Advanced Tin-Free Valsartan Synthesis: Technical Breakthroughs and Commercial Scalability
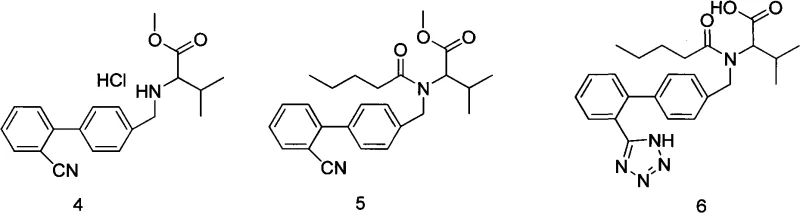
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with environmental safety, particularly for blockbuster antihypertensive agents like Valsartan. Patent CN101817795A introduces a significant technological advancement in this domain by providing an improved method for synthesizing Valsartan that completely eliminates the use of toxic tin compounds. This innovation addresses critical regulatory hurdles associated with heavy metal residues while simultaneously enhancing the overall yield and optical purity of the final Active Pharmaceutical Ingredient (API). By utilizing N-[(2'-cyano-1,1'-biphenyl-4-yl)alkyl]-L-valine ester hydrochloride as a key starting material, the process streamlines the conversion through pentaacylation, diazotization, and saponification steps. The strategic control of the crystallization process described in this patent ensures that the resulting Valsartan meets stringent quality specifications, offering a compelling solution for manufacturers aiming to optimize their production lines for both compliance and efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Valsartan has been plagued by significant challenges related to catalyst toxicity and purification complexity. The original preparation method disclosed in U.S. Patent 5399578A relies heavily on the use of tributyltin chloride as a catalyst for the tetrazole ring formation. While effective in driving the reaction, this approach introduces severe downstream processing issues because organotin compounds are highly toxic and persistent. According to strict International Council for Harmonisation (ICH) guidelines, residual organotin compounds in the finished pharmaceutical product must be controlled at extremely low levels, typically around 1 ppm. Removing these traces to meet such rigorous standards often requires expensive and complex purification steps, such as specialized scavenging resins or multiple recrystallizations, which drastically increase production costs and reduce overall throughput. Furthermore, alternative methods attempting to avoid tin, such as those described in Chinese Patent CN101270096A using triethylamine hydrochloride, have struggled with inconsistent product quality, lower yields, and difficulties in controlling optical isomerism, leaving a gap in the market for a truly optimized industrial process.
The Novel Approach
The methodology outlined in CN101817795A represents a decisive break from these conventional limitations by refining the triethylamine hydrochloride technology to make it suitable for large-scale industrial production. Instead of relying on hazardous tin catalysts, this novel approach utilizes a specific sequence involving valeryl acylation followed by a tetrazole formation step that leverages sodium azide and amine salts in a controlled environment. A distinguishing feature of this invention is the meticulous management of the post-reaction workup, where the organic layer is washed with specific acidic and basic solutions to remove impurities before the critical crystallization phase. By employing ester solvents for extraction and implementing a precise temperature-controlled crystallization protocol, the process effectively suppresses the formation and inclusion of unwanted optical isomers. This results in a high-purity Valsartan product that not only bypasses the regulatory nightmares associated with tin residues but also achieves superior yield and stereochemical integrity compared to previous iterations of tin-free synthesis.
Mechanistic Insights into Tin-Free Tetrazole Formation and Crystallization Control
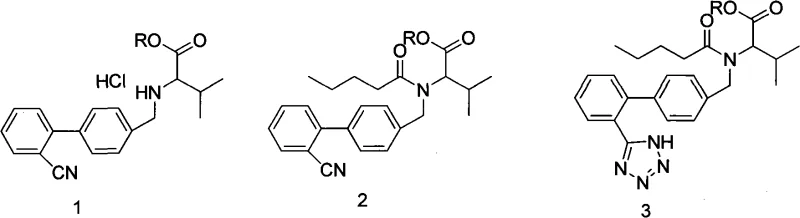
The core chemical innovation in this patent lies in the optimization of the tetrazole ring closure and the subsequent purification mechanics. The reaction begins with the acylation of the biphenyl valine ester hydrochloride using valeryl chloride in the presence of a carbonate base within an aromatic solvent system, typically toluene. This step generates the N-pentanoyl intermediate, which is then subjected to the critical cyclization reaction. Unlike traditional tin-catalyzed routes, this method employs sodium azide in conjunction with an amine salt, such as triethylamine hydrochloride, to facilitate the [2+3] cycloaddition required to form the tetrazole ring. The reaction mixture is refluxed for an extended period, ensuring complete conversion of the nitrile group to the tetrazole moiety. Following the reaction, the mixture undergoes a sophisticated workup involving treatment with alkaline and acidic solutions. This dual pH manipulation is crucial for partitioning the desired product into the organic phase while leaving inorganic salts and polar byproducts in the aqueous layer, thereby setting the stage for high-purity isolation.
Perhaps the most technically significant aspect of this process is the engineered crystallization mechanism designed to control chiral purity. After the organic layer is extracted with an ester solvent like isopropyl acetate and concentrated under reduced pressure, the solution is subjected to a multi-stage cooling regimen. The concentrate is first cooled to a moderate temperature range of 15°C to 35°C, inducing the initial nucleation of the product. This is followed by a holding period to allow crystal growth, and subsequently, a second cooling phase down to sub-zero temperatures between -10°C and 0°C. This gradient cooling strategy is thermodynamically designed to favor the crystallization of the desired enantiomer while keeping impurities and optical isomers in the mother liquor. The specific choice of ester solvents further enhances this selectivity due to their differential solubility profiles for the target molecule versus its stereoisomers. This precise control over the solid-state formation ensures that the final isolated Valsartan possesses an HPLC purity of greater than 99.8% and an optical isomer content of less than 0.2%, meeting the highest standards for pharmaceutical grade materials.
How to Synthesize Valsartan Efficiently
The synthesis of Valsartan via this improved tin-free route offers a practical and scalable pathway for manufacturing facilities aiming to produce high-quality API intermediates. The process is characterized by its reliance on readily available industrial reagents and solvents, avoiding the need for exotic or highly regulated catalysts. The operational procedure involves a straightforward sequence of acylation, cyclization, and controlled crystallization, which can be easily adapted to standard reactor setups found in most fine chemical plants. The detailed标准化 synthesis steps provided in the patent serve as a robust foundation for process engineers to develop Standard Operating Procedures (SOPs) that ensure batch-to-batch consistency. For a comprehensive breakdown of the specific reaction conditions, stoichiometry, and temperature profiles required to replicate this high-yield process, please refer to the technical guide below.
- Perform valeryl acylation on the starting biphenyl valine ester hydrochloride using valeryl chloride in an aromatic solvent with carbonate base.
- Execute tetrazole ring closure using sodium azide and an amine salt (e.g., triethylamine hydrochloride) followed by acid-base workup.
- Control crystallization through specific temperature gradients (concentration at 35-55°C, cooling to 15-35°C, then -10 to 0°C) to maximize optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthesis method described in CN101817795A translates into tangible strategic benefits regarding cost structure and risk mitigation. The elimination of tributyltin chloride from the manufacturing process removes a major bottleneck associated with heavy metal waste disposal and regulatory compliance testing. Traditional tin-based routes require expensive scavenging agents and rigorous analytical testing to prove that residual tin levels are within safe limits, adding significant time and cost to the production cycle. By switching to this tin-free methodology, manufacturers can drastically simplify their waste treatment protocols and reduce the burden on quality control laboratories, leading to substantial cost savings in the overall manufacturing budget. Furthermore, the improved yield and purity profile mean that less raw material is wasted, and the need for reprocessing or re-crystallization batches is minimized, directly enhancing the economic efficiency of the supply chain.
- Cost Reduction in Manufacturing: The removal of toxic tin catalysts eliminates the need for costly heavy metal scavengers and complex purification steps, significantly lowering the cost of goods sold (COGS). Additionally, the high yield reported in the patent examples indicates a more efficient utilization of starting materials, reducing raw material waste and maximizing output per batch. The use of common solvents like toluene and isopropyl acetate further ensures that solvent procurement remains stable and cost-effective, avoiding the price volatility associated with specialized reagents.
- Enhanced Supply Chain Reliability: By avoiding reagents that are subject to strict environmental regulations and handling restrictions, such as organotin compounds, the supply chain becomes more resilient and less prone to regulatory disruptions. The simplified process flow reduces the overall cycle time, allowing for faster turnaround from raw material intake to finished API, which is critical for meeting tight delivery schedules. The robustness of the crystallization process also ensures consistent product quality, reducing the risk of batch failures that could otherwise lead to supply shortages and contractual penalties.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, utilizing standard unit operations like liquid-liquid extraction and controlled cooling crystallization that are easily transferable from pilot plant to commercial production scales. From an environmental perspective, the absence of persistent organic pollutants like tin compounds simplifies effluent treatment and aligns with increasingly stringent global environmental, social, and governance (ESG) mandates. This green chemistry approach not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced Valsartan synthesis route. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the operational benefits and quality outcomes of the method. Understanding these details is essential for technical teams evaluating the feasibility of adopting this process for commercial manufacturing.
Q: Why is the removal of tin compounds critical in Valsartan synthesis?
A: Traditional methods using tributyltin chloride leave toxic organotin residues that are difficult to remove below ICH limits (1ppm). This tin-free route eliminates that regulatory burden entirely.
Q: How does the new crystallization process improve optical purity?
A: By utilizing a multi-stage cooling protocol with specific ester solvents, the process effectively suppresses the incorporation of optical isomers, achieving purity levels exceeding 99.8%.
Q: What are the scalability advantages of this method?
A: The process uses common industrial solvents like toluene and isopropyl acetate, avoids hazardous heavy metals, and offers high yields (approx. 90%), making it ideal for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Valsartan Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced synthetic methodologies to ensure the consistent supply of high-quality pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of Valsartan we produce meets or exceeds the global pharmacopoeia standards required by our international partners.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can drive value and reliability for your organization.
