Advanced Convergent Synthesis of Carfilzomib for Commercial Scale-Up and High Purity
Advanced Convergent Synthesis of Carfilzomib for Commercial Scale-Up and High Purity
The pharmaceutical industry continuously seeks robust manufacturing pathways for complex oncology therapeutics, and the proteasome inhibitor Carfilzomib represents a critical asset in treating multiple myeloma. A pivotal advancement in this domain is detailed in patent CN103864898A, which discloses a novel preparation method that fundamentally shifts the production paradigm from traditional linear sequences to a highly efficient convergent synthesis strategy. This technical breakthrough addresses longstanding challenges regarding yield optimization and process controllability, offering a viable route for the reliable pharmaceutical intermediates supplier market to meet escalating global demand. By streamlining the condensation and deprotection steps among amino acid derivatives, this method ensures that the final active pharmaceutical ingredient meets stringent quality standards while maintaining economic feasibility for large-scale operations. The structural integrity of the target molecule, as depicted below, relies on precise stereochemical control which this new methodology supports effectively.
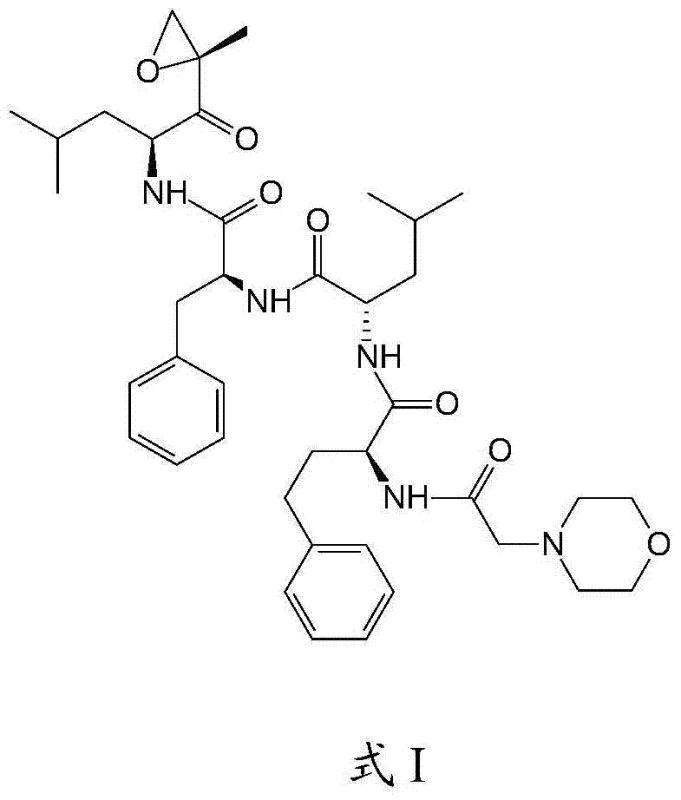
Carfilzomib, chemically known as (2S)-N-((S)-1-((S)-4-methyl-1-((R)-2-methyloxiran-2-yl)-1-oxopentan-2-ylamino)-1-oxo-3-phenylpropan-2-ylamino)-1-oxo-3-phenylpropan-2-yl)-2-(2-morpholinoacetamido)-4-methylpentanamide, requires meticulous assembly to preserve its pharmacological activity. The conventional approaches often suffer from cumulative yield losses and the introduction of difficult-to-remove impurities, which complicates the downstream purification processes essential for a high-purity Carfilzomib product. The innovation presented in the referenced patent utilizes a strategic disconnection of the molecule into manageable fragments that are coupled under mild conditions, thereby reducing the risk of epimerization and degradation. This approach not only enhances the chemical purity but also simplifies the regulatory documentation required for drug master files, as the process is more reproducible and easier to validate. For procurement teams, understanding the underlying chemistry is vital to assessing the long-term viability and cost reduction in API manufacturing associated with adopting this superior synthetic route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Carfilzomib has relied on linear strategies, such as those described in prior art like US20050245435, which involve a sequential buildup of the peptide chain that introduces significant inefficiencies at every stage. These traditional methods typically necessitate the use of sodium iodide for chlorination reactions, a step that is notorious for low yields and the generation of substantial inorganic waste that burdens environmental compliance systems. Furthermore, the reliance on palladium-carbon catalysts for hydrogenation steps introduces the risk of heavy metal contamination, requiring expensive and time-consuming scavenging procedures to meet safety specifications. The complexity of reaction conditions in these linear routes often leads to poor controllability, where slight deviations in temperature or stoichiometry can result in batch failures or off-spec material that cannot be reworked. Consequently, the overall yield of the finished product in these legacy processes is often suppressed to single-digit percentages, driving up the cost of goods sold and creating supply chain vulnerabilities for manufacturers dependent on these inefficient technologies.
The Novel Approach
In stark contrast, the novel convergent synthesis method outlined in the patent data revolutionizes the production landscape by minimizing the number of discrete operational units and eliminating hazardous reagents entirely from the critical path. This approach adopts a strategy where key fragments, specifically the leucine-phenylalanine dipeptide core and the morpholine-containing side chain, are prepared independently and then coupled, which isolates potential failure points and prevents the loss of valuable intermediates early in the sequence. By avoiding the use of chloroacetyl chloride and sodium iodide, the process inherently reduces pollution and improves reaction efficiency, leading to a cleaner crude product that requires less aggressive purification. The reaction conditions are notably mild and controllable, typically operating within a temperature range of -20°C to 60°C, which allows for standard stainless steel reactor usage without the need for specialized cryogenic or high-pressure equipment. This simplification translates directly into enhanced supply chain reliability, as the process is more robust against variability and easier to transfer between different manufacturing sites without loss of performance.
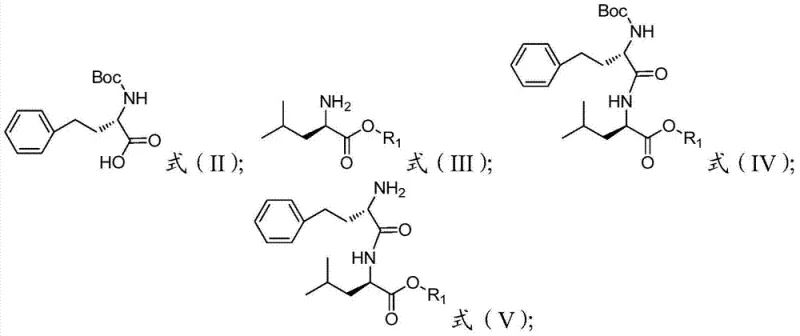
Mechanistic Insights into Peptide Condensation and Deprotection
The core of this technological advancement lies in the precise execution of condensation reactions between protected amino acids, utilizing advanced coupling agents to drive the formation of amide bonds with high fidelity. In the initial phase, N-Boc-L-homophenylalanine reacts with an L-leucine ester salt in the presence of organic bases and condensing agents such as DIC or HATU, forming a protected dipeptide intermediate that serves as the foundation for the entire molecule. The mechanistic advantage here is the use of mild deprotection conditions, typically employing trifluoroacetic acid or hydrochloric acid at controlled temperatures, which removes the Boc group without affecting the stereochemistry of the chiral centers. This preservation of chirality is paramount for the biological activity of the final drug, as even minor racemization can render the product inactive or toxic. The subsequent coupling with morpholin-4-yl-acetic acid further extends the peptide chain, and the careful selection of solvents like acetonitrile or DMF ensures that all reactants remain in solution, maximizing collision frequency and reaction rates. This level of mechanistic control is what enables the consistent production of high-purity Carfilzomib, satisfying the rigorous demands of R&D directors who prioritize impurity profile management.
Furthermore, the impurity control mechanism is intrinsically built into the choice of reagents and the sequence of deprotection steps, which systematically eliminate potential byproducts before they can accumulate. For instance, the avoidance of strong nucleophiles in the deprotection phases prevents unwanted side reactions such as ester hydrolysis or epoxide ring opening, which are common pitfalls in epoxy ketone synthesis. The patent specifies that the final recrystallization steps can achieve purity levels exceeding 99.5%, with single impurities maintained below 0.1%, demonstrating the efficacy of this chemical design in managing the杂质谱 (impurity spectrum). By utilizing commercially available starting materials that are already highly purified, the process reduces the burden on the analytical team to identify and quantify unknown degradants. This transparency in the chemical pathway facilitates faster regulatory approval and reduces the risk of clinical holds due to quality issues, making it a preferred strategy for the commercial scale-up of complex proteasome inhibitors.
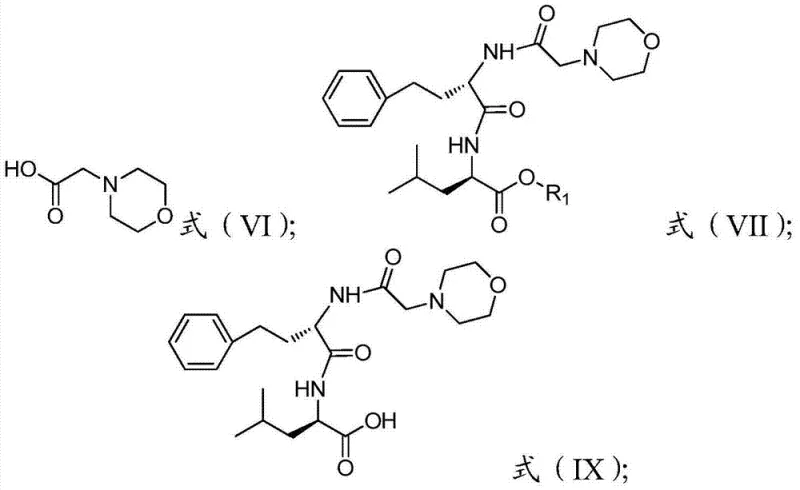
How to Synthesize Carfilzomib Efficiently
Implementing this synthesis route requires a disciplined approach to process chemistry, focusing on the precise stoichiometry and temperature control outlined in the patent embodiments to ensure reproducibility. The procedure begins with the activation of the carboxylic acid component using phosphonium or uronium-based coupling reagents, followed by the addition of the amine component under inert atmosphere to prevent moisture-induced hydrolysis. Detailed standardized synthesis steps are critical for maintaining the integrity of the epoxy ketone warhead, which is sensitive to acidic and basic conditions if not handled correctly. Operators must adhere strictly to the specified molar ratios, typically ranging from 1:1.05 to 1:2.5 for reactants, to drive the reaction to completion while minimizing excess reagent carryover. The workup procedures involve systematic extraction and washing protocols designed to remove urea byproducts and unreacted starting materials, ensuring that the intermediate isolated is of sufficient quality for the next coupling step without additional chromatography. This streamlined workflow is essential for reducing lead time for high-purity peptide intermediates, allowing manufacturing teams to respond quickly to market demands.
- Condense N-Boc-L-homophenylalanine with L-leucine ester followed by deamination protection to obtain Formula V intermediate.
- React Formula V with morpholin-4-yl-acetic acid followed by decarboxylation protection to generate Formula IX.
- Perform final condensation between Formula IX and the epoxy ketone Formula VIII to directly generate Carfilzomib.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this convergent synthesis method offers profound benefits that extend beyond mere technical superiority, directly impacting the bottom line and operational resilience of the supply chain. The elimination of expensive and hazardous reagents such as palladium catalysts and sodium iodide drastically simplifies the procurement landscape, reducing dependency on volatile raw material markets and specialized waste disposal vendors. This simplification allows for more accurate forecasting and budget planning, as the cost structure becomes more stable and predictable over the long term. Additionally, the higher yields achieved through this method mean that less starting material is required to produce the same amount of final product, effectively lowering the material cost per kilogram and improving the overall margin profile for the manufacturing entity. For supply chain heads, the robustness of the process translates to fewer batch failures and less downtime, ensuring a continuous flow of material to fill pipelines and meet contractual obligations without interruption.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the significant reduction in unit operations and the removal of costly purification steps associated with heavy metal scavenging. By avoiding the use of transition metal catalysts, the manufacturer eliminates the need for expensive ligand recovery systems and rigorous testing for residual metals, which are resource-intensive activities. Furthermore, the improved yield means that the fixed costs of labor, energy, and equipment depreciation are spread over a larger output of saleable product, driving down the average cost of production substantially. The use of readily available solvents and reagents also mitigates the risk of price spikes associated with specialty chemicals, providing a more resilient cost base that can withstand market fluctuations. These factors combine to create a compelling value proposition for procurement managers seeking to optimize their spend without compromising on quality standards.
- Enhanced Supply Chain Reliability: Operational continuity is significantly bolstered by the mild reaction conditions and the use of stable intermediates that can be stored or transported with minimal risk of degradation. The process does not rely on cryogenic temperatures or high-pressure hydrogenation, which are often bottlenecks in multi-purpose facilities, thereby increasing the flexibility to schedule production runs across different campaigns. This flexibility is crucial for managing inventory levels and responding to sudden surges in demand from downstream pharmaceutical partners. Moreover, the reduced complexity of the synthesis reduces the likelihood of human error during execution, leading to a higher first-pass yield rate and less need for reprocessing. This reliability fosters stronger relationships with customers who depend on just-in-time delivery models, positioning the supplier as a trusted partner capable of sustaining long-term agreements.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of exothermic hazards and the use of standard unit operations that are well-understood by engineering teams. The reduction in hazardous waste generation aligns with increasingly stringent global environmental regulations, reducing the liability and permitting hurdles associated with chemical manufacturing. By minimizing the use of halogenated solvents and toxic reagents, the facility can operate with a smaller environmental footprint, which is increasingly important for corporate social responsibility reporting and stakeholder engagement. The ability to scale efficiently without proportionate increases in waste treatment costs ensures that the economic benefits of the process are maintained even at multi-ton production volumes. This scalability is essential for supporting the growing patient population requiring access to this life-saving medication.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific Carfilzomib synthesis pathway, derived directly from the patent's experimental data and comparative examples. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into their existing portfolios or supply networks. The answers provided are grounded in the objective findings of the patent documentation, ensuring accuracy and relevance for decision-makers. These insights clarify how the method overcomes specific historical barriers to entry in the proteasome inhibitor market.
Q: How does this convergent method improve yield compared to linear synthesis?
A: The patented convergent method significantly reduces reaction steps and avoids harsh conditions like sodium iodide chlorination, improving overall yield from approximately 10% to over 30%.
Q: What are the key impurities controlled in this process?
A: By avoiding palladium-carbon catalysts and utilizing mild deprotection conditions, the process minimizes heavy metal residues and racemization, ensuring single impurity levels below 0.1%.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method uses commercially available reagents, mild temperatures ranging from -20°C to 60°C, and avoids explosive or highly toxic reagents, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carfilzomib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a manufacturing partner who combines deep technical expertise with the capacity to deliver at scale. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to full-scale supply. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Carfilzomib meets the highest international standards. Our commitment to quality is unwavering, and we understand that consistency is key to maintaining regulatory approval and patient safety. By leveraging our infrastructure, you gain access to a supply chain that is both robust and responsive, capable of adapting to your specific volume requirements without compromising on the integrity of the product.
We invite you to engage with our technical procurement team to discuss how we can tailor our capabilities to support your specific needs. Request a Customized Cost-Saving Analysis today to understand the potential economic benefits of switching to this optimized synthesis route for your projects. We are ready to provide specific COA data and route feasibility assessments to demonstrate our competence and readiness to serve as your long-term strategic partner. Let us help you secure a stable supply of high-quality intermediates that will drive the success of your pharmaceutical developments.
