Advanced Tetracene Derivative Synthesis for High-Mobility OFET Applications and Commercial Scale-Up
The landscape of organic electronics is undergoing a significant transformation driven by the demand for flexible, low-cost, and high-performance semiconductor materials. Patent CN102659752A introduces a groundbreaking class of naphthacene derivative field-effect transistor (OFET) materials that address the critical limitations of traditional acenes. These novel compounds, defined by the general formula (I), represent a strategic evolution in molecular design, combining the high charge carrier mobility of fused ring systems with enhanced processability. 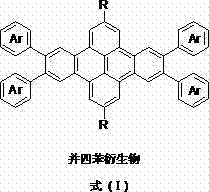 The core innovation lies in the specific substitution of the tetracene backbone with aryl groups and flexible alkyl chains, which fundamentally alters the material's physical properties. This structural modification not only improves solubility in common organic solvents but also enhances environmental stability, a longstanding challenge for p-type organic semiconductors. By leveraging a robust synthetic pathway involving Sonogashira coupling and Bergman cyclization, this technology offers a viable route for the commercial scale-up of complex organic semiconductors. For R&D directors and procurement specialists in the electronic chemical sector, this patent signals a shift towards more manufacturable and reliable OFET materials.
The core innovation lies in the specific substitution of the tetracene backbone with aryl groups and flexible alkyl chains, which fundamentally alters the material's physical properties. This structural modification not only improves solubility in common organic solvents but also enhances environmental stability, a longstanding challenge for p-type organic semiconductors. By leveraging a robust synthetic pathway involving Sonogashira coupling and Bergman cyclization, this technology offers a viable route for the commercial scale-up of complex organic semiconductors. For R&D directors and procurement specialists in the electronic chemical sector, this patent signals a shift towards more manufacturable and reliable OFET materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional organic semiconductor materials, particularly pentacene, have long been the benchmark for high mobility but suffer from severe practical drawbacks that hinder their widespread commercial adoption. Pentacene is notoriously insoluble in most common organic solvents, which restricts its fabrication almost exclusively to physical vapor deposition (PVD) techniques. This reliance on vacuum-based processes necessitates expensive equipment and limits the ability to manufacture devices on flexible or large-area substrates efficiently. Furthermore, pentacene possesses a relatively low highest occupied molecular orbital (HOMO) energy level, making it highly susceptible to oxidation and radical reactions upon exposure to air. This inherent instability leads to rapid degradation of device performance over time, compromising the longevity and reliability required for consumer electronics. In the solid state, pentacene molecules tend to adopt a 'herringbone' packing arrangement, which is not conducive to optimal π-conjugated orbital overlap. This suboptimal molecular stacking prevents the material from achieving its theoretical limit of charge carrier mobility, creating a bottleneck for high-speed organic circuit applications.
The Novel Approach
The methodology disclosed in the patent overcomes these hurdles by engineering a new class of tetracene derivatives that balance electronic performance with processability. By introducing flexible alkyl chains into the one-dimensional fused ring system, the inventors have successfully disrupted the tight crystalline packing that causes insolubility, thereby enabling solution-based processing methods. This solubility enhancement is critical for reducing manufacturing costs, as it allows for the use of techniques like spin-coating or inkjet printing instead of high-vacuum deposition. Moreover, the strategic incorporation of aryl and heteroaryl substituents modifies the electronic structure to improve air stability without sacrificing the high mobility characteristic of acene materials. The use of pyrene as the primary starting material is another distinct advantage, as it is a廉价 (cheap) and readily available industrial chemical compared to specialized acene precursors. This approach effectively decouples high performance from high manufacturing complexity, offering a pathway to cost reduction in electronic chemical manufacturing that was previously unattainable with conventional pentacene systems.
Mechanistic Insights into Sonogashira Coupling and Bergman Cyclization
The synthesis of these advanced tetracene derivatives relies on a sophisticated cascade of transition-metal-catalyzed reactions, primarily centering on the Sonogashira coupling followed by a thermal Bergman cyclization. The process begins with the functionalization of the pyrene core, where Friedel-Crafts alkylation introduces solubilizing groups, followed by halogenation to create a reactive tetrahalo-pyrene intermediate. This intermediate serves as the electrophilic partner in the subsequent Sonogashira cross-coupling reaction with terminal alkynes derived from various aryl or heteroaryl precursors. The palladium-copper catalytic system facilitates the formation of carbon-carbon bonds between the pyrene core and the ethynyl-substituted aromatic rings under mild conditions. Following the coupling, the resulting poly-alkynyl pyrene undergoes a Bergman cyclization, a thermal rearrangement that closes the rings to form the fully conjugated tetracene backbone. This cyclization step is crucial as it restores the planar aromaticity required for efficient charge transport while locking the substituents into their final positions.
From an impurity control perspective, this synthetic route offers significant advantages due to the high selectivity of the palladium-catalyzed steps. The use of specific ligands and controlled reaction temperatures minimizes the formation of homocoupling by-products, which are common pitfalls in alkyne chemistry. Furthermore, the modular nature of the Sonogashira reaction allows for precise tuning of the Ar and R groups, enabling chemists to optimize the HOMO/LUMO levels and solid-state packing without redesigning the entire synthetic pathway. The final cyclization step typically proceeds cleanly, yielding the target fused ring system with minimal side reactions. For quality control teams, this means the impurity profile is predictable and manageable, facilitating the isolation of high-purity OLED material or OFET active layers. The ability to purify intermediates via standard chromatography before the final cyclization ensures that the final product meets stringent purity specifications required for electronic applications.
How to Synthesize Tetracene Derivatives Efficiently
The preparation of these high-performance organic semiconductors follows a logical, multi-step sequence that transforms simple commodity chemicals into complex fused-ring architectures. The process is designed to be scalable, utilizing reagents and conditions that are compatible with standard chemical manufacturing infrastructure. Detailed operational parameters, including specific molar ratios, solvent choices, and temperature profiles, are critical for maximizing yield and minimizing batch-to-batch variability. The following guide outlines the standardized synthesis steps derived from the patent examples, providing a roadmap for technical teams aiming to replicate or adapt this chemistry.
- Functionalize pyrene core via Friedel-Crafts alkylation followed by halogenation to create the reactive tetrahalo-pyrene intermediate.
- Prepare ethynyl-substituted aromatic compounds through Sonogashira coupling of halo-aromatics with trimethylsilylacetylene and subsequent deprotection.
- Execute the final Sonogashira coupling and Bergman cyclization between the tetrahalo-pyrene and ethynyl-aromatics to form the fused tetracene backbone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this tetracene derivative technology presents a compelling value proposition centered on cost efficiency and supply security. The shift from vacuum-deposition-dependent materials to solution-processable semiconductors fundamentally changes the capital expenditure model for device fabrication. By enabling the use of printing technologies, manufacturers can significantly reduce the overhead associated with high-vacuum equipment maintenance and energy consumption. This transition not only lowers the barrier to entry for new market players but also enhances the agility of existing production lines to respond to fluctuating demand. Furthermore, the reliance on pyrene, a bulk chemical with a stable global supply chain, mitigates the risks associated with sourcing exotic or proprietary precursors. This stability ensures consistent lead times and protects against the price volatility often seen with niche fine chemical intermediates.
- Cost Reduction in Manufacturing: The primary driver for cost savings lies in the elimination of expensive vacuum deposition processes. Since these derivatives are soluble, they can be deposited using low-cost solution techniques, which drastically reduces equipment costs and throughput time. Additionally, the use of pyrene as a starting material avoids the premium pricing associated with pre-functionalized acenes, leading to substantial cost savings in raw material procurement. The high selectivity of the synthetic route also minimizes waste generation, further contributing to a leaner and more economical production process.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved by the use of commodity chemicals like pyrene and common aryl halides, which are produced at massive scales globally. This reduces dependency on single-source suppliers for specialized intermediates, thereby diversifying the supply base and reducing geopolitical or logistical risks. The robustness of the synthetic method ensures that production can be scaled up rapidly to meet surges in demand without the long lead times typically required for complex custom synthesis. This reliability is crucial for maintaining continuous production schedules in the fast-paced consumer electronics sector.
- Scalability and Environmental Compliance: The synthetic pathway is amenable to large-scale batch processing, utilizing standard reactors and purification units found in most fine chemical facilities. The reaction conditions are relatively mild, avoiding extreme pressures or temperatures that would require specialized containment or safety measures. Moreover, the ability to recycle solvents and the reduced need for heavy metal scavenging (due to high catalyst efficiency) aligns with increasingly strict environmental regulations. This compliance reduces the administrative burden and potential fines associated with hazardous waste disposal, making the process sustainable for long-term commercial operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this tetracene derivative technology in organic electronic devices. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation. Understanding these nuances is essential for evaluating the feasibility of integrating these materials into your current product development pipeline.
Q: How does this tetracene derivative improve upon traditional pentacene for OFET applications?
A: Unlike pentacene, which requires vacuum deposition due to poor solubility and suffers from oxidative instability, these tetracene derivatives feature flexible alkyl chains that significantly enhance solubility for solution processing. Additionally, the specific substitution pattern improves air stability and promotes favorable molecular packing for higher charge carrier mobility.
Q: What are the key cost advantages of using pyrene as the starting material?
A: Pyrene is a commercially abundant and inexpensive polycyclic aromatic hydrocarbon compared to specialized acene precursors. Utilizing pyrene as the core building block drastically reduces raw material costs and simplifies the supply chain, making large-scale production of organic semiconductors more economically viable.
Q: Can these materials be processed using standard solution-based techniques?
A: Yes, the introduction of long alkyl chains and specific aryl substituents renders these derivatives soluble in common organic solvents. This allows for fabrication via low-cost solution techniques such as spin-coating or inkjet printing, eliminating the need for expensive high-vacuum physical vapor deposition equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetracene Derivative Supplier
As the demand for high-performance organic semiconductors continues to grow, partnering with an experienced CDMO is essential for navigating the complexities of scale-up and commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to factory floor is seamless. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of tetracene derivative meets the exacting standards required for next-generation OFET and OLED applications. We understand the critical nature of supply continuity in the electronics industry and have optimized our logistics to support just-in-time delivery models.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how switching to our solution-processable materials can impact your overall bill of materials. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your project starts on a foundation of technical certainty and commercial viability.
