Optimizing Atorvastatin Intermediate Production: A Technical Breakdown of CN102127060B
Optimizing Atorvastatin Intermediate Production: A Technical Breakdown of CN102127060B
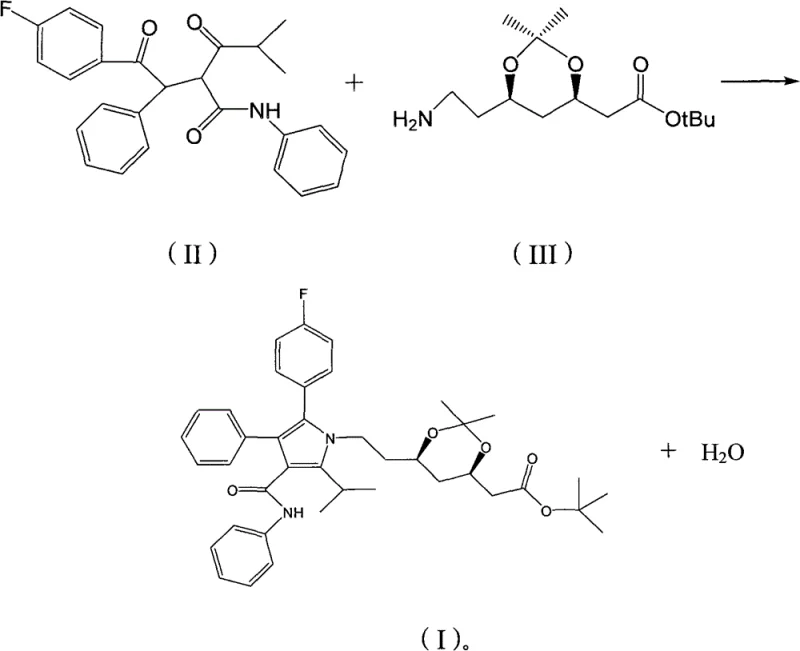
The pharmaceutical industry constantly seeks robust, scalable, and cost-effective routes for the production of statin intermediates, which are critical building blocks for cardiovascular medications. Patent CN102127060B presents a significant technological advancement in the preparation of a key atorvastatin calcium intermediate, specifically the compound designated as Formula (I). This innovation addresses longstanding inefficiencies in the condensation reaction between the pyrrole derivative (M4) and the dioxane side chain (ATS-9). By fundamentally re-engineering the solvent system and the water removal mechanism, this method offers a pathway to higher yields and drastically reduced cycle times compared to traditional United States Patent methodologies. For R&D directors and process chemists, understanding the nuances of this solvent switch is vital for modernizing manufacturing lines.
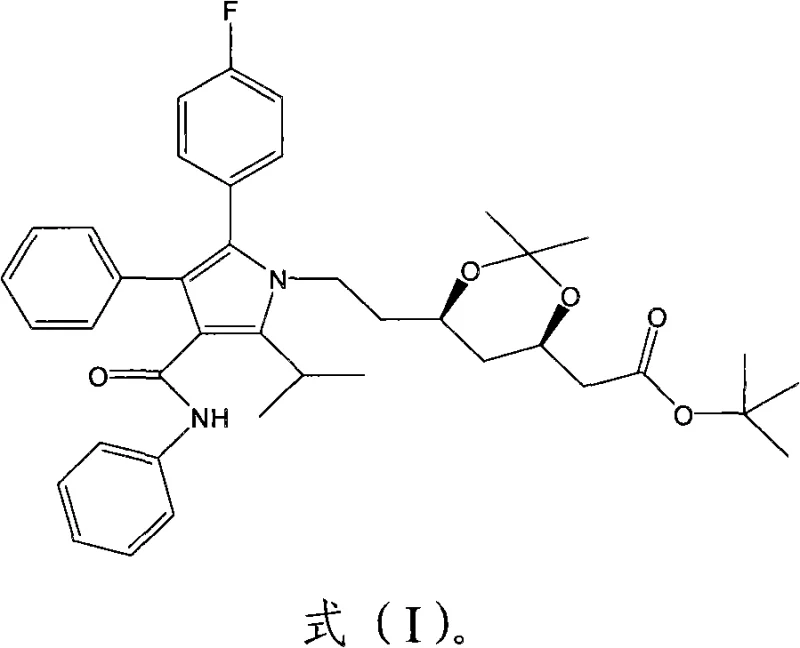
The core of this invention lies in the structural integrity and purity of the final intermediate, which serves as a precursor for one of the world's most prescribed lipid-lowering agents. The compound, chemically known as (4R-cis)-6-[2-[2-(4-fluorophenyl)-5-(1-methylethyl)-3-phenyl-4-[(phenylamino)carbonyl]-1H-pyrrol-1-yl]ethyl]-2,2-dimethyl-1,3-dioxane-4-acetic acid 1,1-dimethylethyl ester, requires precise stereochemical control. The patent details a method that not only preserves the chiral integrity of the side chain but also ensures that the final crystalline form meets stringent quality specifications required for GMP manufacturing. This level of control is essential for any reliable pharmaceutical intermediate supplier aiming to support global generic drug production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, particularly those documented in various United States Patents such as 5,003,080 and 5,273,995, typically rely on a ternary solvent system comprising toluene, n-heptane, and tetrahydrofuran (THF). While effective to a degree, the inclusion of THF introduces a significant thermodynamic bottleneck. Because THF is completely miscible with water, it forms a homogeneous phase with the water byproduct generated during the acid-catalyzed condensation. This miscibility prevents the efficient physical separation of water from the reaction mixture, thereby hindering the shift of the chemical equilibrium toward the desired product. Consequently, these legacy processes often require extended reaction times ranging from 22 to 40 hours to reach completion, leading to excessive energy consumption and potential degradation of sensitive functional groups.
The Novel Approach
The methodology disclosed in CN102127060B overcomes these thermodynamic barriers by eliminating THF entirely and utilizing a binary solvent system of toluene and n-heptane. This strategic modification ensures that the solvent mixture is immiscible with water, facilitating rapid phase separation. When coupled with a specialized constant-temperature reflux water-dividing device, the system allows for the continuous and efficient removal of the water byproduct via azeotropic distillation. This drives the reaction equilibrium strongly to the right, in accordance with Le Chatelier's principle, allowing the reaction to reach completion in merely 12 to 14 hours. This represents a substantial improvement in process intensity and throughput capability.
Mechanistic Insights into Acid-Catalyzed Condensation and Water Removal
The reaction mechanism involves the nucleophilic attack of the amine group on the ATS-9 side chain onto the carbonyl or activated species of the M4 pyrrole derivative, catalyzed by pivalic acid. The efficiency of this transformation is heavily dependent on the removal of the stoichiometric water produced. In the novel process, the use of a glass bead tube within the reflux apparatus plays a critical mechanistic role. This device increases the surface area for heat exchange and vapor-liquid contact within the column. As the solvent vapors rise, they undergo partial condensation and separation within the bead-packed section before reaching the water trap. This ensures that only the water-rich azeotrope is removed, while the organic solvents are returned to the reaction flask, maintaining a constant concentration of reactants and catalyst throughout the 110-120°C reflux period.
Furthermore, the specific ratio of toluene to n-heptane (optimized at 1:4) is crucial for maintaining the correct boiling point and phase behavior. Toluene provides the necessary solvating power for the aromatic reactants, while n-heptane acts as a non-polar diluent that aids in the precipitation of water and helps tune the boiling point of the azeotrope. The stepwise addition of the pivalic acid catalyst—adding roughly half at the start and the remainder after 50% conversion—prevents localized high acidity that could lead to side reactions or decomposition of the acid-sensitive dioxane ring. This controlled addition strategy is a key factor in achieving the reported high purity levels exceeding 98% as detected by gas chromatography.
How to Synthesize Atorvastatin Calcium Intermediate Efficiently
Implementing this synthesis requires precise adherence to the solvent ratios and temperature controls outlined in the patent data. The process begins with the dissolution of the M4 and ATS-9 reactants in the optimized toluene/n-heptane mixture, followed by the initial charge of the pivalic acid catalyst. The reaction is then heated to a vigorous reflux, typically between 110°C and 120°C, utilizing the specialized water-dividing apparatus to continuously strip water from the system. Monitoring via TLC or HPLC is essential to determine the optimal point for the second catalyst addition and solvent swap, ensuring maximum conversion before proceeding to the workup phase which involves washing, drying, and recrystallization.
- Dissolve reactants M4 (Formula II) and ATS-9 (Formula III) in a mixed solvent of toluene and n-heptane.
- Add pivalic acid catalyst and heat to reflux (110-120°C) using a constant-temperature reflux water-dividing device.
- Monitor reaction progress, remove generated water azeotropically, and recrystallize the final product from isopropanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this improved synthetic route offers compelling economic and logistical benefits. The most immediate impact is the drastic reduction in reactor occupancy time. By shortening the reaction cycle from nearly two days to roughly half a day, manufacturing facilities can significantly increase their batch turnover rate without requiring additional capital investment in new reactors. This intensification of the process directly translates to lower fixed costs per kilogram of product, enhancing the overall margin profile for the intermediate. Additionally, the elimination of THF simplifies the solvent recovery infrastructure, as there is no need to manage the complex separation of water-miscible organics.
- Cost Reduction in Manufacturing: The economic advantages are driven primarily by energy efficiency and solvent management. Since the reaction time is significantly shortened, the energy required for heating and stirring is substantially reduced. Furthermore, the use of toluene and n-heptane, which are commodity chemicals with stable pricing and widespread availability, mitigates supply risk compared to more specialized solvent blends. The high yield reported in the embodiments suggests that raw material utilization is optimized, minimizing waste disposal costs associated with unreacted starting materials and byproducts.
- Enhanced Supply Chain Reliability: From a sourcing perspective, the reliance on common industrial solvents like toluene and n-heptane ensures a robust supply chain that is less susceptible to regional shortages or regulatory restrictions often associated with ether-based solvents. The simplicity of the workup procedure, which avoids complex extractions required for THF removal, also reduces the dependency on specialized downstream processing equipment. This reliability is critical for maintaining continuous production schedules and meeting the Just-In-Time delivery expectations of major pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard reflux and distillation unit operations that are well-understood in chemical engineering. The efficient removal of water reduces the volume of aqueous waste streams generated during the reaction phase. Moreover, the high purity of the crude product prior to recrystallization implies that fewer purification cycles are needed, reducing the overall solvent footprint and aligning with green chemistry principles by minimizing the E-factor of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and validation of this synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the operational parameters and quality outcomes expected from this specific route. Understanding these details is crucial for technology transfer teams evaluating the feasibility of adopting this process.
Q: Why is the removal of THF critical in this synthesis?
A: Tetrahydrofuran (THF) is miscible with water, which hinders the separation of the water byproduct generated during the condensation reaction. By replacing THF with a water-immiscible system like toluene and n-heptane, the reaction equilibrium is driven forward more efficiently.
Q: What represents the primary cost advantage of this method?
A: The primary cost advantage stems from the significant reduction in reaction time (from roughly 40 hours in prior art to 12-14 hours) and the elimination of expensive or difficult-to-remove solvents, leading to lower energy consumption and simplified downstream processing.
Q: How is high purity achieved in the final product?
A: High purity (>98%) is achieved through a combination of efficient water removal during the reaction to prevent side reactions, followed by a rigorous workup involving washing, activated carbon decolorization, and recrystallization from isopropanol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Atorvastatin Calcium Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the global pharmaceutical supply chain. Our technical team has extensively analyzed the methodology described in CN102127060B and possesses the expertise to implement this optimized route effectively. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant and finally to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of atorvastatin intermediate meets the exacting standards required for API synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis method can be integrated into your supply chain. By leveraging our manufacturing capabilities, you can secure a stable source of high-purity intermediates while benefiting from the cost efficiencies inherent in this improved process. Please contact us to request a Customized Cost-Saving Analysis, specific COA data from our recent batches, and comprehensive route feasibility assessments tailored to your project requirements.
