate for modern API manufacturing pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of pregnenolone derivatives has been dominated by the Marker degradation method, a process established in the 1940s that, despite numerous modifications, remains fundamentally flawed for modern green chemistry standards. This conventional approach necessitates the use of chromic anhydride as a stoichiometric oxidant, introducing severe environmental liabilities due to the generation of toxic chromium waste streams that require expensive and complex remediation. Furthermore, the process typically involves high-temperature pyrolysis exceeding 200°C in acetic anhydride, which poses significant safety risks and energy costs. The harsh conditions often lead to side reactions and lower selectivity, complicating downstream purification and resulting in substantial material loss. For procurement and sustainability officers, the reliance on hexavalent chromium creates regulatory hurdles and increases the total cost of ownership due to waste disposal fees and the need for specialized equipment to handle corrosive and toxic reagents safely.
The Novel Approach
In stark contrast, the methodology disclosed in CN102286052B utilizes potassium peroxymonosulfate (Oxone) as a clean, non-metallic oxidant, operating under remarkably mild conditions that preserve the integrity of the sensitive steroid skeleton. The process initiates with the oxidation of pseudosteroidal sapogenins in an acetone-water system buffered by sodium bicarbonate, proceeding smoothly at temperatures ranging from -10°C to room temperature. This gentle oxidative step is seamlessly followed by a base-mediated elimination and hydrolysis in a tert-butanol medium, effectively completing the transformation in a single vessel without the need to isolate unstable intermediates. 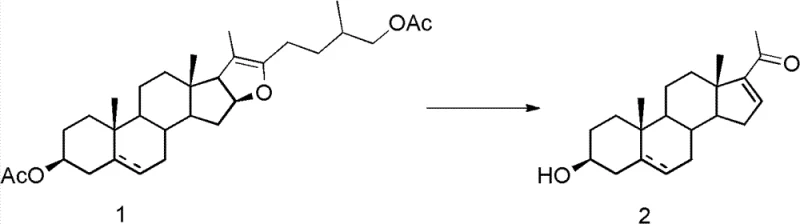 This "one-pot" design drastically reduces solvent usage and operational time, while the absence of heavy metals ensures that the final product meets stringent purity specifications required for pharmaceutical applications without extensive metal scavenging steps.
This "one-pot" design drastically reduces solvent usage and operational time, while the absence of heavy metals ensures that the final product meets stringent purity specifications required for pharmaceutical applications without extensive metal scavenging steps.
Mechanistic Insights into Oxone-Mediated Oxidative Cleavage
The core of this technological advancement lies in the selective oxidative cleavage of the spiroketal side chain of the pseudosteroidal sapogenin. Mechanistically, the reaction begins with the interaction of the Oxone reagent with the electron-rich centers of the substrate, facilitating the formation of a transient 20α-hydroxy steroidal intermediate. Unlike previous attempts that struggled with mixture formation, the precise control of reagent stoichiometry—specifically the molar ratios of substrate to Oxone and bicarbonate—allows for the preferential formation of this key intermediate. The subsequent addition of a strong inorganic base, such as potassium hydroxide or sodium hydroxide, triggers a rapid elimination reaction. This step involves the deprotonation of the hydroxyl group and the concurrent cleavage of the C22-C23 bond, ultimately yielding the desired Δ16-20-ketone structure characteristic of pregnenolone derivatives.  The ability to tune the reaction temperature during the elimination phase, typically between 50°C and reflux, provides an additional handle for optimizing conversion rates and minimizing degradation, ensuring a clean impurity profile that is critical for downstream drug synthesis.
The ability to tune the reaction temperature during the elimination phase, typically between 50°C and reflux, provides an additional handle for optimizing conversion rates and minimizing degradation, ensuring a clean impurity profile that is critical for downstream drug synthesis.
From an impurity control perspective, this mechanism offers distinct advantages over chromium-based oxidations. Traditional methods often generate a plethora of over-oxidized byproducts and chromium-complexed impurities that are notoriously difficult to separate from the lipophilic steroid backbone. In the Oxone-mediated pathway, the byproducts are primarily inorganic salts (potassium sulfate, sodium sulfate) which are easily removed during the aqueous workup. The mild nature of the oxidation prevents the degradation of the sensitive Δ5-double bond often present in the steroid A-ring, preserving the biological activity potential of the intermediate. Furthermore, the instability of the 20α-hydroxy intermediate towards acid is cleverly exploited; by avoiding acidic workups until the final product is formed, the process prevents premature decomposition, thereby maximizing the isolated yield of the target pregnenone compound.
How to Synthesize Pregnenolone Compounds Efficiently
Implementing this synthesis route requires careful attention to the sequential addition of reagents and temperature control to ensure optimal conversion. The process begins by dissolving the pseudosteroidal sapogenin starting material in acetone, followed by the controlled addition of sodium bicarbonate and water to create a buffered heterogeneous mixture. Oxone is then introduced gradually while maintaining the temperature between -10°C and room temperature to manage the exotherm and ensure selective oxidation. After the initial oxidation phase is complete, typically monitored by TLC, the solvent is removed, and the residue is taken up in tert-butanol and water for the elimination step.
- Oxidize the pseudosteroidal sapogenin substrate with Oxone and sodium bicarbonate in an acetone-water mixture at temperatures ranging from -10°C to room temperature.
- Remove the solvent and dissolve the residue in tert-butanol and water, then add an inorganic base such as potassium hydroxide or sodium hydroxide.
- Heat the reaction mixture to reflux or maintain at 50°C to 60°C to facilitate elimination and hydrolysis, followed by extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Oxone-based synthesis protocol presents a compelling value proposition centered on cost reduction and risk mitigation. The elimination of chromic anhydride removes the need for expensive heavy metal scavengers and the associated regulatory compliance costs related to hexavalent chromium handling and disposal. This shift not only lowers the direct material costs but also simplifies the waste management infrastructure required at the manufacturing site. Additionally, the use of commodity chemicals like acetone, tert-butanol, and Oxone ensures a stable and resilient supply chain, reducing the risk of production delays caused by the scarcity of specialized reagents. The streamlined "one-pot" nature of the process further contributes to operational efficiency by reducing the number of unit operations, thereby lowering labor costs and increasing throughput capacity without the need for significant capital investment in new reactor hardware.
- Cost Reduction in Manufacturing: The replacement of toxic chromium reagents with benign Oxone leads to substantial cost savings by eliminating the complex and costly downstream processing steps required to remove trace metals to ppm levels. The simplified workup procedure, which relies on basic extraction and crystallization rather than chromatography or specialized filtration, significantly reduces solvent consumption and processing time. Furthermore, the higher selectivity of the reaction minimizes the formation of hard-to-remove impurities, leading to improved overall yields and reduced raw material waste, which directly impacts the cost of goods sold (COGS) for the final API intermediate.
- Enhanced Supply Chain Reliability: By utilizing widely available and non-restricted reagents, this method decouples production from the volatile supply chains often associated with specialized oxidants or heavy metals. The robustness of the reaction conditions, which tolerate a range of temperatures and do not require high-pressure equipment, enhances the reliability of manufacturing schedules. This stability is crucial for maintaining continuous supply to downstream customers, ensuring that production targets are met consistently without the interruptions frequently caused by equipment maintenance or reagent shortages inherent in more aggressive chemical processes.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively in multi-gram batches with consistent results, indicating a smooth path to ton-scale production. The significant reduction in hazardous waste generation aligns with increasingly stringent global environmental regulations, future-proofing the manufacturing site against tightening emission standards. This eco-friendly profile not only reduces the environmental tax burden but also enhances the corporate sustainability metrics of the organization, making it a preferred partner for multinational pharmaceutical companies committed to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent literature, providing a clear understanding of the process capabilities and limitations.
Q: How does this Oxone-based method improve upon the traditional Marker degradation process?
A: Unlike the traditional Marker degradation which relies on toxic chromic anhydride and harsh high-temperature pyrolysis, this method utilizes Oxone under mild conditions. This eliminates heavy metal contamination, significantly reduces environmental pollution, and simplifies the purification process by avoiding complex chromium removal steps.
Q: What are the key advantages of the 'one-pot' strategy described in patent CN102286052B?
A: The one-pot strategy allows for the direct conversion of the intermediate without isolation. Since the intermediate 20α-hydroxy compound is acid-sensitive and prone to decomposition, avoiding its isolation prevents yield loss and simplifies the operational workflow, leading to higher overall efficiency and reduced solvent consumption.
Q: Is this synthesis method suitable for large-scale industrial production of steroid intermediates?
A: Yes, the method is highly suitable for scale-up. It uses inexpensive and readily available reagents like Oxone and common solvents like acetone and tert-butanol. The mild reaction conditions (-10°C to reflux) and the absence of high-pressure or extreme temperature requirements make it safer and more cost-effective for commercial manufacturing compared to legacy technologies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pregnenolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic routes to maintain competitiveness in the global pharmaceutical market. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity steroidal intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our facility is designed to handle complex organic syntheses with a focus on safety, quality, and environmental stewardship, making us an ideal partner for your long-term supply needs.
We invite you to engage with our technical procurement team to discuss how this Oxone-mediated synthesis can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume needs. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to validate the quality and viability of our manufacturing capabilities before committing to large-scale orders. Let us collaborate to drive efficiency and sustainability in your steroid supply chain.
