Optimizing Etelcalcetide Production: A Deep Dive into Advanced Solid-Phase Synthesis and Commercial Scalability
The pharmaceutical landscape for treating secondary hyperparathyroidism in dialysis patients has been significantly advanced by the development of calcimimetics like Etelcalcetide. As a potent activator of the calcium-sensing receptor on the parathyroid gland, this peptide therapeutic requires a manufacturing process that guarantees exceptional purity and structural fidelity, particularly regarding its critical disulfide bridges. Recent intellectual property developments, specifically patent CN110054662B, disclose a robust solid-phase synthesis method that addresses historical challenges in yield and impurity profiles. This technical insight report analyzes the mechanistic advantages of this novel route, offering a compelling value proposition for R&D directors and procurement strategists seeking a reliable Etelcalcetide supplier. By shifting from traditional post-synthesis modification to a pre-protected coupling strategy, the industry can achieve substantial improvements in process efficiency.
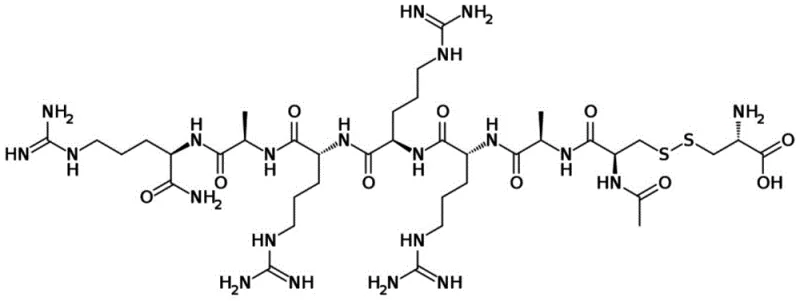
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex peptides containing disulfide bonds, such as Etelcalcetide, has been plagued by inefficiencies inherent in post-synthetic modification strategies. Conventional methods, including those cited in prior art like CN201511029990.2 and CN201511030047.3, typically involve synthesizing the linear peptide chain first and then attempting to form the disulfide bond through oxidation in the liquid phase or on the solid support. This approach introduces significant variability; the oxidation step is notoriously difficult to control, often leading to a heterogeneous mixture of products including incorrect disulfide pairings, oligomers, and over-oxidized species. Furthermore, liquid-phase synthesis of the main chain often suffers from solubility issues as the peptide length increases, resulting in aggregation and incomplete couplings that drastically reduce the total yield. These technical bottlenecks translate directly into higher production costs and extended timelines for cost reduction in peptide manufacturing, making the final API prohibitively expensive for broader clinical application.
The Novel Approach
The methodology outlined in patent CN110054662B represents a paradigm shift by introducing a fully protected D-cysteine derivative, denoted as Ac-D-Cys[R1-Cys-OR2], as a distinct building block. Instead of struggling with oxidation after the chain is complete, this strategy couples the protected cysteine unit directly to the N-terminus of the resin-bound peptide sequence (positions 2-7). This ensures that the sulfur atoms are already in the correct proximity and protection state before the final cleavage. The structural precision of this intermediate is critical, as shown in the detailed chemical representation below. By integrating this specialized building block, the synthesis bypasses the stochastic nature of random oxidation, leading to a crude product purity exceeding 92%. This approach not only simplifies the workflow but also significantly enhances the reproducibility required for commercial scale-up of complex peptides.
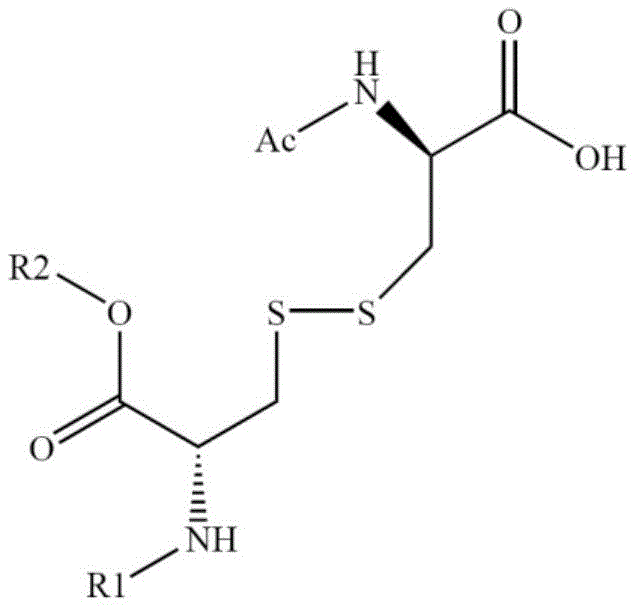
Mechanistic Insights into Solid-Phase Peptide Assembly and Acidolysis
The core of this synthetic success lies in the meticulous orchestration of solid-phase peptide synthesis (SPPS) coupled with a highly optimized cleavage protocol. The process initiates with the loading of Fmoc-D-Arg(Pbf)-OH onto a Rink Amide MBHA resin, establishing the C-terminus. Subsequent amino acids—D-Ala and D-Arg—are added sequentially using potent condensation reagents like DIC/HOBt or HATU/DIPEA. The critical step involves the coupling of the Ac-D-Cys derivative where R1 is typically Boc or Fmoc, and R2 is All or tBu. This specific protection scheme is designed to be orthogonal, meaning the side-chain protecting groups remain stable during the repetitive Fmoc deprotection cycles (using 20-25% piperidine in DMF) but are readily removed during the final acidic cleavage. The use of EDT (1,2-ethanedithiol) in the cleavage cocktail is particularly noteworthy; it acts as a potent scavenger for carbocations generated during acidolysis, preventing alkylation of the electron-rich sulfur atoms which is a common source of impurity in cysteine-rich peptides.
Following the assembly, the global deprotection and cleavage are performed using a tailored mixture of TFA, EDT, and water (optimally 90:5:5 v/v/v). This specific ratio is crucial for balancing the rate of cleavage with the suppression of side reactions. The resulting crude peptide is then subjected to a rigorous two-stage purification process. First, preparative HPLC using a C18 reverse-phase column separates the target peptide from deletion sequences and truncated fragments. Second, a salt exchange step converts the TFA salt into the more pharmaceutically acceptable acetate or hydrochloride form. This dual-purification strategy is what enables the final product to achieve a purity greater than 99.5% with single impurities controlled below 0.1%, meeting the stringent specifications required for injectable high-purity Etelcalcetide.
How to Synthesize Etelcalcetide Efficiently
The synthesis of Etelcalcetide via this protected cysteine coupling method offers a streamlined pathway that minimizes unit operations while maximizing yield. The process relies on standard Fmoc-SPPS equipment but demands precise control over the coupling of the bulky cysteine derivative and the composition of the cleavage cocktail. Operators must ensure that the resin swelling and washing steps are thorough to prevent reagent trapping, which can lead to racemization. The following guide outlines the critical operational phases derived from the patent examples, focusing on the transition from resin loading to the final lyophilized product. For detailed standard operating procedures and specific molar ratios, please refer to the technical guidelines below.
- Synthesize the peptide resin sequence (positions 2-7) on Rink Amide resin using standard Fmoc chemistry.
- Couple the fully protected Ac-D-Cys[R1-Cys-OR2] derivative to the N-terminus of the resin-bound peptide.
- Perform global acidolysis using a TFA/EDT/Water mixture, followed by HPLC purification and salt exchange to obtain high-purity Etelcalcetide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage is the drastic simplification of the downstream processing. By eliminating the need for a separate, difficult-to-control oxidation step, the manufacturing timeline is significantly compressed, and the consumption of specialized oxidizing reagents is reduced. This streamlining directly contributes to cost reduction in peptide manufacturing by lowering both material costs and labor hours associated with monitoring complex reaction endpoints. Furthermore, the high crude purity (>90%) means that the load on the purification columns is reduced, extending the life of expensive chromatography media and increasing the throughput of the purification suite. This efficiency is vital for maintaining a competitive cost structure in the biosimilars and generic peptide market.
- Cost Reduction in Manufacturing: The elimination of post-synthesis oxidation steps removes a major source of yield loss and reagent cost. Traditional methods often require multiple rounds of oxidation and purification to achieve acceptable disulfide bond formation, whereas this method builds the connectivity directly into the synthesis. Additionally, the high total yield of approximately 70% reported in the patent examples implies that less starting material is wasted, optimizing the cost of goods sold (COGS). The use of standard Fmoc amino acids and widely available resins further ensures that raw material costs remain stable and predictable, avoiding the price volatility associated with exotic catalysts or proprietary reagents.
- Enhanced Supply Chain Reliability: The robustness of this solid-phase method enhances supply security by reducing the risk of batch failures. In conventional liquid-phase or post-oxidation routes, minor variations in temperature or pH can lead to catastrophic batch rejection due to incorrect disulfide pairing. The pre-protected strategy mitigates this risk, ensuring consistent batch-to-batch quality. Moreover, the reliance on commodity chemicals like TFA, DIC, and standard resins means that the supply chain is not dependent on single-source suppliers for critical reagents. This diversification of the supply base is crucial for reducing lead time for high-purity API intermediates and ensuring uninterrupted production schedules even during global supply chain disruptions.
- Scalability and Environmental Compliance: Scaling solid-phase synthesis is inherently challenging due to solvent volumes, but this method's high efficiency reduces the overall solvent burden per kilogram of product. The high yield means fewer batches are needed to meet demand, thereby reducing the total volume of hazardous waste generated. The acidolysis step uses TFA, which is recoverable and recyclable in modern facilities, aligning with green chemistry principles. The ability to scale from gram-scale R&D to multi-kilogram commercial production without changing the fundamental chemistry provides a clear path for capacity expansion. This scalability ensures that manufacturers can respond rapidly to market demand surges without compromising on environmental standards or regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Etelcalcetide using this advanced solid-phase methodology. These insights are derived directly from the experimental data and claims within patent CN110054662B, providing a factual basis for decision-making. Understanding these nuances is essential for partners evaluating the feasibility of integrating this molecule into their portfolio. The answers reflect the balance between theoretical chemistry and practical manufacturing constraints.
Q: What is the primary advantage of using the fully protected Ac-D-Cys derivative in Etelcalcetide synthesis?
A: Using the fully protected Ac-D-Cys[R1-Cys-OR2] derivative allows for the direct formation of the disulfide bond precursor on the resin, avoiding complex post-synthesis oxidation steps that often lead to impurities and lower yields.
Q: What purity levels can be achieved with this solid-phase synthesis method?
A: The method described in patent CN110054662B achieves a crude product purity of over 92% and a final purified product purity exceeding 99.5%, with single impurities controlled below 0.1%.
Q: How does the acidolysis step impact the final quality of the peptide?
A: The use of a specific mixed acidolysis solution containing TFA, EDT, and water effectively cleaves the peptide from the resin while scavenging reactive cations, preventing side reactions on the sulfur atoms and ensuring high structural integrity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Etelcalcetide Supplier
The technical sophistication required to execute this synthesis underscores the need for a manufacturing partner with deep expertise in peptide chemistry. NINGBO INNO PHARMCHEM stands at the forefront of this field, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art solid-phase synthesizers and preparative HPLC systems capable of handling the rigorous purification demands of Etelcalcetide. We adhere to stringent purity specifications and operate rigorous QC labs to ensure that every batch meets the >99.5% purity benchmark required for clinical and commercial use. Our commitment to quality assurance ensures that the complex disulfide architecture of Etelcalcetide is preserved throughout the manufacturing process.
We invite potential partners to engage with our technical team to explore how this optimized synthesis route can benefit your supply chain. By leveraging our process expertise, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can accelerate the availability of this critical therapy for patients with chronic kidney disease, ensuring a stable and high-quality supply of this vital pharmaceutical intermediate.
