Advanced Synthesis of Agmatine Sulfate: A Commercial Scale-Up Guide for Pharmaceutical Leaders
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally compliant synthetic routes for high-value intermediates. Patent CN101717352A represents a significant technological breakthrough in the production of Agmatine Sulfate (III), a critical neuroprotective agent and potential therapeutic for opioid withdrawal. This patent details a novel condensation methodology that replaces traditional, hazardous reagents with safer, more efficient alternatives, directly addressing the pain points of modern GMP manufacturing. By shifting from sulfur-based guanidinylation agents to oxygen-based analogues, the process fundamentally alters the impurity profile and waste stream characteristics. For R&D Directors and Supply Chain Heads, understanding this shift is crucial for securing a reliable agmatine sulfate supplier capable of meeting stringent regulatory standards. The following analysis dissects the chemical innovation behind this patent, evaluating its feasibility for commercial scale-up of complex pharmaceutical intermediates and its potential to drive substantial cost reduction in pharmaceutical intermediates manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of agmatine sulfate has relied heavily on the reaction between 1,4-butanediamine and S-methylisothiourea salts. While chemically feasible, this legacy approach presents severe operational and environmental liabilities that hinder large-scale adoption. The primary drawback lies in the leaving group; the displacement of the methylthio group generates alkyl mercaptans as byproducts. These sulfur-containing compounds are notorious for their intensely unpleasant odors, which pose significant challenges for labor protection and community relations around manufacturing sites. Furthermore, alkyl mercaptans are toxic and require specialized scrubbing systems and waste treatment protocols to prevent environmental contamination, directly contradicting modern green chemistry principles. From a yield perspective, conventional methods often struggle to exceed 40% efficiency, leading to poor atom economy and inflated raw material costs. The presence of sulfur also complicates the purification landscape, potentially introducing difficult-to-remove sulfurous impurities that can compromise the quality of high-purity agmatine sulfate required for clinical applications.
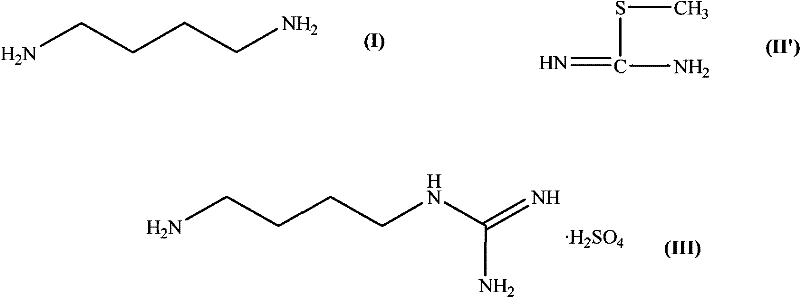
The Novel Approach
The invention disclosed in CN101717352A introduces a paradigm shift by utilizing O-methylisourea sulfate (II) as the guanidinylation reagent instead of its sulfur counterpart. This strategic substitution effectively eliminates the formation of malodorous and toxic mercaptans, replacing them with methanol or other benign alcohol byproducts depending on the specific reaction pathway and workup. The reaction proceeds smoothly in aqueous or alcoholic media at mild temperatures ranging from -20°C to 100°C, with a preferred window of 0°C to 40°C. This thermal flexibility allows for precise kinetic control, minimizing side reactions such as polymerization or over-alkylation. The use of O-methylisourea sulfate not only simplifies the waste treatment process—essentially achieving "zero three-waste" discharge regarding toxic sulfur emissions—but also enhances the overall reaction yield. By optimizing the molar ratio of 1,4-butanediamine to the guanidinylation agent between 1:1 and 1:2.0, manufacturers can achieve conversion rates significantly higher than the historical 40% benchmark, thereby maximizing throughput and resource utilization.
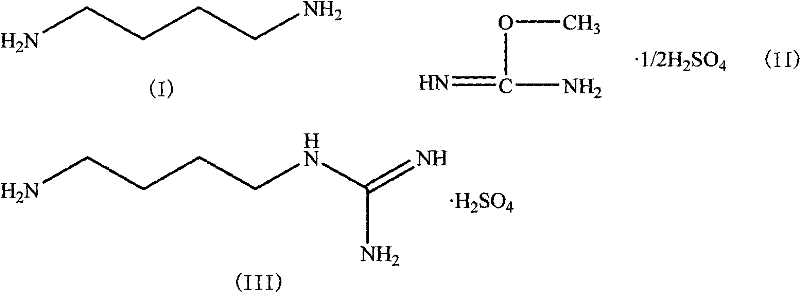
Mechanistic Insights into O-Methylisourea Mediated Guanidinylation
The core of this synthetic advancement lies in the nucleophilic substitution mechanism facilitated by the O-methylisourea cation. In this reaction, the primary amine group of 1,4-butanediamine acts as a potent nucleophile, attacking the electrophilic imino carbon of the O-methylisourea sulfate. The protonated imine structure activates the carbon center, making it highly susceptible to nucleophilic attack even under mild conditions. Unlike the sulfur analogue, where the C-S bond cleavage can be sluggish and prone to generating stable thiolate species that complicate the reaction matrix, the C-O bond cleavage in the O-methyl variant is kinetically favorable and thermodynamically driven towards the formation of the stable guanidine moiety. The sulfate counterion plays a dual role: it stabilizes the reactive isourea species during storage and handling, and subsequently participates in the salt formation of the final product. This mechanistic pathway ensures that the reaction proceeds with high selectivity, minimizing the formation of bis-guanidinated byproducts which are common impurities in diamine functionalization reactions.
Impurity control is further enhanced by the specific workup procedure outlined in the patent. Following the condensation, the reaction mixture is filtered to remove insoluble inorganic salts, and the pH is carefully adjusted to below 7.0 using a sulfuric acid-alcohol solution. This acidification step is critical; it ensures that the free base agmatine is fully converted into its sulfate salt form, which possesses distinct solubility characteristics compared to potential organic impurities. The subsequent concentration and recrystallization from water-ethanol mixtures exploit these solubility differences to exclude unreacted starting materials and minor side products. The choice of recrystallization solvent ratios (e.g., 40-60% water to 60-40% ethanol) is empirically optimized to maximize the recovery of the crystalline sulfate salt while keeping impurities in the mother liquor. This rigorous purification protocol is essential for producing reducing lead time for high-purity agmatine sulfates that meet the strict specifications required for downstream drug substance manufacturing.
How to Synthesize Agmatine Sulfate Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control to replicate the high yields reported in the patent examples. The process begins with the liberation of free 1,4-butanediamine from its hydrochloride salt, followed by the key condensation step with O-methylisourea sulfate. The operational simplicity of using water or ethanol as solvents makes this route particularly attractive for facilities aiming to reduce their reliance on hazardous organic solvents like dichloromethane or DMF. The detailed standardized synthesis steps below outline the precise conditions for reaction, isolation, and purification to ensure consistent batch-to-batch quality.
- Preparation of 1,4-Butanediamine: Liberating the free base from 1,4-butanediamine dihydrochloride using sodium hydroxide followed by distillation.
- Condensation Reaction: Reacting 1,4-butanediamine with O-methylisourea sulfate in water or ethanol at 0-40°C under magnetic stirring.
- Isolation and Purification: Adjusting pH below 7.0 with sulfuric acid/ethanol solution, concentrating, and recrystallizing from water-ethanol mixtures.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to the O-methylisourea sulfate route offers compelling economic and logistical benefits beyond mere chemical elegance. The elimination of sulfur-based waste streams translates directly into reduced operational expenditures related to environmental compliance and waste disposal. Traditional methods requiring mercaptan scrubbing involve significant capital investment in specialized equipment and ongoing maintenance costs, which are entirely avoided with this new methodology. Furthermore, the use of commodity solvents like water and ethanol enhances supply chain resilience, as these materials are globally available and less subject to the price volatility often seen in specialized halogenated solvents. The improved reaction yield means that less raw material is required per kilogram of finished product, driving down the cost of goods sold (COGS) and improving margin potential for high-volume contracts.
- Cost Reduction in Manufacturing: The economic argument for this process is anchored in the removal of expensive waste treatment protocols. By avoiding the generation of toxic alkyl mercaptans, manufacturers eliminate the need for complex odor control systems and hazardous waste incineration services. Additionally, the higher reaction yield observed in the patent examples implies a more efficient utilization of the 1,4-butanediamine starting material. This improved atom economy reduces the quantity of raw materials needed to produce a fixed amount of agmatine sulfate, resulting in substantial cost savings over the lifecycle of the product. The ability to use water as a primary solvent further reduces solvent purchase and recovery costs, contributing to a leaner manufacturing cost structure.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the availability of specialized reagents. O-methylisourea sulfate is a stable, commercially accessible reagent that does not suffer from the supply constraints often associated with sensitive sulfur compounds. The robustness of the reaction conditions—tolerating a wide temperature range and various solvent systems—means that production is less likely to be halted due to minor fluctuations in utility availability or cooling capacity. This operational flexibility ensures a steady output of high-purity agmatine sulfate, allowing downstream pharmaceutical partners to maintain their own production schedules without interruption. The simplified logistics of handling non-hazardous solvents also streamline the inbound supply chain.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production often exposes hidden safety and environmental risks. This patent's methodology is inherently scalable because it avoids exothermic hazards associated with unstable sulfur intermediates and utilizes benign solvents that are easy to handle in large reactors. The "green" nature of the process aligns perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing site against tighter emission standards. The absence of heavy metal catalysts or toxic sulfur byproducts simplifies the regulatory filing process for the API, as the impurity profile is cleaner and easier to characterize. This facilitates faster regulatory approval and market entry for drugs utilizing this intermediate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of agmatine sulfate. These answers are derived directly from the experimental data and claims within patent CN101717352A, providing a factual basis for evaluating the technology's viability. Understanding these nuances is vital for technical teams assessing the feasibility of adopting this route for commercial production.
Q: Why is O-methylisourea sulfate preferred over S-methylisothiourea for agmatine synthesis?
A: O-methylisourea sulfate eliminates the generation of toxic and malodorous alkyl mercaptans associated with S-methylisothiourea, significantly improving environmental compliance and operator safety while maintaining high reaction yields.
Q: What are the optimal solvent conditions for this guanidinylation reaction?
A: The patent specifies water or C1-C3 alcohols (methanol, ethanol) as ideal solvents. Water is particularly preferred for its cost-effectiveness and environmental safety, facilitating easier downstream processing.
Q: How does this process impact the purity profile of the final API intermediate?
A: By avoiding sulfur-based side reactions and utilizing a controlled pH adjustment followed by recrystallization, the process yields agmatine sulfate with superior purity specifications, minimizing difficult-to-remove impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Agmatine Sulfate Supplier
The synthesis of agmatine sulfate via O-methylisourea sulfate represents a mature, high-value opportunity for pharmaceutical development, yet translating patent chemistry into commercial reality requires deep process engineering expertise. NINGBO INNO PHARMCHEM stands ready to bridge this gap as your trusted partner. With extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, our technical team possesses the capability to optimize this specific route for maximum efficiency and purity. Our rigorous QC labs and commitment to stringent purity specifications ensure that every batch of agmatine sulfate meets the exacting standards required for global pharmaceutical markets. We understand that consistency is key, and our state-of-the-art facilities are designed to handle the specific solvent and temperature requirements of this guanidinylation process with precision.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain clear visibility into the potential economic advantages of switching to this cleaner, more efficient method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs. Let us collaborate to secure a sustainable and cost-effective supply of this critical neuroprotective intermediate.
