Advanced Agmatine Sulfate Manufacturing: Technical Insights for Global Procurement Leaders
Advanced Agmatine Sulfate Manufacturing: Technical Insights for Global Procurement Leaders
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally compliant synthetic routes for critical intermediates. Patent CN101717352B introduces a significant technological advancement in the production of Agmatine Sulfate, a compound with growing relevance in neuroprotection and metabolic health applications. This proprietary methodology replaces traditional, hazardous guanidinylation reagents with O-methylisourea sulfate, fundamentally altering the impurity profile and operational safety of the process. For R&D directors and procurement managers evaluating long-term supply contracts, understanding the mechanistic superiority of this route is essential for ensuring consistent quality and regulatory compliance. The following analysis details how this innovation translates into tangible commercial advantages, offering a pathway to cost reduction in pharmaceutical intermediates manufacturing while adhering to strict environmental standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
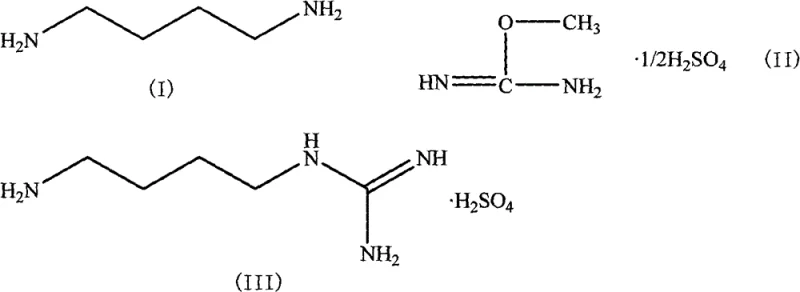
Historically, the industrial synthesis of Agmatine Sulfate relied heavily on the reaction between 1,4-butanediamine and S-methylisothiourea. While chemically feasible, this legacy approach presents severe operational bottlenecks that modern supply chains can ill afford. The primary drawback is the generation of alkyl mercaptans as stoichiometric byproducts, which are notorious for their intensely unpleasant odor and high toxicity. Handling these volatile sulfur compounds requires specialized containment infrastructure, extensive scrubbing systems, and rigorous personal protective equipment, all of which drive up capital expenditure and operational overhead. Furthermore, prior art indicates that this conventional route often suffers from mediocre reaction yields, typically hovering around 40%, which directly impacts material throughput and increases the cost of goods sold. The environmental burden of treating sulfur-containing waste streams also poses a significant compliance risk in regions with tightening emission regulations.
The Novel Approach
In stark contrast, the method disclosed in CN101717352B utilizes O-methylisourea sulfate as the guanidinylation agent, effectively bypassing the formation of noxious sulfur byproducts. This strategic substitution results in a much cleaner reaction profile, where the primary byproduct is methanol or a corresponding alcohol, both of which are far easier to manage and recover than mercaptans. The process operates under mild conditions, typically between 0°C and 40°C, allowing for precise thermal control without the need for extreme cryogenic cooling or high-energy heating. By eliminating the "three wastes" associated with sulfur chemistry, this novel approach not only simplifies the downstream purification workflow but also aligns perfectly with green chemistry principles. For a reliable agmatine sulfate supplier, adopting this technology means delivering a product with a superior impurity profile while drastically reducing the environmental footprint of the manufacturing facility.
Mechanistic Insights into O-Methylisourea Mediated Guanidinylation
The core of this synthetic innovation lies in the nucleophilic attack of the primary amine group of 1,4-butanediamine on the electrophilic carbon of the O-methylisourea moiety. Unlike the S-analog, the oxygen leaving group in O-methylisourea sulfate facilitates a smoother displacement reaction under neutral to slightly acidic conditions. The reaction mechanism proceeds through a tetrahedral intermediate that collapses to release methanol, driving the equilibrium toward the formation of the guanidine bond. This pathway is kinetically favorable and thermodynamically stable, which explains the observed improvement in conversion rates. From a process chemistry perspective, the absence of competing nucleophilic attacks by sulfur species minimizes the formation of thio-ether side products, thereby enhancing the overall purity of the crude reaction mixture before any purification steps are even initiated.
Impurity control is further enhanced by the specific choice of solvent systems and acidification protocols detailed in the patent. The use of water or lower alcohols (C1-C3) ensures high solubility of the ionic intermediates while maintaining a homogeneous reaction phase. Following the condensation, the addition of a sulfuric acid alcohol solution serves a dual purpose: it neutralizes excess amine and simultaneously converts the free base into the stable sulfate salt. This in-situ salt formation prevents the isolation of the hygroscopic free base, which can be difficult to handle on a large scale. The subsequent recrystallization from water-ethanol mixtures leverages the differential solubility of the product versus unreacted starting materials, yielding high-purity agmatine sulfate with a sharp melting point range of 234-236°C. This rigorous control over the crystallization process is critical for meeting the stringent specifications required by pharmaceutical customers.
How to Synthesize Agmatine Sulfate Efficiently
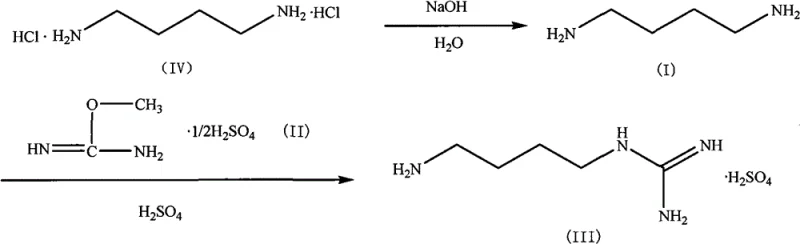
Implementing this synthesis route requires careful attention to the preparation of the diamine precursor and the stoichiometry of the guanidinylation step. The patent outlines a streamlined protocol where 1,4-butanediamine is generated in situ or purified via distillation from its hydrochloride salt before reacting with the O-methylisourea sulfate. Maintaining the molar ratio between the diamine and the guanidinating agent within the range of 1:1 to 1:2 is crucial for maximizing yield while minimizing residual starting materials. The reaction progress is monitored via Thin Layer Chromatography (TLC) to ensure complete consumption of the limiting reagent before proceeding to the workup phase. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis protocol below.
- Prepare 1,4-butanediamine by distilling its hydrochloride salt with sodium hydroxide under reduced pressure at 120°C.
- React 1,4-butanediamine with O-methylisourea sulfate in water or alcohol solvent at 0-40°C until TLC indicates completion.
- Filter the reaction mixture, adjust pH below 7.0 with sulfuric acid alcohol solution, concentrate, and recrystallize from water-ethanol mixture.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this O-methylisourea-based process offers compelling economic and logistical benefits beyond simple yield improvements. The elimination of malodorous and toxic sulfur byproducts removes the need for expensive waste treatment facilities and reduces the regulatory burden associated with hazardous air pollutants. This simplification of the environmental compliance landscape allows for faster permitting and more flexible manufacturing site selection. Furthermore, the use of commodity chemicals like water, ethanol, and sulfuric acid ensures that the supply chain is resilient to raw material shortages, unlike processes relying on specialized or scarce reagents. These factors collectively contribute to a more stable and predictable supply of commercial scale-up of complex pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven primarily by the simplification of the downstream processing train. By avoiding the generation of alkyl mercaptans, manufacturers save significantly on the capital and operational costs associated with scrubbing towers and specialized waste disposal. Additionally, the higher reaction efficiency reduces the amount of raw material required per kilogram of finished product, directly lowering the variable cost of production. The ability to use water as a primary solvent further reduces solvent purchase and recovery costs compared to processes requiring expensive organic solvents. These cumulative efficiencies result in a more competitive pricing structure for the final API intermediate without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for global pharmaceutical companies, and this synthesis route enhances reliability through the use of robust, non-hazardous reagents. The stability of O-methylisourea sulfate allows for easier storage and transportation compared to unstable isothiourea derivatives, reducing the risk of supply disruptions due to reagent degradation. Moreover, the mild reaction conditions (0-40°C) reduce the energy load on manufacturing facilities, making the process less susceptible to utility fluctuations. This operational robustness ensures that production schedules can be maintained consistently, reducing lead time for high-purity agmatine sulfates and enabling just-in-time delivery models for downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but this method is inherently scalable due to its homogeneous nature and lack of gas evolution. The absence of toxic gas release simplifies the engineering requirements for large-scale reactors, allowing for straightforward translation from pilot plant to multi-ton production. From an environmental standpoint, the "clean" nature of the reaction aligns with corporate sustainability goals and increasingly strict global environmental regulations. This future-proofs the supply chain against potential regulatory crackdowns on sulfur emissions, ensuring long-term viability of the manufacturing asset and securing the supply line for years to come.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Agmatine Sulfate using this advanced methodology. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature. Understanding these nuances helps stakeholders make informed decisions regarding vendor qualification and process validation. We encourage technical teams to review these points when assessing the feasibility of integrating this intermediate into their broader drug development pipelines.
Q: What are the primary advantages of using O-methylisourea sulfate over S-methylisothiourea?
A: The use of O-methylisourea sulfate eliminates the generation of toxic and malodorous alkyl mercaptans, significantly improving workplace safety and reducing environmental waste treatment costs compared to traditional S-methylisothiourea methods.
Q: What represents the optimal reaction temperature for this synthesis?
A: While the patent covers a range from -20°C to 100°C, experimental data suggests that mild temperatures between 0°C and 40°C provide the best balance of reaction rate and yield, avoiding the degradation observed at higher temperatures.
Q: Is this process scalable for industrial production?
A: Yes, the process utilizes common solvents like water and ethanol and avoids hazardous gas evolution, making it highly suitable for commercial scale-up with standard stainless steel reactor equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Agmatine Sulfate Supplier
At NINGBO INNO PHARMCHEM, we combine deep technical expertise with world-class manufacturing capabilities to deliver superior pharmaceutical intermediates. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the clinical trial phase or full commercial launch. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Agmatine Sulfate meets the highest industry standards. Our commitment to quality and consistency makes us the preferred partner for multinational corporations seeking a secure and compliant supply chain.
We invite you to engage with our technical procurement team to discuss your specific project needs. By leveraging our process optimization capabilities, we can provide a Customized Cost-Saving Analysis tailored to your volume and purity requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing technologies can add value to your supply chain.
