Advanced Non-Halogen Synthesis Of Substituted Aromatic Amines For Commercial Scale
The chemical landscape for producing high-value aromatic amines is undergoing a significant transformation, driven by the urgent need for more sustainable and economically viable manufacturing processes. Patent CN1084727C introduces a groundbreaking methodology for the preparation of substituted aromatic amines, specifically targeting derivatives of 4-aminodiphenylamine (4-ADPA), which are critical precursors in the synthesis of antioxidants, antiozonants, and various pharmaceutical intermediates. This technology represents a paradigm shift away from traditional halogen-dependent routes, offering a cleaner, more efficient pathway that aligns with modern green chemistry principles. By leveraging the reactivity of substituted aromatic azo compounds in the presence of alcohols and suitable bases, this process eliminates the need for corrosive halogenated reagents and the associated waste disposal burdens. For industry leaders seeking a reliable pharmaceutical intermediates supplier, understanding the nuances of this patent is essential for optimizing supply chains and reducing long-term production costs. The technical depth of this invention provides a robust foundation for scaling complex organic syntheses while maintaining stringent purity standards required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted aromatic amines like 4-ADPA has relied heavily on nucleophilic aromatic substitution mechanisms involving halogenated starting materials. These conventional pathways typically require the reaction of aniline derivatives with halogenated nitrobenzenes, a process that generates substantial quantities of halogenated salt waste. The presence of halogens in the reaction mixture poses severe challenges, including significant corrosion of reaction apparatus, which necessitates the use of expensive, specialized equipment capable of withstanding harsh chemical environments. Furthermore, the disposal of halogenated waste streams is not only environmentally hazardous but also incurs substantial financial costs due to strict regulatory compliance requirements. Traditional methods often involve multiple steps, including separate hydrogenation stages to reduce nitro groups, which adds complexity and increases the overall energy consumption of the manufacturing process. The reliance on specific halogenated precursors also introduces supply chain vulnerabilities, as the availability and price volatility of these raw materials can impact production continuity. Consequently, the industry has long sought alternative routes that can bypass these inherent limitations while delivering high-purity products suitable for sensitive applications in the pharmaceutical and fine chemical sectors.
The Novel Approach
The innovative process described in the patent offers a compelling solution by utilizing substituted aromatic azo compounds as key intermediates, which are reacted with alcohols under basic conditions to yield the desired aromatic amines. This non-halogen method fundamentally alters the reaction mechanism, avoiding the formation of corrosive byproducts and simplifying the purification workflow. By selecting appropriate alcohols, such as aliphatic, cycloaliphatic, or aralkyl alcohols, the process can be tuned to produce specific alkylated derivatives directly, potentially eliminating the need for subsequent alkylation steps. The reaction conditions, typically ranging from 70°C to 200°C, are compatible with standard industrial reactors, reducing the capital expenditure required for specialized corrosion-resistant infrastructure. This approach not only enhances the economic feasibility of producing high-purity pharmaceutical intermediates but also significantly improves the environmental profile of the manufacturing operation. The ability to generate valuable diamines and their derivatives through a streamlined, one-pot or reduced-step sequence represents a major advancement in process chemistry, offering manufacturers a strategic advantage in cost reduction in pharmaceutical intermediates manufacturing. This novel route ensures a more stable supply of critical raw materials while adhering to increasingly stringent global environmental standards.
Mechanistic Insights into Azo Compound Reduction and Alkylation
The core of this technology lies in the unique reactivity of the azo functional group when exposed to nucleophilic attack by alcohols in a basic environment. The reaction mechanism involves the reduction of the azo linkage (-N=N-) coupled with the transfer of alkyl groups from the alcohol to the nitrogen atoms, resulting in the formation of amine bonds. This transformation is facilitated by the presence of strong bases, such as alkali metal hydroxides or alkoxides, which activate the alcohol and promote the cleavage of the azo bond. The versatility of this method is evident in the wide range of substituents that can be accommodated on the aromatic rings, allowing for the synthesis of diverse derivatives tailored to specific application requirements. The patent details various structural formulas for the starting azo compounds, highlighting the flexibility of the system to incorporate different electronic and steric properties. 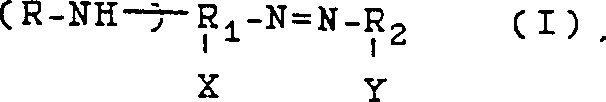
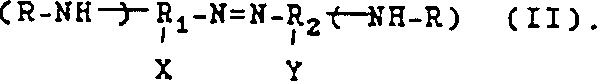 Understanding the electronic effects of substituents X and Y on the aromatic rings is crucial for optimizing reaction rates and selectivity. Electron-withdrawing groups can enhance the electrophilicity of the azo nitrogen, facilitating nucleophilic attack, while electron-donating groups may require adjusted reaction conditions to achieve comparable conversion levels. This deep mechanistic understanding allows chemists to fine-tune the process for maximum efficiency, ensuring that the commercial scale-up of complex pharmaceutical intermediates proceeds with minimal deviation from laboratory-scale results.
Understanding the electronic effects of substituents X and Y on the aromatic rings is crucial for optimizing reaction rates and selectivity. Electron-withdrawing groups can enhance the electrophilicity of the azo nitrogen, facilitating nucleophilic attack, while electron-donating groups may require adjusted reaction conditions to achieve comparable conversion levels. This deep mechanistic understanding allows chemists to fine-tune the process for maximum efficiency, ensuring that the commercial scale-up of complex pharmaceutical intermediates proceeds with minimal deviation from laboratory-scale results.
Impurity control is a critical aspect of this synthesis, particularly given the stringent quality requirements for pharmaceutical and high-performance additive applications. The patent emphasizes the importance of controlling the amount of proton transfer materials, such as water, within the reaction system. Excessive water can lead to the hydrolysis of intermediates or the deactivation of the base, resulting in lower yields and the formation of unwanted byproducts. By employing drying agents or azeotropic distillation techniques to maintain low water content, the process ensures high selectivity towards the desired aromatic amine products. Additionally, the choice of solvent plays a pivotal role in managing impurity profiles; aprotic polar solvents can be used to dissolve reactants effectively without interfering with the reaction mechanism. The ability to recycle byproducts, such as amides formed in certain embodiments, further enhances the atom economy of the process. This rigorous approach to impurity management guarantees that the final product meets the high-purity standards expected by R&D directors and quality assurance teams. The detailed guidance on reaction parameters provides a clear roadmap for maintaining consistent product quality across different production batches, thereby reinforcing supply chain reliability.
How to Synthesize 4-Aminodiphenylamine Derivatives Efficiently
Implementing this synthesis route requires careful attention to reaction conditions and reagent selection to maximize yield and purity. The process begins with the preparation of the substituted aromatic azo compound, which can be generated in situ or used as a pre-formed intermediate. Following this, the azo compound is combined with the chosen alcohol and base in a suitable reactor system. The temperature must be carefully controlled within the specified range to ensure complete conversion while preventing thermal degradation of sensitive functional groups. Monitoring the reaction progress via techniques such as HPLC allows for precise determination of the endpoint, ensuring that resources are not wasted on over-processing. The patent provides specific examples of molar ratios and catalyst loadings that serve as a starting point for process optimization. For those seeking to adopt this technology, it is essential to validate these parameters against specific substrate combinations to account for steric and electronic variations. The following guide outlines the standardized steps for executing this synthesis, providing a foundation for technical teams to develop robust manufacturing protocols. Detailed standardized synthesis steps are provided in the guide below.
- Combine substituted aromatic azo compounds with aliphatic or aralkyl alcohols in the presence of a suitable base.
- Maintain reaction temperature between 70°C and 200°C to facilitate the reduction and alkylation process.
- Control proton transfer material levels to optimize yield and minimize side reactions during the synthesis.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this non-halogen synthesis route offers substantial benefits for procurement and supply chain management. The elimination of halogenated reagents directly translates to a reduction in raw material costs associated with waste treatment and equipment maintenance. Traditional processes often require significant investment in corrosion-resistant reactors and specialized waste disposal services, which can erode profit margins. By switching to this cleaner technology, manufacturers can achieve significant cost savings in pharmaceutical intermediates manufacturing without compromising on product quality. Furthermore, the simplified reaction sequence reduces the overall production time, allowing for faster turnaround on orders and improved responsiveness to market demand. The use of readily available alcohols and bases ensures a stable supply of raw materials, mitigating the risk of shortages that can disrupt production schedules. This stability is crucial for supply chain heads who are tasked with ensuring continuous availability of critical intermediates for downstream customers. The scalability of the process means that production volumes can be adjusted flexibly to match market fluctuations, providing a competitive edge in a dynamic global market.
- Cost Reduction in Manufacturing: The primary economic driver for this technology is the removal of expensive halogen removal and waste disposal steps. Traditional halogenated routes generate large volumes of saline waste that require costly treatment before discharge. By avoiding halogens entirely, this process eliminates these downstream costs, leading to a leaner cost structure. Additionally, the reduced corrosion extends the lifespan of manufacturing equipment, lowering capital replacement expenditures over time. The ability to use alcohols as both reactants and solvents further simplifies the material balance, reducing the need for additional solvent recovery systems. These cumulative efficiencies result in a more competitive pricing model for the final aromatic amine products, making them attractive to cost-sensitive buyers in the pharmaceutical and agrochemical sectors.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by the use of commodity chemicals as primary feedstocks. Alcohols and common bases are widely available from multiple global suppliers, reducing dependency on single-source vendors for specialized halogenated intermediates. This diversification of the supply base minimizes the risk of disruptions caused by geopolitical issues or production outages at specific facilities. The robust nature of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output even when sourcing from different suppliers. For procurement managers, this reliability translates to more predictable lead times and reduced inventory holding costs. The ability to maintain steady production flows is essential for meeting the just-in-time delivery requirements of major pharmaceutical clients, thereby strengthening long-term business relationships.
- Scalability and Environmental Compliance: Scaling this process to industrial levels is straightforward due to the use of standard unit operations and benign reagents. The absence of hazardous halogenated waste simplifies the permitting process for new production facilities, accelerating time-to-market for new capacity. Environmental compliance is easier to achieve, as the waste streams are less toxic and easier to treat, aligning with corporate sustainability goals. This environmental advantage is increasingly important for companies facing pressure from investors and regulators to reduce their carbon footprint and chemical impact. The process design supports continuous manufacturing possibilities, which can further enhance efficiency and reduce the physical footprint of the production plant. These factors combined make the technology a future-proof solution for growing demand in high-performance chemical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. They are intended to clarify the operational feasibility and strategic benefits of adopting this non-halogen route for aromatic amine production. Understanding these details is vital for decision-makers evaluating the potential integration of this technology into their existing manufacturing portfolios. The insights provided here bridge the gap between theoretical chemistry and practical industrial application, ensuring that stakeholders have a clear understanding of the value proposition.
Q: What are the primary advantages of this non-halogen method over traditional nucleophilic substitution?
A: The primary advantage is the elimination of halogenated waste streams and equipment corrosion. Traditional methods using halogenated intermediates require expensive waste disposal and corrosion-resistant reactors. This patent describes a route using azo compounds and alcohols, which significantly simplifies the downstream processing and reduces environmental compliance costs.
Q: Can this process be scaled for industrial production of 4-ADPA derivatives?
A: Yes, the patent explicitly outlines conditions suitable for industrialization. The use of common alcohols as solvents or reactants, combined with standard bases like potassium hydroxide or alkoxides, allows for straightforward scale-up. The process avoids complex hydrogenation steps in certain embodiments, further enhancing its feasibility for large-scale manufacturing.
Q: How does the control of proton transfer materials impact the reaction yield?
A: Controlling the amount of proton transfer material, such as water, is critical for maximizing yield. The patent indicates that minimizing water content or removing it continuously via azeotropic distillation can drive the reaction towards higher conversion rates. This precise control ensures that the base remains active and prevents the hydrolysis of sensitive intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Aminodiphenylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes for high-value chemical intermediates like 4-aminodiphenylamine. Our technical team has extensively analyzed the potential of the non-halogen methods described in patent CN1084727C and is well-equipped to translate this intellectual property into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly converted into industrial output. Our facilities are designed to handle complex organic syntheses with stringent purity specifications, supported by rigorous QC labs that verify every batch against global standards. We understand that the transition to new manufacturing technologies requires a partner with deep technical expertise and a commitment to quality. Our CDMO capabilities allow us to adapt this specific azo-reduction chemistry to meet your unique volume and purity requirements, providing a secure source for your supply chain.
We invite you to collaborate with us to optimize your supply chain for aromatic amines and related derivatives. Our team is ready to conduct a Customized Cost-Saving Analysis to quantify the potential economic benefits of switching to this non-halogen process for your specific application. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project needs. By leveraging our expertise in process development and scale-up, we can help you reduce lead time for high-purity pharmaceutical intermediates and secure a competitive advantage in the market. Let us partner with you to drive innovation and efficiency in your chemical manufacturing operations, ensuring a reliable and cost-effective supply of critical materials for your global business.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
