Advanced Terlipressin Manufacturing: Leveraging Novel MOBHA Resin for Superior Yield and Purity
Advanced Terlipressin Manufacturing: Leveraging Novel MOBHA Resin for Superior Yield and Purity
The pharmaceutical landscape for vasoactive peptides is undergoing a significant transformation driven by the innovations detailed in patent CN107778353B. This pivotal intellectual property introduces a groundbreaking methodology for the synthesis of Terlipressin, a critical therapeutic agent used in the management of esophageal variceal bleeding. Unlike traditional approaches that rely on costly and sometimes inefficient resin systems, this invention deploys a brand-new solid phase carrier known as MOBHA resin. The technical breakthrough lies not only in the chemical structure of the resin itself but also in the optimized timing of intramolecular cyclization, which collectively drives the total yield of the pure product to exceed 75%. This represents a substantial leap forward from the industry standard, offering a robust pathway for manufacturers seeking to enhance supply chain reliability and reduce production costs without compromising on the stringent purity specifications required for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Terlipressin has been plagued by inefficiencies inherent to the use of conventional Rink Amide series resins. Prior art, such as that disclosed in patent CN105367627A, typically yields a total recovery of merely 50% to 55% after purification, a figure that severely impacts the economic viability of large-scale production. The reliance on these older resin technologies often necessitates harsh acidolysis conditions and complex scavenger systems, such as high concentrations of EDT in TFA solutions, which can introduce difficult-to-remove impurities into the crude product. Furthermore, the theoretical yield calculations in existing patents often fail to align with practical outcomes due to the presence of protective agents and scavengers that inflate the mass of the crude intermediate, leading to significant discrepancies between expected and actual output. These limitations create a bottleneck for procurement teams, as the low yield translates directly into higher raw material consumption and increased waste disposal costs, thereby inflating the overall cost of goods sold for this essential API.
The Novel Approach
In stark contrast to these legacy methods, the novel approach utilizing MOBHA resin fundamentally restructures the synthesis workflow to maximize efficiency and minimize waste. The invention strategically modifies the linker structure attached to the polystyrene backbone, creating a carrier that is not only more cost-effective than Rink Amide resins but also chemically superior for this specific peptide sequence. By adjusting the parameters of the intramolecular cyclization step and employing a streamlined acidolysis protocol using hydrogen bromide in trifluoroacetic acid, the process achieves a total yield of the pure product that surpasses 75%. This improvement is not marginal; it represents a paradigm shift in process chemistry that allows for a drastic reduction in the amount of starting materials required per kilogram of final API. The simplicity of the operation, characterized by mild reaction conditions and straightforward washing procedures, ensures that the method is highly amenable to industrial scale-up, providing a competitive edge for manufacturers aiming to secure a stable and economical supply of high-purity Terlipressin.
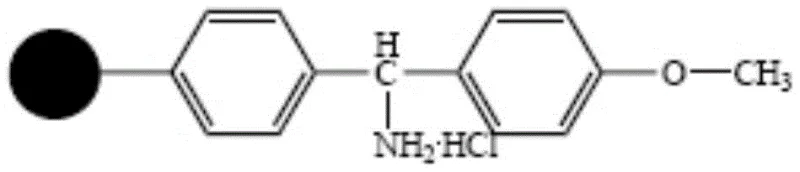
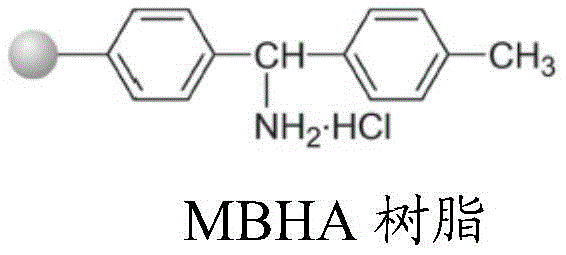
Mechanistic Insights into MOBHA-Mediated Solid Phase Peptide Synthesis
The core of this technological advancement lies in the precise orchestration of solid-phase peptide synthesis (SPPS) mechanics using the specialized MOBHA resin. The synthesis initiates with the sequential coupling of protected amino acids—specifically Glycine, Lysine, Proline, Cysteine, Asparagine, Glutamine, Phenylalanine, and Tyrosine—from the C-terminus to the N-terminus. The use of condensation reagents such as DIC (N,N-Diisopropylcarbodiimide) paired with activation reagents like HOBt ensures high coupling efficiency, while the specific substitution value of the resin (optimized between 0.5 to 1.0 mmol/g) prevents steric hindrance that often plagues long peptide chains. Crucially, the protecting group strategy employs Fmoc or Boc for the N-terminus and specific side-chain protectors like Trt for Cysteine and tBu for Tyrosine, which are orthogonal to the final cleavage conditions. This meticulous selection prevents premature deprotection and racemization, ensuring that the linear peptide backbone is assembled with high fidelity before the critical cyclization step occurs.
Following the assembly of the linear peptide chain, the process employs a sophisticated iodine oxidation method to facilitate intramolecular cyclization, forming the essential disulfide bond between the two Cysteine residues at positions 4 and 9. This step is performed directly on the resin using a 5% I2/DMF solution, a condition that is mild enough to preserve the integrity of other sensitive functional groups while being potent enough to drive the cyclization to completion. The subsequent acidolysis step utilizes a hydrogen bromide/trifluoroacetic acid solution, which effectively cleaves the peptide from the MOBHA resin while simultaneously removing acid-labile protecting groups. This dual-action cleavage mechanism minimizes the formation of by-products and simplifies the downstream purification process. The result is a crude product with a purity exceeding 85%, which significantly reduces the burden on the preparative HPLC purification stage, ultimately yielding a final product with a purity greater than 99.5% and a maximum single impurity of less than 0.12%.
How to Synthesize Terlipressin Efficiently
The synthesis of Terlipressin via this patented route requires strict adherence to the optimized coupling and cyclization parameters to achieve the reported high yields. The process begins with the swelling of the MOBHA resin in DMF, followed by the iterative addition of activated protected amino acids. Each coupling cycle involves activation with DIC and HOBt, followed by thorough washing to remove excess reagents, ensuring that deletion sequences are minimized. The critical cyclization step must be monitored carefully to ensure complete disulfide bond formation without over-oxidation. For a comprehensive breakdown of the specific molar ratios, reaction times, and temperature controls required for each step of this synthesis, please refer to the standardized protocol below.
- Couple protected amino acids sequentially from C-terminus to N-terminus onto the MOBHA resin using condensation reagents like DIC/HOBt.
- Perform intramolecular cyclization of the peptide resin using an iodine oxidation method to form the critical disulfide bond.
- Execute acidolysis using HBr/TFA solution to cleave the peptide from the resin, followed by HPLC purification and salt conversion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this MOBHA resin-based synthesis method offers profound strategic advantages that extend far beyond simple chemical yield improvements. The transition from expensive Rink Amide resins to the more economical MOBHA variant directly addresses the escalating costs associated with peptide API manufacturing. By eliminating the need for premium-priced carriers and reducing the stoichiometric excess of amino acids required to drive reactions to completion, the overall material cost per batch is significantly reduced. Furthermore, the simplified operational workflow, which avoids complex scavenger mixtures and harsh conditions, lowers the barrier for entry for contract manufacturing organizations, thereby increasing the pool of potential suppliers and enhancing supply chain resilience against disruptions.
- Cost Reduction in Manufacturing: The implementation of this novel resin system eliminates the dependency on high-cost Rink Amide series resins, which are traditionally a major cost driver in peptide synthesis. By utilizing a carrier that is chemically optimized for Terlipressin, the process achieves a much higher conversion rate of raw materials into the final active ingredient, effectively spreading the fixed costs of production over a larger output volume. Additionally, the reduction in impurity formation during the acidolysis step means that less solvent and stationary phase are consumed during the purification stage, leading to substantial savings in chromatography consumables and waste treatment expenses. This holistic reduction in variable costs allows for a more competitive pricing structure without sacrificing margin.
- Enhanced Supply Chain Reliability: The robustness of the MOBHA resin synthesis method ensures a more predictable and consistent production schedule, which is critical for maintaining uninterrupted supply to downstream pharmaceutical formulators. The mild reaction conditions and tolerance to standard laboratory equipment mean that the process can be easily replicated across different manufacturing sites, reducing the risk of technology transfer failures. Moreover, the high crude purity achieved prior to purification reduces the likelihood of batch failures due to out-of-specification impurity profiles, thereby stabilizing the lead time for finished goods. This reliability is paramount for securing long-term supply agreements and mitigating the risks associated with single-source dependencies in the global API market.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method offers a cleaner and more sustainable manufacturing profile. The use of DMF as a primary solvent is standard, but the reduction in the number of purification cycles and the elimination of heavy metal catalysts or exotic scavengers simplifies the effluent treatment process. The high yield means that less waste is generated per kilogram of product, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The process is designed for seamless scale-up from pilot batches to multi-ton commercial production, ensuring that the supply can grow in tandem with market demand for Terlipressin without requiring prohibitive capital investment in new reactor infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Terlipressin using the MOBHA resin methodology. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing pipelines or for procurement specialists assessing the quality credentials of potential suppliers.
Q: What is the primary advantage of MOBHA resin over Rink Amide resin in Terlipressin synthesis?
A: The MOBHA resin offers a significantly lower cost profile compared to the conventional Rink Amide series while simultaneously improving the total yield of the pure product to over 75%, addressing both economic and efficiency bottlenecks.
Q: How does the novel method control impurities during the cyclization step?
A: The process utilizes a controlled iodine oxidation method for intramolecular cyclization, which effectively connects the two Cysteine residues via a disulfide bond while minimizing side reactions, resulting in a crude product purity exceeding 85%.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method features mild reaction conditions and simple operational steps, such as standard DMF washing and straightforward acidolysis, making it highly scalable for commercial manufacturing of complex peptide APIs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Terlipressin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies like the MOBHA resin method to meet the growing global demand for high-quality Terlipressin. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of this vital API. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the low impurity profiles achievable through this novel synthesis route. We are committed to leveraging our technical expertise to optimize every step of the manufacturing process, from resin loading to final lyophilization, guaranteeing a product that meets the highest international pharmacopeial standards.
We invite pharmaceutical companies and research institutions to collaborate with us to explore the full potential of this cost-effective and high-yield synthesis method. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how the switch to this advanced manufacturing route can impact your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to secure a sustainable and economical supply chain for Terlipressin and other complex peptide therapeutics.
