Advanced Manufacturing of Fluvastatin Intermediates via Novel Indole Derivative Processes
The pharmaceutical industry continuously seeks robust synthetic pathways for high-value active pharmaceutical ingredients (APIs), particularly for cardiovascular treatments like HMG-CoA reductase inhibitors. Patent CN1217930C presents a significant technological advancement in the preparation of indole derivatives, specifically targeting intermediates essential for the synthesis of Fluvastatin. This document outlines a comprehensive process that addresses historical challenges in yield optimization and stereochemical control, offering a viable route for the commercial scale-up of complex pharmaceutical intermediates. By leveraging novel reduction techniques and coupling strategies, this methodology ensures the production of high-purity compounds necessary for meeting stringent regulatory standards in global markets.
The core innovation lies in the strategic manipulation of the indole nucleus and the subsequent attachment of the heptenoic acid side chain with precise stereochemistry. Traditional methods often struggle with the formation of unwanted diastereomers or require expensive chiral auxiliaries that complicate downstream processing. In contrast, the approach detailed in CN1217930C utilizes a sequence involving Vilsmeier-Haack formylation, followed by condensation and specialized borane-mediated reductions. This not only streamlines the synthetic route but also enhances the overall economic feasibility of producing these critical pharmaceutical intermediates on an industrial scale.
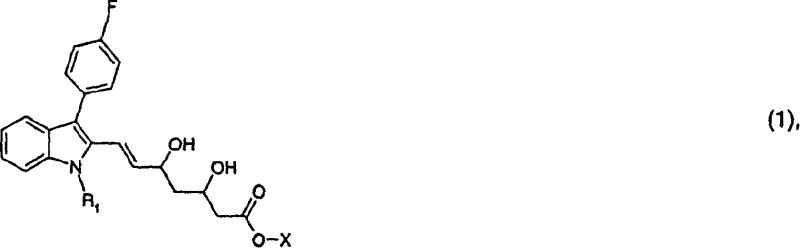
For procurement specialists and supply chain managers, understanding the nuances of this synthetic pathway is vital for securing a reliable pharmaceutical intermediates supplier. The process described minimizes the reliance on exotic reagents, favoring commercially available starting materials such as 3-(4-fluorophenyl)-1-isopropylindole and standard esters. This shift towards accessible raw materials directly contributes to cost reduction in pharmaceutical intermediates manufacturing by stabilizing input costs and reducing lead times associated with sourcing specialized chemicals. Furthermore, the robustness of the reaction conditions suggests a high degree of reproducibility, which is a cornerstone of supply chain continuity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing Fluvastatin intermediates often relied on multi-step sequences that suffered from cumulative yield losses and poor stereochemical outcomes. Conventional reduction methods using simple metal hydrides frequently resulted in mixtures of diastereomers, necessitating costly and time-consuming purification steps such as preparative HPLC or repeated crystallizations. These inefficiencies not only drove up the cost of goods sold (COGS) but also introduced variability in the final product quality, posing risks for regulatory compliance. Additionally, older routes sometimes employed harsh reaction conditions or unstable intermediates that were difficult to handle safely in large-scale reactors, limiting their practical application for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
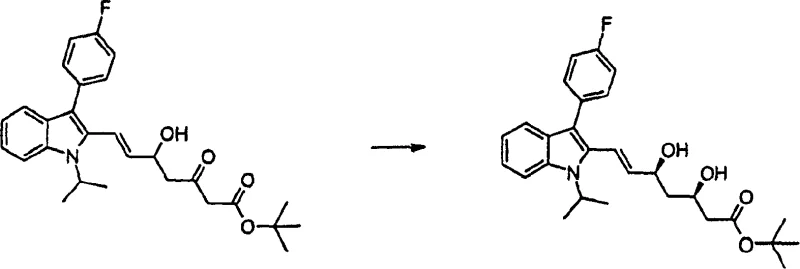
The methodology disclosed in CN1217930C overcomes these hurdles through a refined sequence that prioritizes both chemoselectivity and stereoselectivity. By introducing specific protecting groups and utilizing controlled reduction environments, the process effectively directs the formation of the desired (3R, 5S) configuration. For instance, the use of diethylmethoxyborane in conjunction with sodium borohydride allows for a highly selective reduction of the ketone functionality without affecting other sensitive parts of the molecule. This precision eliminates the need for extensive downstream purification, thereby simplifying the workflow. The integration of Heck coupling reactions for side-chain attachment further demonstrates the versatility of this approach, enabling the construction of the carbon skeleton with high fidelity and minimal byproduct formation.
Mechanistic Insights into Stereoselective Reduction and Coupling
A deep dive into the reaction mechanics reveals why this process is superior for R&D teams focused on impurity control. The reduction of the beta-keto ester or enone intermediates is the critical step where stereochemistry is established. The patent describes a mechanism where the borane species coordinates with the carbonyl oxygen, creating a rigid transition state that favors hydride delivery from a specific face. This chelation-controlled reduction is far more effective than non-directed hydride attacks, which occur randomly. By maintaining reaction temperatures between -78°C and 0°C, the kinetic control is maximized, ensuring that the thermodynamic product does not dominate. This level of control is essential for achieving the high enantiomeric excess required for bioactive molecules.
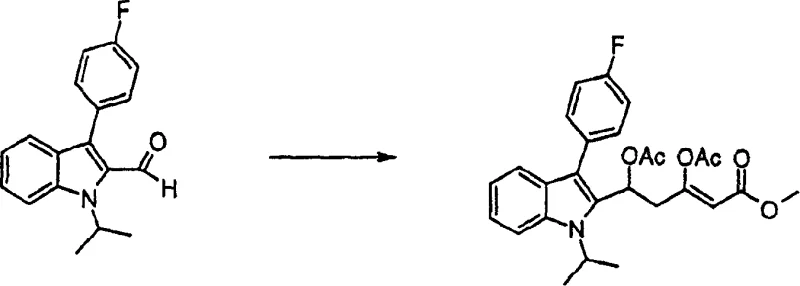
Furthermore, the coupling reactions described, such as the palladium-catalyzed Heck reaction, proceed through a well-defined catalytic cycle involving oxidative addition, migratory insertion, and beta-hydride elimination. The choice of ligands and bases, as detailed in the examples, optimizes the turnover number of the catalyst, reducing the residual metal content in the final API. This is a crucial consideration for high-purity pharmaceutical intermediates, as heavy metal limits are strictly enforced by health authorities. The ability to tune the electronic properties of the indole ring through substituents like the 4-fluorophenyl group also enhances the reactivity of the coupling partner, leading to faster reaction rates and higher conversions. Understanding these mechanistic details allows process chemists to troubleshoot potential scale-up issues proactively.
How to Synthesize Fluvastatin Intermediate Efficiently
The synthesis of these advanced indole derivatives requires precise adherence to the reaction parameters outlined in the patent to ensure optimal yield and purity. The process begins with the formylation of the indole core, followed by chain extension and stereoselective reduction. Each step demands careful monitoring of temperature and stoichiometry to prevent side reactions. For a detailed breakdown of the operational procedures, including specific solvent grades and workup protocols, please refer to the standardized guide below which summarizes the critical unit operations derived from the patent examples.
- Perform Vilsmeier-Haack formylation on 3-(4-fluorophenyl)-1-isopropylindole using DMF and phosphorus oxychloride at controlled temperatures.
- Execute a condensation reaction with methyl acetoacetate followed by elimination to form the unsaturated side chain precursor.
- Conduct stereoselective reduction using sodium borohydride and diethylmethoxyborane to establish the critical (3R, 5S) diol configuration.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers tangible benefits that extend beyond the laboratory bench. For procurement managers, the shift towards more stable and readily available reagents translates into a more resilient supply chain. The elimination of hard-to-source chiral catalysts in favor of stoichiometric borane reductions simplifies the vendor landscape, reducing the risk of supply disruptions. Moreover, the improved yield profile means that less raw material is required to produce the same amount of finished intermediate, driving down the overall material cost. This efficiency is critical in a competitive market where margin pressure is constant, allowing companies to maintain profitability while adhering to quality standards.
- Cost Reduction in Manufacturing: The streamlined process significantly lowers manufacturing costs by reducing the number of purification steps required. Traditional methods often necessitate multiple chromatographic separations to remove diastereomeric impurities, which are expensive and slow. By achieving high stereocontrol directly in the reaction vessel, this method minimizes waste and solvent consumption. The use of common solvents like THF, toluene, and ethyl acetate further reduces operational expenses compared to processes requiring specialized or hazardous solvents. Additionally, the higher throughput per batch due to improved yields means that fixed costs such as labor and equipment depreciation are amortized over a larger volume of product.
- Enhanced Supply Chain Reliability: Supply chain stability is bolstered by the use of robust intermediates that can be stored and transported without significant degradation. The intermediates described, such as the bromo-indole derivatives, are chemically stable, allowing for strategic stockpiling to buffer against market fluctuations. The process does not rely on single-source suppliers for exotic catalysts, diversifying the supply base and mitigating geopolitical risks. Furthermore, the scalability of the reactions ensures that production can be ramped up quickly to meet sudden increases in demand without compromising quality, providing a competitive edge in reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The environmental footprint of this process is reduced through efficient atom economy and minimized waste generation. The avoidance of heavy metal catalysts in the final reduction steps simplifies wastewater treatment and reduces the burden of hazardous waste disposal. The reactions are designed to operate at concentrations that are safe for large-scale reactors, preventing thermal runaways and ensuring operator safety. This alignment with green chemistry principles not only meets regulatory requirements but also enhances the corporate sustainability profile, which is increasingly important for stakeholders and customers alike. The ability to recycle solvents and recover byproducts adds another layer of economic and environmental value.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and claims found within the patent documentation, providing clarity on the feasibility and advantages of the method. Understanding these aspects is crucial for technical teams evaluating the transfer of this technology to pilot or production scales.
Q: What is the key advantage of the reduction method in CN1217930C?
A: The patent utilizes a borane-mediated reduction strategy that offers superior stereocontrol for generating the (3R, 5S) diol motif compared to traditional hydride reductions, minimizing diastereomeric impurities.
Q: How does this process improve supply chain stability?
A: By employing robust intermediates like the 2-bromo-indole derivative and avoiding unstable transition metal catalysts in later stages, the process ensures consistent batch-to-batch quality and reduces dependency on scarce reagents.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the described reactions utilize common solvents like THF and toluene and operate at manageable temperature ranges (-78°C to reflux), making the protocol highly adaptable for kilogram to metric ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluvastatin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN1217930C can be translated into reliable manufacturing reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Fluvastatin intermediate meets the exacting standards required by global regulatory bodies. Our commitment to technical excellence ensures that your supply chain remains uninterrupted and compliant.
We invite you to collaborate with us to optimize your production costs and secure your supply of critical materials. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in indole derivative synthesis can add value to your pharmaceutical portfolio. Together, we can drive efficiency and innovation in the production of essential cardiovascular therapies.
