Revolutionizing Carfilzomib Production: A Novel Organocatalytic Epoxidation Strategy for Industrial Scale
The pharmaceutical industry continuously seeks robust synthetic pathways for complex oncology therapeutics, and the recent disclosure in patent CN111646958B presents a significant advancement in the manufacturing of Carfilzomib, a potent proteasome inhibitor approved for treating multiple myeloma. This patent details a highly efficient preparation method for the critical epoxy ketone intermediate, designated as Formula I, which serves as the chiral foundation for the final active pharmaceutical ingredient. By leveraging a novel asymmetric epoxidation strategy utilizing hydrogen peroxide and specialized chiral organic catalysts, this technology addresses long-standing challenges regarding selectivity and cost that have plagued previous synthetic routes. The innovation lies in the replacement of expensive transition metal catalysts with readily available organocatalysts, facilitating a greener and more economically sustainable production process. For global procurement teams and R&D directors, this development signals a potential shift towards more reliable supply chains for high-value anticancer intermediates, ensuring consistent quality and reduced dependency on scarce metal resources.
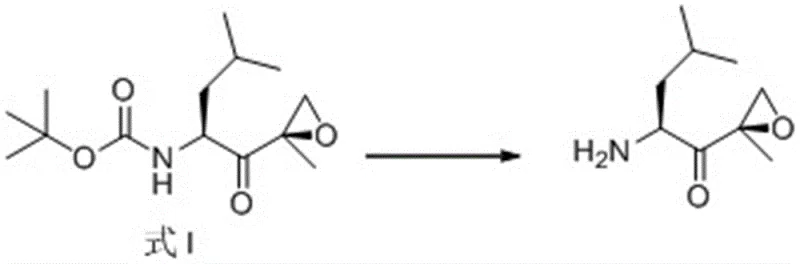
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the key epoxy ketone moiety in Carfilzomib has been fraught with inefficiencies that hinder large-scale industrial adoption. Early methodologies, such as those reported in Bioorganic & Medicinal Chemistry Letters, suffered from inherently low yields and poor control over the epoxidation step, resulting in difficult purification profiles and substantial material loss. Subsequent improvements, like the route disclosed in WO2009045497, attempted to enhance stereocontrol through a multi-step sequence involving chiral hydroxyl induction followed by Dess-Martin oxidation. However, this approach introduced significant operational complexity due to the length of the synthetic route and the prohibitive cost of stoichiometric oxidizing reagents. Furthermore, while manganese-complex catalyzed routes offered improved yields, they were severely limited by the high expense and commercial scarcity of the chiral ligands required, alongside the regulatory burden of removing trace heavy metals from the final product.
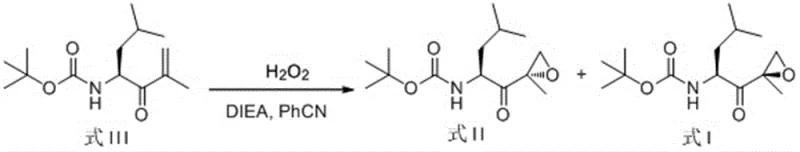
The Novel Approach
In stark contrast to these legacy methods, the technology outlined in CN111646958B introduces a direct and streamlined asymmetric epoxidation of the alpha,beta-unsaturated ketone precursor, Formula III. This novel approach bypasses the need for pre-functionalization of the substrate or the use of precious metal catalysts, relying instead on a synergistic combination of hydrogen peroxide and a silyl-protected proline derivative. The reaction conditions are remarkably mild, operating effectively at temperatures between -20°C and 10°C, which reduces energy consumption compared to deep cryogenic processes. By achieving high diastereoselectivity directly from the enone substrate, this method drastically shortens the overall process timeline and minimizes the generation of hazardous waste. The simplicity of the workup procedure, involving standard aqueous quenching and extraction, further underscores its suitability for seamless integration into existing GMP manufacturing facilities without requiring specialized equipment.
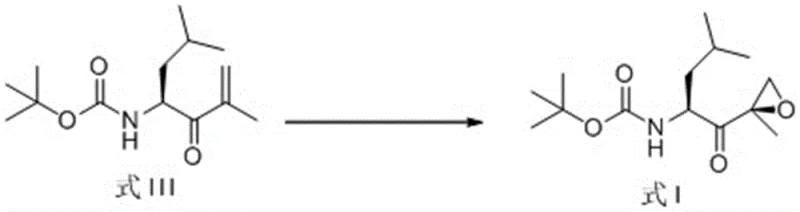
Mechanistic Insights into Silyl-Pyrrolidine Catalyzed Epoxidation
The core of this technological breakthrough resides in the unique structural features of the chiral organocatalysts employed, specifically the (2S)-2-[diphenyl[(trialkylsilyl)oxy]methyl]-pyrrolidine derivatives. These catalysts function by activating the hydrogen peroxide oxidant through hydrogen bonding interactions while simultaneously organizing the substrate within a well-defined chiral pocket created by the bulky diphenylsilyl and alkyl groups. This precise spatial arrangement ensures that the oxygen transfer occurs exclusively from one face of the enone double bond, thereby enforcing high stereochemical fidelity. The presence of the silyl ether moiety is critical, as it provides the necessary steric bulk to differentiate between the prochiral faces of the substrate, a feature that simple proline derivatives lack. Understanding this mechanism allows process chemists to fine-tune the catalyst loading and solvent environment to maximize the formation of the desired (R)-epoxide isomer while suppressing the formation of the unwanted optical isomer, Formula II.
![Chemical structure of the chiral organocatalyst (2S)-2-[diphenyl[(trimethylsilyl)oxy]methyl]-pyrrolidine](/insights/img/carfilzomib-intermediate-organocatalysis-pharma-supplier-20260306232953-06.webp)
Furthermore, the choice of solvent system plays a pivotal role in modulating the catalyst's performance and the reaction's selectivity profile. The patent highlights the efficacy of binary solvent mixtures, particularly combinations of toluene or acetonitrile with N-methylpyrrolidone (NMP), in stabilizing the transition state and enhancing the solubility of both the organic substrate and the aqueous oxidant. This biphasic or homogeneous mixing capability ensures efficient mass transfer of the hydrogen peroxide to the catalytic site, preventing localized over-oxidation or decomposition of the sensitive epoxy ketone product. The robustness of this catalytic system is evidenced by its ability to maintain high selectivity even with varying equivalents of oxidant, providing a wide operational window for manufacturing. Such mechanistic resilience is essential for scaling up reactions, as it mitigates the risks associated with exothermic events and ensures batch-to-batch consistency in the critical quality attributes of the intermediate.
How to Synthesize Carfilzomib Intermediate Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters to fully realize the benefits of high yield and purity described in the patent documentation. The process begins with the dissolution of the starting enone in the optimized solvent blend, followed by the addition of the chiral catalyst under inert atmosphere to prevent moisture interference prior to the intended oxidation step. Temperature control is paramount during the addition of hydrogen peroxide to manage the exotherm and preserve the integrity of the epoxide ring. Detailed standardized operating procedures regarding addition rates, stirring speeds, and quenching protocols are essential for safe and effective execution. For comprehensive technical guidance on executing this transformation, please refer to the step-by-step synthesis guide provided below.
- Dissolve the alpha,beta-unsaturated ketone precursor (Formula III) in a mixed solvent system of toluene and N-methylpyrrolidone.
- Add the chiral organocatalyst, such as (2S)-2-[diphenyl[(triethylsilyl ester)oxy]methyl]-pyrrolidine, and cool the reaction mixture to sub-zero temperatures.
- Slowly introduce aqueous hydrogen peroxide as the oxidant, maintain the temperature, and proceed with standard aqueous workup and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this organocatalytic methodology offers profound advantages that extend beyond mere chemical efficiency, directly impacting the bottom line and supply chain resilience. By eliminating the reliance on manganese complexes, manufacturers can avoid the volatility associated with the pricing and availability of transition metal catalysts, which are often subject to geopolitical supply constraints. The substitution of expensive stoichiometric oxidants like mCPBA or Dess-Martin periodinane with commodity-grade hydrogen peroxide results in a drastic reduction in raw material costs. Additionally, the simplified purification process, driven by the high diastereomeric ratio achieved, reduces the consumption of chromatography media and solvents, further lowering the cost of goods sold. These factors collectively contribute to a more predictable and cost-effective manufacturing model for this high-value pharmaceutical intermediate.
- Cost Reduction in Manufacturing: The economic benefits of this route are primarily derived from the replacement of noble metal catalysts and expensive oxidizing agents with affordable organocatalysts and hydrogen peroxide. This shift eliminates the need for costly metal scavenging steps and reduces the overall reagent expenditure per kilogram of product. Furthermore, the high selectivity minimizes the loss of valuable starting materials to side products, maximizing the atom economy of the process. Consequently, the total production cost is significantly lowered, allowing for more competitive pricing strategies in the global market for oncology intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: Utilizing widely available reagents such as hydrogen peroxide and common organic solvents ensures that the supply chain is not vulnerable to single-source bottlenecks often associated with specialized chiral ligands. The robustness of the reaction conditions allows for flexible manufacturing scheduling, as the process is less sensitive to minor fluctuations in environmental parameters. This reliability translates to shorter lead times and a higher degree of certainty in meeting delivery commitments for downstream API manufacturers. By securing a stable source of raw materials, companies can mitigate the risks of production delays and ensure continuous availability of this critical therapeutic building block.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing reaction conditions that are easily managed in large-scale reactors without requiring extreme cryogenic capabilities. The use of hydrogen peroxide generates water as the primary byproduct, aligning with green chemistry principles and simplifying waste treatment protocols. This environmental compatibility reduces the regulatory burden and disposal costs associated with hazardous chemical waste. Moreover, the straightforward workup procedure facilitates rapid throughput, enabling manufacturers to respond quickly to market demand surges. The combination of operational simplicity and environmental sustainability makes this route an ideal candidate for long-term commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis presented in the patent specification, aiming to clarify the practical implications for industrial adoption. Understanding these nuances is crucial for technical teams evaluating the feasibility of integrating this new route into their existing manufacturing portfolios. The answers provided reflect the specific advantages in selectivity, yield, and operational safety that distinguish this method from prior art.
Q: What are the primary advantages of this organocatalytic route over manganese-based methods?
A: Unlike manganese complex catalysts which are expensive and commercially scarce, the silyl-protected proline catalysts described in CN111646958B are more accessible and eliminate the need for heavy metal removal steps, significantly simplifying downstream processing and reducing environmental impact.
Q: How does this method improve the diastereomeric ratio compared to earlier literature?
A: The patent demonstrates that specific silyl-protected pyrrolidine catalysts can achieve diastereomeric ratios as high as 20:1, vastly superior to the 7:1 ratio observed in previous manganese-catalyzed methods, thereby reducing the burden on purification processes.
Q: Is this process suitable for large-scale manufacturing of API intermediates?
A: Yes, the process utilizes inexpensive oxidants like hydrogen peroxide and avoids cryogenic conditions lower than -20°C, making it thermally manageable and economically viable for multi-kilogram to ton-scale production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carfilzomib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing high-quality intermediates for the production of life-saving oncology therapies like Carfilzomib. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of our epoxy ketone intermediates meets the highest international standards. Our expertise in asymmetric synthesis allows us to optimize processes for maximum efficiency, delivering products that facilitate smoother downstream API manufacturing for our partners.
We invite you to collaborate with us to leverage this advanced organocatalytic technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can support your strategic goals in the competitive landscape of pharmaceutical manufacturing.
