Advanced Convergent Synthesis of Paricalcitol: Overcoming Traditional Vitamin D2 Limitations for Commercial Scale
The pharmaceutical industry continuously seeks robust synthetic routes for active pharmaceutical ingredients (APIs) that balance high purity with economic viability. Patent CN103086937A introduces a transformative methodology for the synthesis of Paricalcitol, widely known by its brand name Zemplar, which is critical for treating secondary hyperparathyroidism in dialysis patients. Unlike conventional linear modifications of the Vitamin D2 nucleus, this invention proposes a convergent synthetic strategy that fundamentally alters the manufacturing landscape. By utilizing a Julia olefination reaction to couple two distinct molecular fragments, the process circumvents the historical reliance on prohibitively expensive 25-hydroxyvitamin D2 starting materials. This technical breakthrough not only streamlines the carbon skeleton construction but also ensures that each intermediate exists as a single, well-defined substance rather than a complex mixture of diastereomers. For R&D directors and process chemists, this represents a significant leap forward in impurity profile control and structural verification, laying a solid foundation for reliable high-purity pharmaceutical intermediates supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Paricalcitol has been plagued by significant economic and technical bottlenecks inherent to semi-synthetic approaches derived directly from Vitamin D2. The primary constraint lies in the sourcing of the raw material; specifically, the requirement for 25-hydroxyvitamin D2 creates a severe cost burden due to its scarcity and complex isolation from natural sources or multi-step enzymatic processes. Furthermore, the chemical modification of the Vitamin D2 parent nucleus often involves harsh conditions that lack regioselectivity, leading to the formation of multiple diastereomeric byproducts. These stereoisomers possess physical properties remarkably similar to the target molecule, making their separation via standard chromatographic techniques extremely difficult and yield-destructive. Consequently, the final evaluation of compound purity becomes an arduous task, often requiring repetitive recrystallization or preparative HPLC, which drastically inflates the cost of goods sold (COGS) and extends production lead times. This inefficiency renders many traditional routes unsuitable for the rigorous demands of modern commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN103086937A employs a convergent strategy that decouples the synthesis of the side chain from the CD-ring system until the final stages. This approach utilizes two relatively simple, structurally defined small molecules—Compound I (a ketone fragment) and Compound II (a sulfone fragment)—which are readily available and easy to prepare in large quantities. The core innovation is the application of the Julia reaction to splice these fragments together, forming the critical exocyclic double bond with high stereocontrol. Because the starting fragments are achiral or easily resolved prior to coupling, the resulting intermediate (Compound III) is obtained as a single substance, eliminating the diastereomer nightmare associated with direct Vitamin D2 modification. This clarity in molecular identity facilitates straightforward structural identification using standard NMR and MS techniques. Moreover, the subsequent deprotection steps utilize mild fluoride reagents and acidic hydrolysis, avoiding the use of toxic heavy metals or extreme thermal conditions, thereby enhancing the overall safety and environmental profile of the manufacturing process.
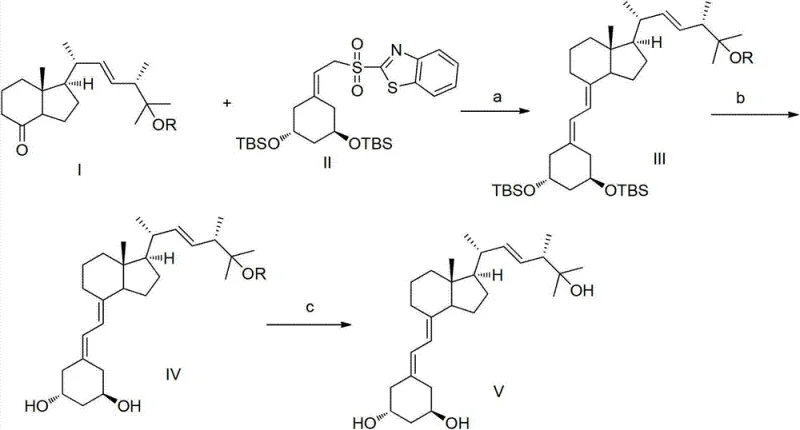
Mechanistic Insights into Julia Olefination and Deprotection
The heart of this synthetic route is the Julia olefination, a powerful carbon-carbon bond-forming reaction that connects the A-ring fragment (Compound II) with the CD-ring side chain precursor (Compound I). Mechanistically, this process begins with the deprotonation of the sulfone moiety in Compound II using a strong, non-nucleophilic base such as Lithium Diisopropylamide (LDA) or Lithium Hexamethyldisilazide (LHMDS). This generates a reactive carbanion species which then nucleophilically attacks the carbonyl carbon of the ketone in Compound I. The reaction is conducted at cryogenic temperatures ranging from -60°C to -40°C in solvents like tetrahydrofuran (THF) or diethyl ether to suppress side reactions and control the stereochemistry of the initial adduct. Following the addition, the intermediate beta-hydroxy sulfone undergoes elimination, typically facilitated by warming to room temperature or through specific activation, to forge the trans-alkene linkage characteristic of the Paricalcitol structure. The precision of this step is paramount, as it establishes the geometry of the triene system essential for biological activity.
Following the coupling, the synthesis proceeds through a carefully orchestrated deprotection sequence to reveal the final pharmacophore. The first deprotection stage targets the silyl protecting groups (specifically tert-butyldimethylsilyl or TBS) located on the A-ring hydroxyls. This is achieved using fluorine-containing reagents such as tetrabutylammonium fluoride (TBAF) or potassium fluoride (KF) at elevated temperatures between 50°C and 70°C. The silicon-fluorine bond formation provides a strong thermodynamic driving force for this cleavage, yielding Compound IV with high efficiency. The final step involves the removal of the ether-based protecting group (R group, such as methoxymethyl or MOM) at the C25 position. This is accomplished under acidic conditions using reagents like hydrochloric acid, sulfuric acid, or p-toluenesulfonic acid in alcoholic solvents. The acid-catalyzed hydrolysis cleaves the acetal linkage without disturbing the sensitive triene system, delivering the final Paricalcitol product with the requisite 1-alpha, 3-beta, 25-trihydroxy configuration intact.
How to Synthesize Paricalcitol Efficiently
The operational simplicity of this three-step sequence makes it highly attractive for process development teams aiming to establish a robust manufacturing protocol. The procedure begins with the preparation of the sulfone anion under inert atmosphere, followed by the controlled addition of the ketone fragment to manage exotherms and ensure selectivity. After the Julia coupling is complete, standard aqueous workup and silica gel chromatography provide the coupled intermediate in high purity. The subsequent fluoride-mediated desilylation is equally straightforward, utilizing common laboratory reagents that are easily sourced at scale. Finally, the acidic hydrolysis step requires careful pH monitoring to ensure complete deprotection while minimizing degradation of the vitamin D analog. For a detailed breakdown of the specific molar ratios, solvent volumes, and purification parameters described in the patent examples, please refer to the standardized synthesis guide below.
- Perform Julia reaction between sulfone Compound II and ketone Compound I using strong bases like LDA or LHMDS at -60°C to -40°C to generate intermediate III.
- React Compound III with fluorine-containing reagents such as TBAF or KF at 50°C to 70°C to remove silyl protecting groups and obtain Compound IV.
- Hydrolyze Compound IV under acidic conditions (e.g., HCl, Tosic acid) at 0°C to 70°C to remove the R protecting group and yield final Paricalcitol.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this convergent synthesis route offers substantial strategic advantages over legacy Vitamin D2 modification pathways. The most immediate impact is seen in raw material sourcing; by shifting away from the bottleneck of 25-hydroxyvitamin D2, manufacturers can utilize commodity chemicals and simpler intermediates that are subject to less market volatility. This diversification of the supply base significantly enhances supply chain reliability and reduces the risk of production stoppages due to raw material shortages. Furthermore, the ability to produce single-substance intermediates simplifies quality control (QC) workflows. When intermediates are not complex mixtures, analytical testing becomes faster and more accurate, reducing the time batches spend in quarantine and accelerating the release of finished goods. This efficiency translates directly into improved cash flow and the ability to respond more agilely to market demand fluctuations.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive starting materials and the reduction of purification complexity. Traditional routes often suffer from low overall yields due to the loss of material during the separation of diastereomers; in contrast, this novel method delivers high yields at each step because the intermediates are chemically homogeneous. Additionally, the avoidance of precious metal catalysts or exotic reagents lowers the direct material costs. The simplified workup procedures, which rely on standard extraction and chromatography rather than specialized preparative techniques, further reduce operational expenditures related to solvent consumption and labor hours. These factors combine to create a significantly lower cost base for cost reduction in pharmaceutical intermediates manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, stable intermediates that can be stockpiled if necessary. Compound I and Compound II are structurally simple molecules that do not suffer from the light and heat sensitivity typical of fully formed vitamin D analogs, allowing for more flexible inventory management. The modular nature of the convergent synthesis also means that the two fragments can be produced in parallel at different facilities if needed, decoupling the production timeline and reducing the critical path duration. This flexibility ensures reducing lead time for high-purity pharmaceutical intermediates and provides a buffer against logistical disruptions, ensuring a continuous flow of material to downstream formulation partners.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates. The reaction conditions, while requiring temperature control, do not demand extreme pressures or specialized reactor configurations that are difficult to replicate at the tonnage scale. The use of common solvents like THF, ethanol, and ethyl acetate simplifies solvent recovery and recycling systems, aligning with green chemistry principles. Moreover, the absence of heavy metal residues eliminates the need for costly and time-consuming metal scavenging steps, which are often a regulatory hurdle in API production. This streamlined profile facilitates easier regulatory filing and approval, accelerating the time to market for generic versions of Paricalcitol.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. They are derived from the specific experimental data and beneficial effects outlined in the patent documentation, providing clarity on the practical aspects of adopting this technology. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of the supply source.
Q: Why is this convergent synthesis superior to traditional Vitamin D2 modification?
A: Traditional methods rely on expensive 25-OH Vitamin D2 and often produce difficult-to-separate diastereomer mixtures. This new route uses simple, commercially available fragments that yield single-substance intermediates, significantly simplifying purification and structural identification.
Q: What are the critical reaction conditions for the Julia coupling step?
A: The Julia reaction requires precise low-temperature control between -60°C and -40°C using strong non-nucleophilic bases like Lithium Hexamethyldisilazide (LHMDS) or LDA in solvents such as THF or ether to ensure high stereoselectivity and yield.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for industrial suitability. It avoids expensive transition metal catalysts, utilizes standard workup procedures like column chromatography and extraction, and employs stable intermediates that are easy to handle and store.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paricalcitol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the competitive landscape of vitamin D analogs. Our technical team has thoroughly analyzed the convergent strategy presented in CN103086937A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry to life. We are committed to delivering stringent purity specifications through our rigorous QC labs, ensuring that every batch of Paricalcitol intermediate meets the highest global regulatory standards. Our facility is equipped to handle the low-temperature Julia coupling and the subsequent deprotection steps with precision, guaranteeing consistent quality and batch-to-batch reproducibility.
We invite pharmaceutical partners to collaborate with us to leverage this advanced manufacturing capability. By optimizing this route further, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can secure a sustainable and cost-effective supply chain for this vital therapeutic agent, ensuring patient access while maximizing value for your organization.
