Revolutionizing Aromatic Amine Synthesis: A Ligand-Free Zinc Complex Approach for Industrial Scale
Revolutionizing Aromatic Amine Synthesis: A Ligand-Free Zinc Complex Approach for Industrial Scale
The synthesis of aromatic amines represents a cornerstone capability in the modern fine chemical industry, serving as critical building blocks for active pharmaceutical ingredients (APIs), agrochemicals, and advanced functional materials. Historically, the construction of the aromatic carbon-nitrogen (C-N) bond has been fraught with challenges ranging from expensive catalytic systems to harsh reaction conditions that compromise atom economy. However, a significant technological breakthrough documented in patent CN102134176A introduces a novel methodology utilizing organic amine zinc chloride complexes. This innovation provides a robust, ligand-free pathway for converting halogenated aromatic hydrocarbons directly into valuable arylamine compounds. For global procurement and R&D leaders, this technology signals a paradigm shift away from reliance on precious transition metals like palladium towards more abundant, cost-effective, and environmentally benign zinc-based reagents. The implications for supply chain stability and manufacturing cost structures are profound, offering a sustainable route for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional strategies for synthesizing aromatic amines have long been dominated by three primary methodologies, each carrying distinct operational burdens that hinder efficient industrial application. The classical N-alkylation method, often exemplified by the Eschweiler-Clarke reaction, typically requires a two-step sequence involving nitration followed by reduction, which suffers from poor atom economy and generates significant waste streams. Furthermore, controlling selectivity in N-alkylation is notoriously difficult, often leading to mixtures of secondary and tertiary amines that require energy-intensive separation processes. Alternatively, direct nucleophilic aromatic substitution (SNAr) is heavily dependent on the presence of strong electron-withdrawing groups on the aromatic ring to activate the leaving group, severely limiting the scope of accessible substrates. 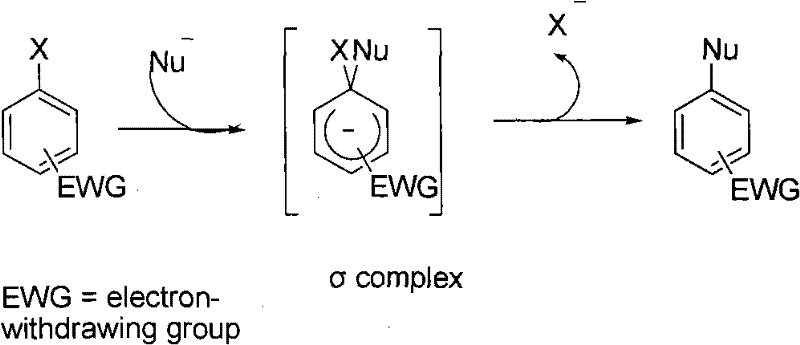 While transition metal-catalyzed cross-coupling reactions, such as the Buchwald-Hartwig amination, expanded the scope of accessible substrates, they introduced new bottlenecks related to the high cost of palladium catalysts and the toxicity of phosphine ligands.
While transition metal-catalyzed cross-coupling reactions, such as the Buchwald-Hartwig amination, expanded the scope of accessible substrates, they introduced new bottlenecks related to the high cost of palladium catalysts and the toxicity of phosphine ligands. 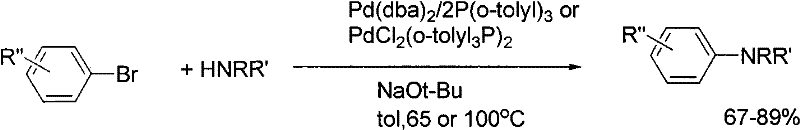 The removal of trace palladium residues to meet stringent pharmaceutical purity specifications adds substantial downstream processing costs, while the sensitivity of these catalysts to air and moisture complicates reactor operations and increases the risk of batch failures.
The removal of trace palladium residues to meet stringent pharmaceutical purity specifications adds substantial downstream processing costs, while the sensitivity of these catalysts to air and moisture complicates reactor operations and increases the risk of batch failures.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in patent CN102134176A leverages pre-formed organic amine zinc chloride complexes to drive the amination of halogenated aromatics with exceptional efficiency. This approach fundamentally alters the reaction landscape by utilizing a solid-state amination reagent that is stable, non-volatile, and easy to handle, effectively mitigating the safety hazards associated with free liquid or gaseous amines. The reaction proceeds smoothly in common polar aprotic solvents such as dimethyl sulfoxide (DMSO) or toluene, utilizing inexpensive inorganic bases like potassium carbonate or cesium carbonate. Crucially, this method operates without the need for external ligands or expensive transition metals, thereby simplifying the reaction mixture and facilitating easier product isolation. The general reaction scheme demonstrates the versatility of this approach across a wide range of aryl halides, including those substituted with nitro, trifluoromethyl, and aldehyde groups, which are often sensitive to harsher reducing conditions found in traditional routes. 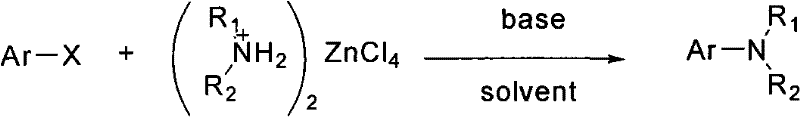 By bypassing the need for noble metals and complex ligand systems, this novel approach offers a streamlined, green chemistry solution that aligns perfectly with modern sustainability goals and cost reduction imperatives in fine chemical manufacturing.
By bypassing the need for noble metals and complex ligand systems, this novel approach offers a streamlined, green chemistry solution that aligns perfectly with modern sustainability goals and cost reduction imperatives in fine chemical manufacturing.
Mechanistic Insights into Zinc-Mediated C-N Bond Formation
The mechanistic underpinning of this zinc-mediated amination represents a fascinating departure from standard palladium-catalyzed cycles. While the precise electronic interactions are still a subject of academic inquiry, the prevailing understanding suggests that the zinc center in the amine zinc chloride complex acts as a Lewis acid activator. It likely coordinates with the halogenated aromatic substrate or stabilizes the developing negative charge on the nitrogen nucleophile during the transition state, thereby lowering the activation energy for the C-N bond formation. Unlike the oxidative addition and reductive elimination steps characteristic of Pd(0)/Pd(II) cycles, this zinc-mediated process appears to follow a more direct nucleophilic substitution pathway that is facilitated by the unique electronic environment provided by the zinc chloride lattice. This mechanism allows for the activation of aryl chlorides and fluorides, which are typically inert in uncatalyzed SNAr reactions unless activated by very strong electron-withdrawing groups. The absence of phosphine ligands eliminates the possibility of ligand-induced side reactions or catalyst deactivation pathways, resulting in a cleaner reaction profile with fewer byproducts. This mechanistic simplicity translates directly to higher reproducibility and robustness when scaling from gram-scale laboratory experiments to multi-ton commercial production runs.
From an impurity control perspective, the use of zinc complexes offers distinct advantages over traditional amine sources. Free amines, particularly low molecular weight alkyl amines, are prone to over-alkylation or oxidation, leading to complex impurity profiles that are difficult to purge. By locking the amine into a stable zinc complex, the reactivity is modulated, ensuring that the nucleophilic attack occurs selectively at the desired aromatic carbon center. Furthermore, the zinc byproducts generated during the reaction are generally inorganic salts that are water-soluble and easily removed during the aqueous workup phase, unlike organotin byproducts from Stille-type couplings or palladium black which can colloidalize and contaminate the product. This inherent cleanliness of the reaction mechanism significantly reduces the burden on downstream purification units such as chromatography columns or recrystallization tanks. For R&D directors focused on impurity fate and purge studies, this technology provides a clearer path to meeting ICH Q3 guidelines for residual metals and organic impurities, ultimately accelerating the regulatory approval timeline for new drug candidates derived from these intermediates.
How to Synthesize Aromatic Amines Efficiently
Implementing this synthesis route in a pilot or production facility requires careful attention to reagent stoichiometry and thermal management, although the overall protocol is remarkably straightforward. The process begins with the precise weighing of the halogenated aromatic hydrocarbon and the specific organic amine zinc chloride complex, typically in a molar ratio ranging from 1:1 to 1:10 depending on the reactivity of the halide. These solids are charged into a reactor along with a suitable solvent system, with dimethyl sulfoxide (DMSO) often proving optimal for solubilizing the zinc complex while maintaining thermal stability. A base, such as potassium carbonate, is then introduced to scavenge the hydrogen halide byproduct and drive the equilibrium toward the desired amine product. The detailed standardized synthesis steps, including specific temperature ramps and workup procedures validated in the patent examples, are outlined below to ensure consistent quality and yield.
- Mix halogenated aromatic hydrocarbon and organic amine zinc chloride complex in a molar ratio of 1: 1 to 1:10 within a reaction vessel.
- Add a solvent such as DMSO or toluene (1 to 10 times the volume of the halogenated hydrocarbon) and a base like potassium carbonate.
- Heat the mixture to 60°C-200°C, monitor via TLC, and upon completion, extract and purify the target aromatic amine compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this zinc-complex mediated amination technology presents a compelling value proposition centered on cost predictability and operational resilience. The most immediate impact is the drastic reduction in raw material expenditure associated with eliminating palladium catalysts and specialized phosphine ligands from the bill of materials. Precious metal prices are subject to extreme volatility and geopolitical supply risks, whereas zinc is an abundant, base metal with a stable and transparent global market price. This substitution not only lowers the direct cost of goods sold but also hedges the manufacturing process against future spikes in noble metal costs. Additionally, the use of solid amine zinc complexes simplifies logistics and storage; unlike volatile liquid amines that require specialized containment and ventilation systems to manage odor and flammability, these solid reagents can be stored in standard warehouse conditions, reducing infrastructure overhead and improving workplace safety compliance.
- Cost Reduction in Manufacturing: The economic benefits extend beyond simple reagent costs to encompass significant savings in downstream processing. Because the reaction does not generate heavy metal waste streams requiring specialized treatment or expensive scavenging resins, the environmental compliance costs are substantially lowered. The simplified workup procedure, often involving basic aqueous extraction and crystallization, reduces solvent consumption and energy usage compared to the multi-step purifications needed for transition-metal catalyzed reactions. This holistic reduction in operational complexity allows for a leaner manufacturing footprint, enabling facilities to produce high-purity pharmaceutical intermediates with a smaller environmental impact and lower utility consumption, directly contributing to improved profit margins.
- Enhanced Supply Chain Reliability: Supply chain continuity is critically dependent on the availability of key starting materials. The reagents required for this method—halogenated aromatics, zinc chloride, and common amines—are commodity chemicals produced at massive scales globally, ensuring a diversified and secure supply base. This stands in contrast to proprietary ligands or custom-synthesized catalysts that may have single-source suppliers and long lead times. By relying on widely available commodities, manufacturers can mitigate the risk of production stoppages due to raw material shortages. Furthermore, the stability of the zinc-amine complexes allows for longer shelf-life and bulk purchasing opportunities, enabling strategic stockpiling without the degradation concerns associated with sensitive catalytic systems.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden inefficiencies, but the robust nature of this zinc-mediated chemistry makes it highly amenable to commercial scale-up. The reaction tolerates a broad range of temperatures and concentrations, providing process engineers with wide operating windows to optimize throughput. From an environmental standpoint, the avoidance of toxic tin reagents (as seen in Migita couplings) and the reduction of heavy metal load align with increasingly stringent global environmental regulations such as REACH and TSCA. This proactive compliance posture minimizes the risk of regulatory fines and facilitates smoother audits from international clients who prioritize green chemistry principles in their supplier selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aromatic amine synthesis technology. These answers are derived directly from the experimental data and claims presented in the underlying patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific project needs. Understanding these nuances is essential for making informed decisions about process development and vendor qualification.
Q: What are the primary advantages of using organic amine zinc chloride complexes over traditional palladium catalysts?
A: The use of organic amine zinc chloride complexes eliminates the need for expensive palladium catalysts and toxic organophosphorus ligands. This significantly reduces raw material costs and simplifies the purification process by avoiding heavy metal residue removal, making it ideal for large-scale pharmaceutical intermediate production.
Q: Does this method require harsh reaction conditions compared to classical nucleophilic substitution?
A: No, the method operates under relatively mild conditions ranging from 60°C to 200°C depending on the substrate reactivity. Unlike classical methods that often require extreme temperatures or strong electron-withdrawing groups exclusively, this zinc-mediated approach offers broader substrate tolerance and improved safety profiles.
Q: Is the organic amine zinc chloride complex stable for long-term storage and transport?
A: Yes, one of the key innovations is that the amination reagent exists as a stable solid complex. This avoids the handling issues associated with volatile, malodorous, or hazardous liquid and gaseous free amines, thereby enhancing supply chain reliability and workplace safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Amines Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to innovative synthetic methodologies requires a partner with both technical expertise and proven manufacturing capacity. As a leading CDMO and supplier, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this zinc-complex technology are fully realized in a commercial setting. Our rigorous QC labs and stringent purity specifications guarantee that every batch of aromatic amines meets the exacting standards required for pharmaceutical and agrochemical applications. We are committed to leveraging advanced green chemistry techniques to deliver high-purity pharmaceutical intermediates that drive efficiency and sustainability for our global partners.
We invite you to engage with our technical procurement team to discuss how this novel synthesis route can be tailored to your specific molecule requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a reliable pharmaceutical intermediates supplier dedicated to your long-term success.
