Advanced Synthetic Route for Sofosbuvir Intermediate: Enhancing Purity and Scalability for Global Supply Chains
Advanced Synthetic Route for Sofosbuvir Intermediate: Enhancing Purity and Scalability for Global Supply Chains
The pharmaceutical landscape for Hepatitis C treatment has been revolutionized by nucleotide analogues, with Sofosbuvir standing as a cornerstone therapy. At the heart of manufacturing this critical antiviral agent lies the efficient production of its key precursor, 2R-2-deoxy-2-fluoro-2-methyl-D-erythropentonic acid-gamma-lactone-3, 5-dibenzoate. Patent CN109574961B, published in late 2020, introduces a transformative synthetic methodology that addresses long-standing inefficiencies in this supply chain. This technical insight report analyzes the novel four-step pathway detailed in the patent, which leverages ester-amine exchange, selective fluorination, and strategic rearrangement to achieve superior yields and purity profiles. For R&D directors and procurement strategists, understanding this route is essential for securing a reliable pharmaceutical intermediates supplier capable of meeting the rigorous demands of modern API manufacturing.
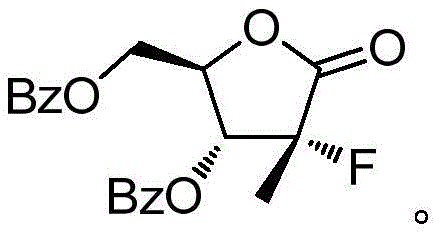
The patented process distinguishes itself by integrating a chiral auxiliary strategy that streamlines stereocontrol while minimizing waste. By initiating the synthesis with a cyclic sulfate derivative and employing 4-phenyl-2-oxazolidone, the method establishes the necessary chiral framework early in the sequence. This approach not only enhances the optical purity to greater than 98% but also simplifies downstream processing by avoiding complex chromatographic separations. As global demand for direct-acting antivirals remains robust, adopting such high-efficiency routes is critical for cost reduction in API manufacturing and ensuring the continuity of supply for life-saving medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of Sofosbuvir intermediates was plagued by low overall yields and cumbersome purification requirements, creating bottlenecks for commercial scale-up of complex pharmaceutical intermediates. Historical methods, such as those described by Wang et al., relied on R-glyceraldehyde acetonide and Wittig reactions, followed by oxidation and multiple protection-deprotection cycles. These routes often suffered from poor atom economy and generated significant impurities, necessitating expensive purification steps that drove up costs. Similarly, approaches by Mayers et al. utilizing six-membered lactone rings faced challenges with regioselectivity during fluorination and cyclization, resulting in total yields as low as 16.4% to 24.9%. Such inefficiencies translate directly into higher raw material consumption and extended production timelines, posing significant risks to supply chain stability.
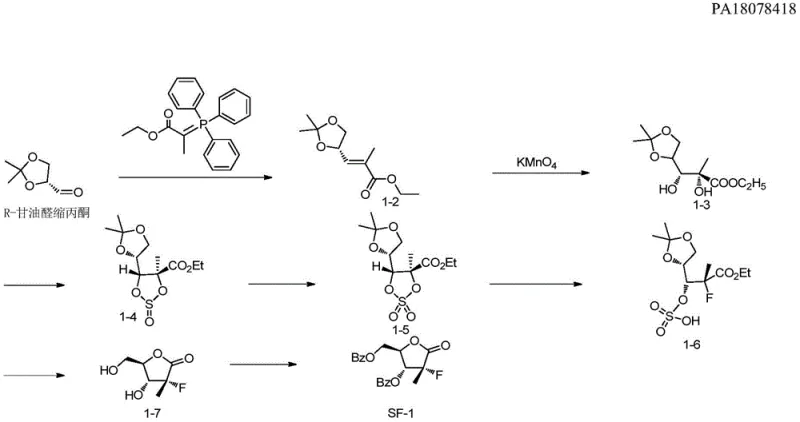
The reliance on harsh reagents and transition metal catalysts in older methodologies further complicated the impurity profile, often requiring specialized equipment for heavy metal removal. For procurement managers, these factors meant volatile pricing and longer lead times, as manufacturers struggled to optimize these inherently flawed processes. The accumulation of side products not only reduced the final yield but also increased the environmental burden of waste disposal, conflicting with modern green chemistry initiatives. Consequently, the industry has urgently required a paradigm shift towards more robust and linear synthetic strategies that can deliver high-purity materials without compromising on scalability or economic viability.
The Novel Approach
The methodology disclosed in patent CN109574961B offers a decisive break from these limitations by introducing a streamlined, four-step sequence that maximizes efficiency at every stage. By utilizing 2-C-methyl-4,5-O-(1-methylethylidene)-D-arabinonic acid ethyl ester cyclic sulfate as the starting material, the process bypasses the need for early-stage oxidations that typically degrade yield. The introduction of the chiral oxazolidone auxiliary allows for highly stereoselective functionalization, effectively solving the selectivity issues that plagued previous fluorination attempts. This new route achieves an impressive average total yield of 59-70%, representing a substantial improvement over the double-digit yields of the past. Furthermore, the reaction conditions are mild and utilize commercially available solvents like toluene and ethanol, facilitating easier technology transfer and reducing lead time for high-purity pharmaceutical intermediates.
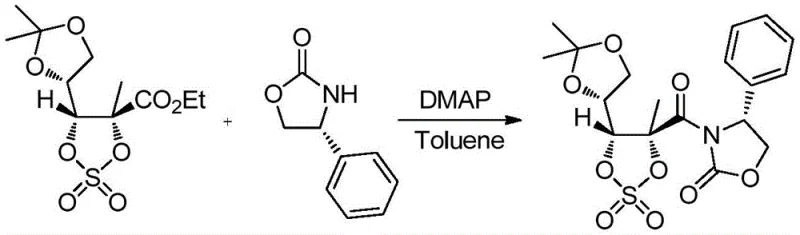
Crucially, this novel approach eliminates the need for chiral column chromatography, a major cost driver in fine chemical synthesis. The post-treatment steps involve standard aqueous workups and crystallization, which are far more amenable to multi-tonne production scales. The strategic use of tetraethylammonium fluoride for fluorination ensures high conversion rates with minimal elimination byproducts, preserving the integrity of the sugar backbone. For supply chain heads, this translates to a more predictable manufacturing schedule and a significant reduction in the cost of goods sold (COGS), enabling more competitive pricing strategies in the global marketplace.
Mechanistic Insights into the Multi-Step Cascade Synthesis
The success of this synthetic route lies in the precise orchestration of nucleophilic substitutions and rearrangement reactions. The process begins with a condensation reaction where the cyclic sulfate reacts with 4-phenyl-2-oxazolidone in the presence of 4-dimethylaminopyridine (DMAP). DMAP acts as a potent nucleophilic catalyst, activating the sulfate moiety for attack by the nitrogen of the oxazolidone. This step is conducted in toluene at temperatures between 60-80°C, optimizing the kinetic energy to drive the reaction to completion within 8-12 hours while maintaining thermal stability. The resulting first compound retains the stereochemical integrity of the starting material, setting the stage for the critical fluorination step.
Following condensation, the system undergoes a selective fluorination using tetraethylammonium fluoride (TEAF) in anhydrous 1,4-dioxane. The use of TEAF is pivotal; it provides a source of naked fluoride ions that can effectively displace the leaving group without inducing unwanted elimination reactions that would destroy the alkene or sugar framework. The reaction is heated to 105-115°C for 16-18 hours, ensuring complete conversion. Subsequent acid-catalyzed deprotection removes the acetonide protecting group, revealing the diol functionality required for the final lactonization. This tandem fluorination-deprotection sequence is a masterclass in step economy, combining two transformations into a single operational workflow to minimize handling losses.
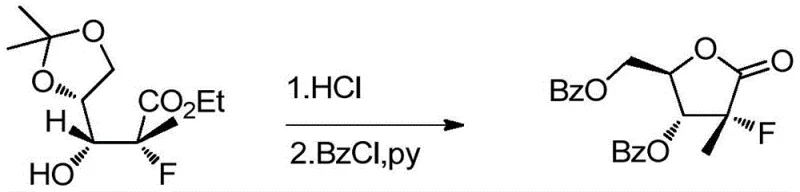
The final stages involve an ester exchange with sodium ethoxide followed by an acid-catalyzed rearrangement and benzoylation. The treatment with sodium ethoxide in absolute ethanol facilitates the migration of the ester group, preparing the molecule for cyclization. Upon addition of hydrochloric acid, the molecule undergoes a rearrangement to form the gamma-lactone ring, a thermodynamically stable structure characteristic of the Sofosbuvir core. Finally, benzoyl chloride is introduced under controlled temperatures (-2 to 30°C) to protect the hydroxyl groups as benzoates. This exothermic reaction is carefully managed to prevent over-benzoylation or degradation, yielding the final white crystalline intermediate with a melting point of 136°C and exceptional purity.
How to Synthesize Sofosbuvir Intermediate Efficiently
The synthesis of this critical antiviral intermediate requires strict adherence to the optimized reaction parameters outlined in the patent to ensure reproducibility and high yield. The process involves a sequential cascade of condensation, fluorination, ester exchange, and rearrangement-benzoylation, each demanding specific solvent systems and temperature controls. Operators must monitor reaction progress via TLC to determine precise endpoints, particularly during the fluorination and rearrangement steps where over-reaction can lead to impurity formation. The detailed standardized synthesis steps below provide a roadmap for implementing this high-efficiency route in a GMP-compliant environment.
- Condense 2-C-methyl-4,5-O-(1-methylethylidene)-D-arabinonic acid ethyl ester cyclic sulfate with 4-phenyl-2-oxazolidone using DMAP in toluene.
- Perform fluorination using tetraethylammonium fluoride followed by acid-catalyzed deprotection to introduce the fluoro group.
- React the resulting compound with sodium ethoxide to facilitate ester exchange and prepare for rearrangement.
- Execute acid-catalyzed rearrangement followed by benzoylation with benzoyl chloride to yield the final dibenzoate intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthesis method described in CN109574961B offers tangible strategic benefits beyond mere technical elegance. The primary value proposition lies in the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures. By eliminating the need for expensive chiral separation columns and reducing the number of unit operations, manufacturers can significantly lower the barrier to entry for large-scale production. This efficiency gain allows for a more agile response to market fluctuations, ensuring that critical API intermediates remain available even during periods of surging demand.
- Cost Reduction in Manufacturing: The most immediate impact of this new route is the substantial reduction in raw material and processing costs. By achieving total yields nearly three times higher than conventional methods, the consumption of starting materials per kilogram of final product is drastically decreased. Furthermore, the replacement of precious metal catalysts and complex oxidants with inexpensive reagents like DMAP and TEAF removes a significant line item from the bill of materials. The simplified workup procedures, which rely on standard extractions and crystallizations rather than preparative HPLC, also reduce solvent usage and energy consumption, leading to comprehensive cost optimization throughout the production lifecycle.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals and robust reaction conditions. The reliance on readily available solvents such as toluene, ethanol, and acetonitrile mitigates the risk of supply disruptions associated with specialty reagents. Additionally, the high stereoselectivity of the process ensures consistent quality output, reducing the incidence of batch failures and the need for re-processing. This reliability allows for tighter inventory management and shorter lead times, enabling pharmaceutical companies to maintain leaner stock levels without compromising on production schedules or regulatory compliance.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this method aligns perfectly with modern green chemistry principles. The reduction in side reactions means less hazardous waste generation, simplifying effluent treatment and lowering disposal costs. The process is inherently scalable, as demonstrated by the successful execution of the steps in standard laboratory glassware which can be directly translated to industrial reactors. The absence of toxic heavy metals and the use of benign workup procedures facilitate easier regulatory approval and support sustainable manufacturing goals, making it an attractive option for companies aiming to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical application of this technology for industrial partners.
Q: What is the primary advantage of this new synthesis method over prior art?
A: The primary advantage is a significant increase in total yield, reaching 59-70% compared to 16.4-24.9% in conventional methods, alongside reduced side reactions and simplified purification.
Q: How does the process ensure high stereoselectivity?
A: The process utilizes a chiral auxiliary (4-phenyl-2-oxazolidone) during the initial condensation and fluorination steps, which effectively controls stereochemistry and eliminates the need for chiral chromatographic separation.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the method uses common solvents like toluene and ethanol, avoids expensive transition metal catalysts, and features robust post-treatment steps, making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sofosbuvir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to production requires a partner with deep technical expertise and unwavering commitment to quality. Our team of process chemists has extensively analyzed the route disclosed in CN109574961B and is fully prepared to adapt this methodology for your specific manufacturing needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity specifications achieved in the lab are faithfully reproduced at the plant scale. Our rigorous QC labs employ state-of-the-art analytical instrumentation to verify every batch against stringent purity specifications, guaranteeing that the Sofosbuvir intermediate you receive meets the highest global standards.
We invite you to collaborate with us to leverage this advanced synthetic technology for your antiviral drug programs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this efficient route can improve your bottom line. We encourage you to reach out today to request specific COA data and discuss route feasibility assessments for your upcoming projects, securing a competitive edge in the rapidly evolving pharmaceutical market.
