Advanced Tetracene Derivatives for High-Performance Organic Field-Effect Transistors
Advanced Tetracene Derivatives for High-Performance Organic Field-Effect Transistors
The rapid evolution of the organic electronics sector demands materials that balance high charge carrier mobility with processability and environmental stability. Patent CN102659752B introduces a groundbreaking class of tetracene derivative field-effect transistor materials that address the critical bottlenecks of traditional acene-based semiconductors. By leveraging a sophisticated synthetic strategy involving Sonogashira coupling and Bergman cyclization, this technology enables the production of solution-processable organic semiconductors with exceptional performance metrics. The core innovation lies in the strategic functionalization of the tetracene backbone with specific aryl and alkyl substituents, which not only modulates the electronic properties but also dramatically improves solubility. For R&D directors and procurement specialists seeking a reliable OLED material supplier or partners for electronic chemical manufacturing, this patent represents a pivotal shift towards scalable, high-performance organic electronics.
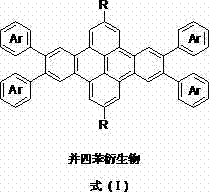
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the organic semiconductor industry has been heavily reliant on pentacene, a material known for its high hole mobility but plagued by severe practical limitations that hinder widespread commercial adoption. Pentacene suffers from extremely poor solubility in most common organic solvents, effectively restricting its deposition to physical vapor deposition (PVD) methods which require expensive high-vacuum equipment and limit throughput. Furthermore, pentacene possesses a relatively low highest occupied molecular orbital (HOMO) energy level, making it highly susceptible to oxidation and radical reactions upon exposure to ambient air, leading to rapid device degradation. In the condensed state, pentacene molecules tend to arrange in a "herringbone" pattern, a packing motif that is suboptimal for π-orbital overlap, thereby capping the achievable device mobility well below the theoretical limits of the material. These intrinsic defects create significant supply chain vulnerabilities and inflate the cost of goods sold for organic field-effect transistors (OFETs).
The Novel Approach
The methodology disclosed in CN102659752B offers a transformative solution by synthesizing tetracene derivatives that retain high mobility while overcoming the solubility and stability barriers of their predecessors. By introducing flexible alkyl chains and diverse aryl or heteroaryl groups into the one-dimensional fused ring system, the new materials exhibit excellent solubility, allowing for solution-based film formation techniques such as spin-coating or inkjet printing. This shift from vacuum deposition to solution processing is a game-changer for cost reduction in electronic chemical manufacturing, as it utilizes standard, lower-cost infrastructure. Additionally, the incorporation of heteroatoms like sulfur facilitates stronger intermolecular interactions through S-S contacts and enhanced π-orbital overlapping, resulting in tighter molecular packing and superior air stability. This approach ensures that the final OFET devices maintain high performance over extended operational lifetimes without the need for rigorous encapsulation.
Mechanistic Insights into Sonogashira Coupling and Bergman Cyclization
The synthetic pathway relies on a dual-reaction mechanism that efficiently constructs the complex polycyclic aromatic hydrocarbon framework. The process initiates with the preparation of ethynyl-substituted aryl precursors, typically achieved through a palladium-catalyzed Sonogashira cross-coupling reaction between halogenated aryl compounds and trimethylsilylacetylene. This step is critical for installing the necessary carbon-carbon triple bonds that serve as the handles for the subsequent ring-closing reaction. Following the deprotection of the trimethylsilyl group to reveal the terminal alkyne, these precursors are reacted with a tetrahalogenated pyrene core. The presence of the palladium catalyst and cuprous iodide facilitates the initial coupling, while the thermal conditions drive the intramolecular Bergman cyclization. This cascade reaction elegantly fuses the aromatic rings, extending the conjugation length of the system which is directly correlated with improved charge transport properties.
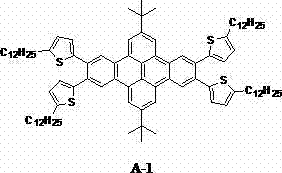
Controlling the impurity profile during this multi-step synthesis is paramount for ensuring high device yield and consistent batch-to-batch performance. The use of pyrene as the central scaffold is advantageous not only for its cost but also for its symmetry, which simplifies the regioselectivity of the substitution reactions. The reaction conditions, typically maintained between 60°C and 135°C in solvents like tetrahydrofuran or diisopropylamine, are optimized to minimize side reactions such as homocoupling of the alkynes or incomplete cyclization. The resulting tetracene derivatives, such as the thiophene-substituted variant shown above, demonstrate how specific heterocyclic integration can further tune the HOMO/LUMO levels. This precise mechanistic control allows manufacturers to tailor the electronic properties of the semiconductor to match specific device architectures, ensuring optimal compatibility with various gate dielectrics and electrode materials.
How to Synthesize Tetracene Derivative Efficiently
The synthesis of these high-performance materials follows a robust, modular protocol that is amenable to scale-up from gram-scale laboratory optimization to multi-kilogram commercial production. The process begins with the functionalization of the pyrene core via Friedel-Crafts alkylation, followed by selective halogenation to activate the 4,5,9,10-positions. Parallel to this, the aryl side chains are prepared and converted into terminal alkynes. The convergence of these two fragments via the final coupling-cyclization step yields the target tetracene derivative. Detailed standard operating procedures regarding stoichiometry, solvent grades, and purification methods are essential for maintaining the stringent purity specifications required for electronic applications.
- Perform Friedel-Crafts alkylation on pyrene using aluminum chloride and alkyl halides at 10-50°C to obtain 2,7-dialkylpyrene.
- Halogenate the 2,7-dialkylpyrene at 0-50°C to generate 2,7-dialkyl-4,5,9,10-tetrahalopyrene.
- Prepare ethynyl-substituted aryl compounds via Sonogashira coupling of halogenated aryls with trimethylsilylacetylene followed by deprotection.
- Conduct the final Sonogashira coupling and Bergman cyclization between the tetrahalopyrene and ethynyl-aryl compounds at 60-135°C using Pd/Cu catalysts.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this tetracene derivative technology offers substantial strategic benefits beyond mere technical performance. The reliance on pyrene, a commodity chemical derived from coal tar or petroleum cracking, ensures a stable and abundant supply of the core raw material, mitigating the risks associated with sourcing exotic or proprietary intermediates. The synthetic route avoids the use of ultra-expensive transition metal catalysts in stoichiometric quantities, utilizing instead catalytic amounts of palladium that can potentially be recovered, thereby driving down the direct material costs. Furthermore, the ability to process these materials from solution eliminates the need for capital-intensive vacuum deposition tools, significantly lowering the barrier to entry for device manufacturers and accelerating time-to-market for new organic electronic products.
- Cost Reduction in Manufacturing: The elimination of high-vacuum physical vapor deposition processes in favor of solution processing represents a massive reduction in operational expenditure. By enabling the use of roll-to-roll coating or printing technologies, manufacturers can achieve higher throughput rates with lower energy consumption. The use of inexpensive starting materials like pyrene and standard organic solvents further compresses the cost structure, making high-mobility OFETs economically viable for mass-market applications such as flexible displays and RFID tags.
- Enhanced Supply Chain Reliability: The synthetic pathway utilizes widely available reagents such as alkyl halides, thiophenes, and standard palladium catalysts, reducing dependency on single-source suppliers for niche chemicals. The robustness of the Sonogashira and Bergman reactions ensures high reproducibility, minimizing batch failures and ensuring consistent delivery schedules. This reliability is crucial for maintaining continuous production lines in the fast-paced consumer electronics sector, where supply disruptions can lead to significant revenue losses.
- Scalability and Environmental Compliance: The reaction conditions operate at moderate temperatures and pressures, facilitating safe scale-up in standard stainless steel reactors without the need for specialized high-pressure equipment. The byproducts generated during the synthesis are primarily inorganic salts and simple organic volatiles that can be managed through standard wastewater treatment protocols. This alignment with green chemistry principles simplifies regulatory compliance and reduces the environmental footprint of the manufacturing process, a key consideration for modern sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this tetracene derivative technology in organic electronic devices. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating these materials into existing product lines.
Q: How does this tetracene derivative overcome the solubility issues of pentacene?
A: Unlike pentacene, which requires vacuum deposition due to poor solability, this invention introduces flexible alkyl chains into the tetracene core. This structural modification significantly enhances solubility in common organic solvents, enabling solution-processing techniques which are far more cost-effective for large-scale manufacturing.
Q: What represents the primary cost advantage of this synthesis route?
A: The synthesis utilizes pyrene as the starting material, which is a cheap and readily available industrial feedstock compared to specialized precursors. Furthermore, the elimination of high-vacuum physical vapor deposition (PVD) equipment in favor of solution processing drastically reduces capital expenditure and operational costs in device fabrication.
Q: Does the introduction of heteroatoms affect the stability of the material?
A: Yes, the introduction of heteroatoms such as sulfur (in thiophene derivatives) enhances intermolecular interactions through S-S interactions and van der Waals forces. This leads to tighter molecular packing in the solid state, improving air stability and charge carrier mobility compared to traditional hydrocarbon acenes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetracene Derivative Supplier
As the demand for high-performance organic semiconductors continues to surge, partnering with an experienced CDMO is essential for navigating the complexities of process development and commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial reality is seamless. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of tetracene derivative meets the exacting standards required for next-generation OFET and OLED applications. We understand the critical nature of impurity profiles in electronic materials and employ advanced analytical techniques to ensure product consistency.
We invite you to engage with our technical procurement team to discuss how we can optimize your supply chain for these advanced materials. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into potential efficiency gains specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a robust foundation for your electronic material sourcing strategy.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
