Advanced Synthesis of Beta-Aminopropenones for Scalable Pharmaceutical Manufacturing
Advanced Synthesis of Beta-Aminopropenones for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for critical intermediates that balance cost efficiency with high purity standards. Patent CN101260062A introduces a transformative methodology for the synthesis of β-aminopropenone compounds, a class of molecules indispensable for constructing quinolone antibiotics and various heterocyclic drug scaffolds. This technology addresses long-standing bottlenecks in intermediate manufacturing by replacing hazardous or expensive precursors with accessible commodity chemicals. By leveraging a novel two-step condensation and aminolysis sequence, the process achieves superior yields under mild reaction conditions, positioning it as a cornerstone for modern API intermediate production. For R&D directors and procurement strategists, understanding this patent is crucial for optimizing supply chains and reducing the overall cost of goods sold in complex drug synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of β-aminopropenones has relied heavily on the addition of amines to alkynes, specifically utilizing propiolate esters as key starting materials. While chemically straightforward, this conventional pathway suffers from severe economic and logistical drawbacks. Propiolate esters are notoriously difficult to source in bulk quantities and command premium pricing due to their specialized synthesis requirements, rendering the final drug substance cost-prohibitive for mass market applications. Alternative historical methods, such as the cyanuric chloride route involving Gold reagents, introduce significant safety hazards and generate excessive waste streams, complicating environmental compliance and downstream purification. Furthermore, emerging industrial attempts using carbon monoxide insertion require high-pressure equipment and rigorous safety protocols, creating barriers to entry for many contract manufacturing organizations seeking flexible production capabilities.
The Novel Approach
The methodology disclosed in CN101260062A circumvents these challenges by employing a base-catalyzed condensation between readily available ketones or esters and formate esters. This strategic shift allows manufacturers to utilize low-cost feedstocks such as acetone, ethyl acetate, and ethyl formate, which are produced globally at massive scales. The reaction proceeds through a stable intermediate salt, which is subsequently converted to the target enamine structure upon treatment with a secondary amine. This approach not only drastically simplifies the reaction setup by eliminating the need for high-pressure reactors but also streamlines the workup procedure, often requiring only filtration and distillation. The versatility of this route is demonstrated by its compatibility with a wide range of substrates, enabling the efficient production of diverse β-aminopropenone derivatives essential for medicinal chemistry campaigns.

Mechanistic Insights into Base-Catalyzed Condensation and Aminolysis
The core of this synthetic innovation lies in the precise control of enolate chemistry and subsequent nucleophilic substitution. In the first stage, a strong base such as sodium methoxide, sodium ethoxide, or sodium hydride deprotonates the alpha-carbon of the ketone or ester substrate. This generates a reactive nucleophile that attacks the carbonyl carbon of the formate ester, leading to the formation of an α-formyl ketone or ester intermediate which exists primarily as a stable enolate salt (Formula IV). The stability of this intermediate is critical, as it prevents premature polymerization or side reactions that often plague enamine synthesis. The choice of base and solvent plays a pivotal role here; polar protic solvents like ethanol or methanol facilitate the dissolution of the base, while aprotic solvents like toluene or isopropyl ether can aid in the precipitation of the intermediate salt, driving the equilibrium forward and simplifying isolation.
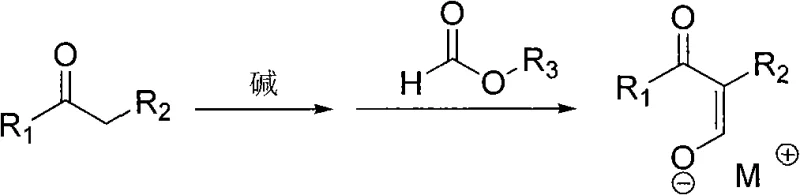
In the second mechanistic phase, the intermediate salt undergoes aminolysis upon exposure to a secondary amine or its hydrochloride salt. The amine acts as a nucleophile, attacking the electrophilic center of the enolate system. This is followed by an elimination step where the alkoxide or hydroxide leaving group is expelled, establishing the characteristic carbon-carbon double bond of the β-aminopropenone structure. A key advantage of this mechanism is the ability to perform the reaction at moderate temperatures ranging from -10°C to 80°C, which minimizes thermal degradation of the sensitive enamine product. To further ensure product integrity during the final isolation via vacuum distillation, the patent recommends the inclusion of polymerization inhibitors such as hydroquinone or N-isopropyl-N′-phenyl-p-phenylenediamine. This attention to detail in the mechanism ensures that the final API intermediate meets stringent purity specifications required for regulatory filing.
How to Synthesize Beta-Aminopropenone Efficiently
Implementing this synthesis requires careful attention to stoichiometry and solvent selection to maximize yield and minimize byproduct formation. The patent outlines a generalized protocol where the molar ratio of ketone/ester to formate to base to amine is optimized between 1:0.8~1.25:0.8~1.25:0.6~1.25. Operators typically dissolve the base in a suitable solvent like isopropyl ether or ethanol, followed by the sequential addition of the ketone and formate ester under controlled stirring. Once the intermediate salt suspension is formed, the amine component is introduced, and the mixture is agitated until conversion is complete. The detailed standardized synthesis steps, including specific temperature ramps and quenching procedures validated for GMP environments, are provided below to guide process engineers in scaling this technology.
- React a ketone or ester with a formate ester in the presence of a strong base (e.g., sodium methoxide) at -10 to 80°C to form an intermediate enolate salt.
- Treat the intermediate salt with a secondary amine (or its salt) at -10 to 80°C to effect nucleophilic addition and elimination.
- Purify the resulting beta-aminopropenone via filtration and vacuum distillation, optionally using polymerization inhibitors.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process offers tangible strategic benefits beyond mere chemical elegance. The primary value driver is the substantial reduction in raw material costs achieved by substituting exotic alkynes with commodity chemicals. Acetone and ethyl acetate are among the most produced organic solvents globally, ensuring a stable and competitive pricing landscape that shields manufacturers from volatile market fluctuations associated with specialty reagents. Additionally, the simplified reaction conditions eliminate the need for specialized high-pressure infrastructure, allowing production to be shifted to multipurpose facilities with lower capital expenditure requirements. This flexibility enhances supply chain resilience, enabling faster response times to market demand surges without the lead time associated with building dedicated high-pressure plants.
- Cost Reduction in Manufacturing: The elimination of expensive propiolate esters and hazardous cyanuric chloride reagents results in a significantly lower bill of materials. By utilizing sodium methoxide or sodium hydroxide as catalysts, the process avoids the use of costly transition metal catalysts that require complex removal steps to meet residual metal limits in pharmaceutical products. This simplification of the downstream purification train reduces solvent consumption and waste disposal costs, contributing to a leaner and more profitable manufacturing operation that aligns with green chemistry principles.
- Enhanced Supply Chain Reliability: Sourcing reliability is markedly improved as the key inputs—formate esters and simple ketones—are available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction across a broad temperature window (-10°C to 80°C) means that production is less susceptible to minor deviations in utility availability, such as cooling water temperature fluctuations. This operational tolerance ensures consistent batch-to-batch quality and reliable delivery schedules, which is critical for maintaining the continuity of API production lines and avoiding costly stockouts in the downstream pharmaceutical value chain.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, with examples demonstrating successful execution from laboratory to pilot scales using standard agitation and distillation equipment. The absence of toxic heavy metals and the use of recyclable solvents like ethanol and toluene simplify wastewater treatment and reduce the environmental footprint of the manufacturing site. Regulatory bodies increasingly favor such cleaner technologies, potentially accelerating the approval timeline for new drug applications that utilize intermediates produced via this environmentally benign and scalable pathway.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios and assess the potential impact on their product lifecycle management strategies.
Q: What are the primary advantages of this formate-based route over traditional alkyne methods?
A: Unlike traditional methods relying on expensive and hard-to-source propiolates, this patent utilizes readily available commodity ketones and formate esters. This significantly lowers raw material costs and simplifies the supply chain, making it ideal for industrial scale-up.
Q: How does the process handle impurity control during the reaction?
A: The process operates under mild temperatures (-10 to 80°C) and uses specific solvent systems like isopropyl ether or ethanol which facilitate the precipitation of inorganic salts. Furthermore, the addition of polymerization inhibitors during the final vacuum distillation prevents product degradation, ensuring high purity specifications.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the patent explicitly highlights favorable industrial production characteristics. The avoidance of high-pressure carbon monoxide and the use of standard atmospheric pressure conditions with common bases like sodium methoxide make the process highly scalable and safe for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Aminopropenone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped to handle the specific solvent systems and temperature controls required for this base-catalyzed condensation, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of beta-aminopropenone intermediate performs flawlessly in your subsequent synthetic steps.
We invite you to leverage our technical capabilities to optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our implementation of CN101260062A can become a cornerstone of your competitive advantage in the global pharmaceutical market.
