Scalable Production of Beta-Aminopropenone Compounds via Novel Base-Catalyzed Addition
Scalable Production of Beta-Aminopropenone Compounds via Novel Base-Catalyzed Addition
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more cost-effective and scalable synthetic routes. A pivotal advancement in this domain is detailed in patent CN101260062B, which discloses a highly efficient method for synthesizing β-aminopropenone compounds. These versatile molecules serve as critical building blocks in organic synthesis, particularly for the construction of heterocyclic systems found in numerous active pharmaceutical ingredients (APIs). The significance of this technology lies in its ability to bypass traditional bottlenecks associated with alkyne-based syntheses, offering a pathway that is not only chemically robust but also commercially viable for large-scale production. For R&D directors and procurement strategists alike, understanding the nuances of this base-catalyzed condensation and subsequent amination process is essential for optimizing supply chains and reducing the overall cost of goods sold (COGS) in complex drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of β-aminopropenone compounds has relied heavily on the addition reaction between alkynes and amines. While this laboratory-scale approach offers mild reaction conditions, it suffers from severe economic and logistical drawbacks when translated to an industrial setting. The primary constraint is the reliance on propiolate esters as starting materials, which are notoriously difficult to source in bulk quantities and command premium pricing due to their specialized nature. Furthermore, alternative routes such as the cyanuric chloride method, which utilizes Gold reagents, have been plagued by significant side reactions, low yields, and the generation of substantial hazardous waste, rendering them environmentally unsustainable and economically unfeasible for modern green chemistry standards. Even emerging carbon monoxide routes, while promising, often require high-pressure equipment and complex safety protocols that increase capital expenditure and operational risk.
The Novel Approach
In stark contrast to these legacy methods, the technology outlined in CN101260062B introduces a streamlined two-step sequence that leverages abundant and inexpensive feedstocks. The process initiates with a base-catalyzed addition reaction between a ketone or ester and a formate ester, generating a stable intermediate salt. This intermediate is then subjected to a second addition reaction with an amine to yield the target β-aminopropenone. This methodology effectively decouples the synthesis from expensive alkyne precursors, replacing them with commodity chemicals like acetone, ethyl acetate, and ethyl formate. The operational simplicity is further enhanced by the use of common organic solvents and moderate temperature ranges, facilitating a smoother transition from benchtop discovery to commercial manufacturing without the need for exotic catalysts or extreme process conditions.
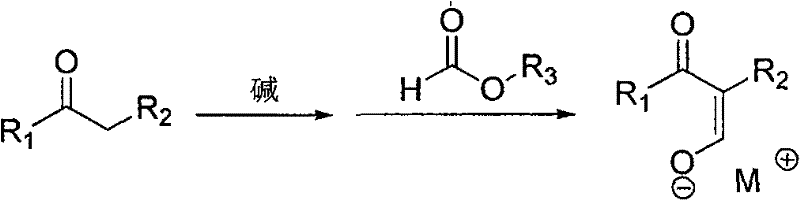
Mechanistic Insights into Base-Catalyzed Condensation and Amination
The core of this synthetic strategy relies on a classic yet optimized Claisen-type condensation mechanism followed by nucleophilic substitution. In the first stage, a strong base such as sodium methoxide, sodium ethoxide, or sodium hydride deprotonates the alpha-carbon of the ketone or ester substrate. This generates a reactive enolate species which subsequently attacks the carbonyl carbon of the formate ester. The result is the formation of an enolate salt intermediate, characterized by a conjugated system that stabilizes the negative charge through resonance with the adjacent carbonyl group. This step is critical as it establishes the carbon backbone of the final product, and the choice of base and solvent plays a pivotal role in driving the equilibrium towards the desired intermediate while minimizing self-condensation of the ketone.
Following the formation of the intermediate salt, the second stage involves the introduction of a secondary amine, such as dimethylamine or diethylamine. The amine acts as a nucleophile, attacking the electrophilic center of the intermediate salt. This displacement reaction results in the formation of the C-N bond, yielding the final β-aminopropenone structure. A crucial aspect of this mechanism, particularly for maintaining product integrity during isolation, is the management of polymerization risks. The patent explicitly details the use of polymerization inhibitors like N-isopropyl-N'-phenyl-p-phenylenediamine (4010NA) or hydroquinone during the vacuum distillation phase. This additive strategy prevents the thermal degradation of the sensitive enaminone double bond, ensuring that the final product meets stringent purity specifications required for pharmaceutical applications.
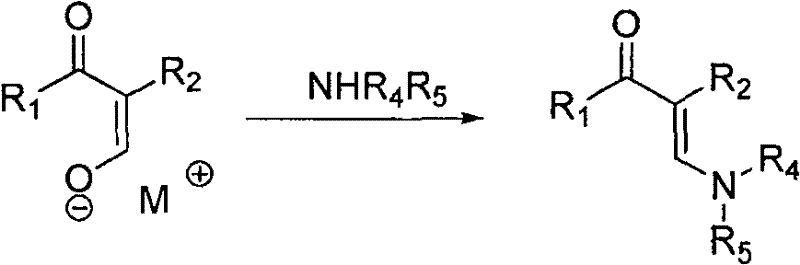
How to Synthesize Beta-Aminopropenone Efficiently
Implementing this synthesis route requires precise control over stoichiometry and reaction parameters to maximize yield and minimize impurities. The patent provides specific guidance on molar ratios, suggesting a balanced approach where the ketone or ester, formate, strong base, and amine are typically employed in ratios ranging from 1:0.8–1.25:0.8–1.25:0.6–1.25. The reaction can be conducted in a variety of protic or aprotic solvents, including methanol, ethanol, toluene, and isopropyl ether, offering flexibility based on solubility profiles and downstream processing requirements. Temperatures are maintained between -10°C and 80°C, allowing for energy-efficient operations that do not require cryogenic cooling or high-energy heating inputs. The following guide outlines the standardized procedural steps derived from the patent's preferred embodiments.
- React a ketone or ester with a formate ester in the presence of a strong base (e.g., sodium methoxide) at -10 to 80°C to form an intermediate salt.
- React the resulting intermediate salt with a secondary amine (or its salt) at -10 to 80°C to form the beta-aminopropenone product.
- Purify the final reaction mixture by filtration and vacuum distillation, optionally adding polymerization inhibitors to prevent degradation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route represents a strategic opportunity to enhance supply security and reduce manufacturing costs. By shifting away from niche alkyne derivatives to commodity ketones and formates, companies can mitigate the risks associated with single-source suppliers and volatile raw material markets. The simplified workup procedure, which primarily involves filtration and distillation rather than complex chromatographic purification, translates directly into reduced processing time and lower solvent consumption. This efficiency gain is compounded by the high yields reported in the patent examples, which minimize material loss and maximize the throughput of existing production assets, thereby improving the overall return on investment for manufacturing campaigns.
- Cost Reduction in Manufacturing: The elimination of expensive propiolate esters and complex Gold reagents fundamentally alters the cost structure of producing β-aminopropenones. By utilizing widely available starting materials like acetone and ethyl formate, the direct material costs are drastically lowered. Furthermore, the avoidance of transition metal catalysts removes the need for costly metal scavenging steps and rigorous heavy metal testing, which are often required to meet regulatory limits in pharmaceutical products. This streamlining of the process chemistry leads to substantial operational savings that can be passed down the value chain.
- Enhanced Supply Chain Reliability: The reliance on bulk commodity chemicals ensures a stable and continuous supply of raw materials, insulating production schedules from the disruptions often seen with specialized fine chemical intermediates. The robustness of the reaction conditions, which tolerate a range of temperatures and solvents, adds another layer of resilience, allowing for flexible manufacturing across different facilities. This reliability is crucial for maintaining just-in-time inventory levels and meeting the demanding delivery timelines of downstream API manufacturers without compromising on quality or consistency.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind. The use of standard unit operations such as stirred tank reactors and distillation columns facilitates easy scale-up from pilot plants to multi-ton commercial facilities. Additionally, the reduction in hazardous waste generation compared to the cyanuric chloride route aligns with increasingly strict environmental regulations. The ability to recycle solvents and the minimization of toxic byproducts contribute to a smaller environmental footprint, supporting corporate sustainability goals and reducing waste disposal costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific application needs. Understanding these details is vital for making informed decisions about process adoption and vendor selection.
Q: What are the primary advantages of this synthesis method over traditional alkyne addition?
A: Unlike traditional methods relying on expensive and hard-to-source propiolates, this method utilizes readily available ketones or esters and formate esters. This significantly reduces raw material costs and simplifies the supply chain logistics for large-scale manufacturing.
Q: How does the process handle potential polymerization during purification?
A: The patent specifically addresses the risk of polymerization during vacuum distillation by recommending the addition of specific polymerization inhibitors such as N-isopropyl-N'-phenyl-p-phenylenediamine (4010NA) or hydroquinone, ensuring high purity and stability of the final enaminone product.
Q: Is this method suitable for industrial scale-up?
A: Yes, the process is designed for industrial viability. It operates under mild temperatures (-10 to 80°C), uses common solvents like methanol or isopropyl ether, and involves straightforward workup procedures like filtration and distillation, avoiding complex chromatographic separations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Aminopropenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development programs. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify identity and assess impurity profiles. Our capability to implement the base-catalyzed synthesis described in CN101260062B allows us to offer a competitive and reliable supply of β-aminopropenone compounds to the global market.
We invite you to engage with our technical procurement team to discuss how this optimized synthetic route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this methodology. We encourage potential partners to contact us for specific COA data and route feasibility assessments, ensuring that our solutions are perfectly aligned with your quality standards and production timelines. Let us collaborate to drive efficiency and innovation in your supply chain.
